-
Our Products
- Medical Equipment
- Beauty Equipment
- Wood Black Portable Massage Bed Foldable, For Spa, 12 kg
- wooden portable therapy bed portable massage table for travel
- portable massage table foldable tattoo bed wooden spa table
- wooden massage table foldable tattoo bed spa table with carry case
- wooden portable massage table professional portable therapy bed
- Folding Massage Table 84'' Professional Massage Bed 2 Fold Lash Bed with Head-& Armrest, Black
- Facial Massage Bed Folding Massage Table Beauty Massage Spa Bed Spa Massage Bed
- wooden massage bed with armrests portable therapy bed with adjustable features
- portable wooden therapy bed with carry case 2-section portable therapy table
- portable treatment table wooden massage bed with carry bag
- foldable therapy bed portable treatment table ergonomic design sturdy frame
- wooden portable massage bed with accessories lightweight therapy bed
- Massage bed therapy table salon bed 2-section massage table
- foldable massage bed for home portable therapy table with headrest
- wooden portable beauty bed with carry case portable therapy table for tattoo studios
- portable beauty bed with high load capacity massage table for portable use
- portable therapy bed for beauty treatments wooden massage bed for professional salons
- portable massage bed for mobile therapists foldable wooden spa massage table
- portable tattoo bed with adjustable headrest wooden massage table with carry case
- foldable wooden beauty bed professional portable massage bed
- Portable Foldable Massage Table Bed Spa Salon Bed with Carry Case 2 Fold
- Portable 2 Fold Massage Reiki Facial Table Bed (Black) Carrying Bag & Headrest, Armrests (Black)
- Massage Bed Table Portable Foldable Wooden for Spa Tattoo Parlor (2-Section Wooden)
- Wooden Massage Bed, For Spa, 15 kg
- Wood Portable Massage Table Folding Massage Table 2 section, For Parlour
- wooden portable table for beauty treatments portable spa table with ergonomic design
- Portable Folding Lightweight Foam Padding Leather Cover Massage Bed with Carrying Bag (Black)
- Foam Black Portable Massage Bed, For Spa, 9 kg
- massage table for therapists foldable massage bed for salons
- massage table with 250kg load capacity foldable spa bed
- carry bag 250kg capacity adjustable headrest portable spa bed
- Bed for massage therapists portable treatment table with sturdy design
- spa bed with carry case portable tattoo bed with strong support
- professional massage table massage table for therapists
- Biotronix Portable Sauna Heating IR Blanket 2 Zone
- Vacuum Therapy Slimming Equipment
- 5 in 1 Cavitation RF Vacuum Slimming Machine
- Ultrasonic Cavitation RF Vacuum and LIPO LASER
- Adjustable Professional Physical Multifunctional Spa Beauty Table Salon bed Massage Couch Massage
- Portable Massage Table Therapy Bed with Carrying Bag for Spa Salon Clinic Lash Tattoo Shop
- foldable therapy bed for home use portable massage table with armrests
- portable tattoo bed with adjustable headrest wooden massage table with carry case
- Slimming Equipment
- 6 in 1 Cavitation Multipolar RF Vacuum and Laser Therapy Slimming Machine
- Electrodes Reusable Sticking Pads For Tens
- Cavitation Slimming Device with Radio Frequency RF and Vacuum Fat loss Device
- 5 in 1 Lipo Ultrasonic Cavitation RF and Vacuum Therapy Weight Loss Device
- Ultrasonic Cavitation Slimming Equipment
- 3 In 1 OPT SHR Q Switch ND YAG Laser
- Biotronix Cavitation 5 in 1 Body Massage Shaping Machine with RF Vacuum
- Self Adhesive Electrode Pads Corded Electric Electronic Stimulators IFT Tens Ems Pulse Massagers
- Chiropractor Tools
- chiropractic adjustment gun, electric chiropractic gun, 400N force chiropractic
- Next-Generation Chiropractic Therapy Electrical 400N Force Gun
- 400N Digital Spine Adjustment Tool
- Digital 4 Levels Chiropractor Gun Electrical Spine Adjuster Force Gun
- Superior Spine Alignment: Digital Chiropractor Gun with 400N Force, For Electric Chiropractic Tool
- Chiropractor Gun Transform Your Practice with Digital Spine Adjustor and 400N Force
- Revolutionize Chiropractic Gun Digital Spine Adjustor with 400N Force
- Chiropractor Gun Empower Your Practice Electrical 400N
- PlasticsABS Flexible Optimal Patient Comfort Electrical 400N Force Chiropractic Tool
- Precision Chiropractic Adjustments with 400N Digital Spine Adjustor
- Plastics ABS Efficient Chiropractic Adjustments Electrical 400N Force Gun
- Chiropractic adjustment Analogue chiropractor gun 6-gear chiropractor tool 300N chiropractor gun
- Plastics ABS Chiropractor Gun Precision Chiropractic Care Electrical 400N Force Spine Adjustor
- Digital Spine Manipulation Tool Electrical Chiropractor tool 400N
- Electric chiropractic gun Chiropractic device 400N activator
- ABS Plastic Chiropractor Gun Precision Chiropractic Care: Electrical 400N Force Spine Adjustor
- Plastics ABS Chiropractor Gun Chiropractic Impulse Adjustor 400N
- Electric chiropractic tool for spine 400N force adjusting device
- Chiropractic Adjusting Tool,6 Levels Manual Spine Adjusting Corrector Tool (4 Heads)
- Manual spine therapy Back pain relief tool Spine alignment tool
- Spine Chiropractor Professional Spine Adjusting Massager Manual Spine Chiropractic Gun
- Chiropractic Adjusting Tool 6Levels Manual Back Spine Adjuster Adjusting Corrector
- Transformative Chiropractic Therapy Electrical 400N Force Gun
- Chiroprator Gun Manual Chiropractic Tool 300N 6 gears 4 Heads Spine Adjuster Correction
- Manual spine adjuster Spinal adjustment tool Chiropractor adjustment tool Chiropractic instrument
- Plastics ABS Innovative Chiropractic Solutions Electrical Gun with 400N Force
- Chiropractic Impulse Gun Digital Spine Adjustor Chiropractic correction tool 400N
- Spine Activator Massager Machine LED Display Chiropractic Correction Gun 400N
- Biotronix 300N Manual Chiropractor Gun with 6 Gears 50N-300N Adjustable Manually Analogue
- Electric chiropractic tool for spinal adjustment 400N
- Chiropractic Adjusting Tool 6 Levels Manual Spine Chiropractic Adjustment Correction Gun
- Chiropractic instrument Electric spine tool 400N adjustment
- Chiropractic gun for spine care Manual spine adjustment equipment
- Chiropractor Chiropractic Gun Manual Analogue 6 Levels 300N Spine Adjustor Correction Gun
- Chiropractor Gun 400N Force Spine Correction Device, For Adjustable Chiropractic Stool
- Impulse Generator Manual Spine Therapy Chiropractor Gun Spine Adjustor Correction Manual
- Biotronix IASTM Tools kit 5 pc Imported Myofascial Release with Carry Case
- Chiropractor Spine Adjuster Gun 400N, For Comprehensive Chiropractic Kit
- Handy Handheld Portable Chiropractor Adjustment Tool 6 Gears 300N Manual type
- Digital Force Chiropractor Tool Chiropractic gun 400 N for Spinal Adjustments
- Chiropractor Gun Manual Spine Correction Manual Analogue 6 Gears 300N
- Stainless Steel Gua Sha Scraping Massage Tool IASTM Tools Help Relieve Sore Muscles
- chiropractic force adjuster chiropractic adjusting instrument
- Plastics ABS Maximize Efficiency: Electrical Chiropractic Gun with 400N Force
- Chiropractic back adjustment tool Manual chiropractic equipment
- Plastics ABS Chiro Gun 400N Electrical Model 4 LEvels
- Electric chiropractic gun Chiropractic device 400N activator
- Biotronix Spine Gun Alignment Tool Manual Analogue Model 300N Chiropractor Gun
- Handheld Manual Chiropractor Gun Spine Adjustor Tool Manual Type Analogue 6 Levels
- Chiropractic Adjuster Gun Manual 6 Levels Gears Adjustable Force 50N-300N
- Levels Manual Spinal Portable Chiropractic Adjustment Kit Spine Back Tool Improve Joint
- Chiropractic spine adjustment tool
- Chiropractor Physiotherapy Gun 300N Chiro Spine Therapy Device 6 Levels
- Chiropractor Gun Precision Treatment Digital Spine Adjustor with 400N Force
- Plastics ABS Exercise Equipment Digital Chiropractor Instrument for Chiropractic Gun 400N
- Chiropractic tool 400N adjuster Electric chiropractic
- Plastics ABS Chiropractic Impulse Gun 400N
- Chiropractic Adjusting Tool Correction Gun Spine Therapy Impulse Device 4 Level 6 Heads 400N
- Chiropractic Spine Adjuster Gun 400N
- Plastics ABS Chiropractor Gun Empower Your Practice Electrical 400N Force Spine Adjustor
- Chiropractor Gun Electric 400N Spine Adjustor 4 Levels Digital 9 Heads
- Biotronix Chiropractic Corrector Spine Gun 6 Levels Manual Chiropractic Adjusting Tool 4Heads
- 400N Force Chiropractic Manipulation Tool Gun Electrical
- electric chiropractic adjustment device chiropractic manipulation tool
- Chiropractic Adjusting Tool 400N
- Chiropractor gun Spine correction tool Chiropractic gun Manual chiropractor tool
- Biotronix Chiropractic Gun,Chiropractic Adjusting Tool Instrument Electric Impulse Gun GINHA HL750
- Iastm Tools Gua Sha Tools Graston Tools 5 pc Imported Stainless Steel with Carry Case
- Biotronix SS 304 IASTM GuaSha Scraping Massage Tools,5 Pc Soft Tissue Mobilization PhysicalTherapy
- Tool IASTM Tools Help Relieve Sore Muscles
- ergonomic IASTM set imported muscle therapy tools
- Electric adjustment tool Chiropractic gun 400N Electric chiropractor activator
- 400N chiropractic equipment Electric chiropractor device 400N spine adjuster
- 400N chiropractor equipment Electric chiropractic instrument Chiropractic activator device
- Electric chiropractic gun 400N adjustment device Chiropractic adjuster electric
- Electric chiropractor tool 400N spine tool Chiropractic tool electric
- Plastics ABS Chiropractor Gun 400N Force Spine Adjustor
- electric chiropractic tool chiropractic gun
- Chiropractic device 400N 400N chiropractic adjuster tool Electric chiropractor adjuster tool
- Electric activator tool Chiropractic device tool
- 400N chiropractic activator Electric chiropractic adjusting
- 400N force tool Electric adjuster 400N chiropractor
- Electric spine adjuster Chiropractic tool 400N 400N adjustment tool
- Chiropractic gun Spine adjuster Chiropractic activator
- Plastics ABS Exercise Equipment 400N Electric Chiropractic Gun Digital Adjuster
- Plastics ABS Spine Adjustor Gun Electric Chiropractic Adjusting Tool 400N
- Chiropractic tool 400N electric Electric chiropractic adjusting tool
- 400N electric chiropractic instrument Chiropractic adjusting device electric
- Electric Chiropractor Gun Digital Chiropractic Tool Adjustable Force Spine Adjustor 400N
- 400N Chiropractic Adjusting Tool Physical Therapy Device Chiropractic Activator Massage Gun
- Electric chiropractic adjustment device for spine 400N instrument electric
- Plastics ABS Exercise Equipment Advanced Precision Electrical 400N Force Chiropractor Gun
- Achieve Optimal Results: Electrical 400N Force Chiropractic Tool Digital
- 400N electric chiropractic activator device
- Next-Level Chiropractic Care Electrical 400N Force Impulse Gun, For Digital Spine Realignment Tool
- Chiropractic Adjusting Tool Instrument Electric Impulse Gun Actuator 400N
- Experience Superior Chiropractic Therapy with Electrical 400N Force Gun
- Impulse Gun Chiropractor Small Electronic Vibration High Pressure Massage Fascia Instrument 400N
- Plastics ABS Exercise Equipment Digital Electric Chiropractic Gun 400N Adjuster
- 400N electric chiropractic activator instrument
- Chiropractic Equipment with 400 N Force Electric Spine Adjustor Gun for Chiropractors
- High-Force Chiropractic Tool 400N Electric Spine Adjustor with Digital control
- spinal therapy gun chiropractic adjuster
- chiropractor adjuster chiropractic gun 400N
- chiropractor force adjustment gun
- electric adjustment gun spine adjustment tool
- electric chiropractic adjustment equipment
- chiropractor gun chiropractic tool
- chiropractor tool spinal adjustment gun
- 400 N chiropractor tool adjustable chiropractic gun
- hiropractic instrument for professionals high-force chiropractic tool
- professional chiropractic tool chiropractic treatment gun spinal adjustment tool
- electric chiropractic gun chiropractic adjusting tool chiropractor adjustment device
- electric chiropractor tool 400 N chiropractic gun adjustable force chiropractic tool
- Exercise Equipment Chiro Gun 400 N Digital Electrical 4 Gear 4 LEvels 6 Heads, For Handheld Chiropractic Device
- Chiropractor Gun 400 N Digital Physiotherapy Device
- Electrical Impulse Gun 400N, For Chiropractic Tool Kit
- Maximize Efficiency Electrical 400N Force Chiropractor Gun Digital
- Chiropractor Gun 400 N, For Chiropractic Positioning Tools
- Innovative Solutions Digital Chiropractor Gun with 400N Force, For Chiropractic Spine Adjuster
- Optimal Spine Alignment Electrical 400N Force Chiropractic Gun
- Elevate Your Practice: Electrical 400N Force Chiropractic Impulse Gun
- Advanced Chiropractic Impulse Gun for Precise Spine Correction 400 N
- Biotronix 6 Gears 300N Manual type Handy Handheld Portable Chiropractor Adjustment Tool
- Spine Adjuster Correction Setting Chiropractor Professional Gun
- Electric 750N GINHA HL750 Chiropractor Gun
- Air Compression Recovery Boots Leg Massager
- SS 304 IASTM GuaSha Scraping Massage Tools
- chiropractic force adjuster chiropractic adjusting instrument
- 300N Spine Adjustor Tool Gun manual Type Chiropractor Spine Adjustment Gun 6 Gears
- Spine Adjustor Correction Alignment Gun Manual with Carry Case 4 Heads 300N
- Chiropractic gun for spine care Manual spine adjustment equipment
- Stainless Steel Gua Sha Scraping Massage Tool Set IASTM Tools Great Soft Tissue Mobilization Tool
- Physiotherapy Equipment
- Dvt Remote Intermittent Pneumatic Compression Therapy Unit with Remote
- Konmed KM-560 4 Channel Biofeedback Device with Muscle Recovery Modes
- Konmed KM-560 EMG Biofeedback System with Advanced Rehab Technology
- Advanced Brainwave And Neural Modulation System
- Konmed KM-560 EMG Biofeedback Device with Therapy Control Settings
- Konmed KM-560 Physiotherapy Biofeedback Equipment with EMG Training
- Phymaster Shockwave Therapy Machine for Non-Invasive Pain Therapy
- Konmed KM-560 EMG Biofeedback System for Muscle Re-education Therapy
- Pelvic Floor Muscle Recovery Chair for Women & Men Rehabilitation
- Konmed KM-560 EMG Biofeedback Device for Hospital Physiotherapy Setup
- Medical Grade Pelvic Muscle Training Chair for Urinary Incontinence Care
- Advanced Class 4 Laser Therapy System for Professional Physiotherapy Practice
- Konmed KM-560 Biofeedback Equipment with Therapy Customization
- Back, Leg and Chest Dynamometer for Accurate Rehab Progress Tracking
- Konmed KM-560 EMG Biofeedback Device with Multi Therapy Settings
- Professional Strength Testing Equipment-Back Leg Chest Dynamometer
- Physiotherapy Strength Evaluation Tool Back, Leg & Chest Dynamometer
- Efficient Motorized Ankle CPM With Quiet Operation And Smooth Movement
- Konmed KM-560 Professional Biofeedback Machine for Advanced Rehab
- Phymaster Shockwave Shockwave Therapy Machine for Physiotherapy Centers
- KM521 Digital Pelvic Muscle Training Device with TFT Screen
- Pelvic Floor Muscle Therapy Chair for Women's & Men's Health
- Clinical Stroke Rehabilitation And Brain Activation Machine
- IASTM Tools 5 Pc IASTM for Physiotherapy, Chiropractor ,Technique & Osteopath
- KM521 Pelvic Muscle Rehab Device for Post Delivery & Surgery Recovery
- Portable Brain Therapy Equipment tDCS tPCS tACS tVNS
- KM521 Advanced Pelvic Floor Muscle Rehab Machine with Preset Programs
- KM521 Smart Pelvic Muscle Trainer for Strength, Control & Recovery
- Back, Leg and Chest Dynamometer for Functional Capacity Evaluation
- Konmed KM-560 Rehab Biofeedback Equipment with EMG
- Advanced Muscle Strength Testing System-Back, Leg & Chest
- Medical Strength Testing Equipment-Back, Leg & Chest Dynamometer
- KM521 Rechargeable Pelvic Floor Muscle Training Device for Home Use
- Back, Leg and Chest Muscle Measurement Dynamometer for Medical Use
- Smart Tecar RF Therapy Machine for Pain Relief and Rehabilitation, For Sports Injury Recovery
- Exoskeleton Rehabilitation Technology for Consistent Therapy Results
- Smart Digital Pelvic Floor Muscle Therapy Chair for Pelvic Health Clinics
- Economical Class 4 Laser Therapy Equipment for Orthopedic Clinics
- Professional Class IV Laser Therapy Machine for Physiotherapy Treatment
- High Power Class 4 Laser Therapy System for Pain Relief Clinics
- Economical Model Class IV Laser Therapy Equipment for Rehabilitation Clinics
- High Intensity Class 4 Laser Physiotherapy Machine for Sports Injury Treatment
- Class IV Laser Therapy Equipment for Pain Relief and Recovery Centers
- High Power Class IV Laser Therapy Equipment for Physiotherapy Rehabilitation
- Economical Class 4 Laser Therapy Machine for Pain Relief Treatment Clinics
- Professional Class 4 Laser Physiotherapy Device for Orthopedic Pain Relief
- Class IV Laser Therapy Machine for Sports Injury Recovery Clinics
- Physiotherapy Professional Dual Probe Laser Pain Relief Device LCD Display 46 Pre Programmed Digita
- Professional Neuro Therapy Machine tDCS tPCS tACS tVNS
- PEMF Loop Therapy Machine Premium Physiotherapy Equipment
- PEMF Loop Therapy Device for Orthopedic Pain Treatment
- Pulse Electromagnetic Therapy Machine for Joint Pain Relief
- PEMT Loop Therapy Device for Sports Injury Recovery
- Electromagnetic Pulse Therapy Machine for Pain Relief Therapy
- Pulse Electromagnetic Field Therapy Loop Machine Clinical Model
- PEMT Loop Therapy Machine for Rehabilitation and Pain Management
- Premium PEMF Loop Therapy Machine for Pain Relief Treatment
- PEMT Pulse Electromagnetic Therapy Loop Device for Physiotherapy Clinics
- PEMT Loop Therapy Machine Advanced Physiotherapy Equipment
- High Power Class 4 Laser Physiotherapy Equipment
- Economical Class 4 Laser Therapy Machine for Rehabilitation
- Professional Class IV Laser Physiotherapy Machine
- High Power Class 4 Therapy Laser Machine
- Class 4 Laser Therapy Device for Physiotherapy Clinics
- Class IV Laser Therapy Device for Pain Management
- Class 4 Laser Therapy System for Sports Injury Recovery
- High Power Class 4 Laser Therapy Equipment for Physiotherapy
- Clinical Class IV Laser Therapy Device for Pain Treatment
- Class 4 Laser Physiotherapy Equipment Economical Model
- Professional Class IV Laser Pain Relief Therapy Machine
- Economical Class 4 Laser Therapy Equipment for Physiotherapy Clinics
- Medical Class IV Laser Therapy Equipment for Muscle Pain Relief
- Class 4 Laser Therapy Machine for Sports Injury Treatment
- Clinical Class IV Laser Physiotherapy Equipment for Pain Therapy
- Economical Model Class 4 Laser Pain Relief Therapy Device
- Professional Class 4 Laser Physiotherapy Device for Rehabilitation
- Class IV Laser Therapy System for Muscle and Joint Pain
- Professional High Power Class IV Laser Therapy Equipment
- Medical Grade Class IV Laser Therapy Equipment for Rehabilitation
- Economical Class 4 Laser Therapy Device for Clinics and Hospitals
- Professional Class IV Laser Machine for Physiotherapy Treatment
- Class IV Laser Therapy Machine for Joint Pain Treatment
- High Power Class 4 Laser Physiotherapy System for Clinics
- Advanced Class IV Laser Therapy Equipment for Pain Relief
- Economical Model Class 4 Laser Therapy Equipment for Hospitals
- Economical Class IV Laser Therapy Machine for Physiotherapy Clinics
- High Power Physiotherapy Laser Therapy Machine Class IV
- Class 4 Laser Therapy Equipment for Sports Injury Recovery
- Professional Class IV Laser Physiotherapy Equipment for Clinics
- High Power Class 4 Laser Therapy Device for Pain Relief Treatment
- Clinical Class IV Laser Therapy Device for Rehabilitation Clinics
- Economical Class 4 Laser Physiotherapy Machine for Pain Relief
- Advanced Class 4 Laser Therapy System for Pain Relief Clinics
- Economical Class IV Laser Machine for Physiotherapy Treatment
- High Intensity Class IV Laser Therapy Machine for Pain Management
- Class 4 Laser Pain Relief Machine for Physiotherapy Centers
- High Power Class IV Laser Therapy Equipment for Orthopedic Clinics
- Professional Class 4 Laser Therapy Device for Rehabilitation Clinics
- Medical Grade Class 4 Laser Physiotherapy Machine for Pain Therapy
- Class 4 Laser Therapy Equipment for Physiotherapy and Sports Clinics
- Class IV Laser Physiotherapy Device for Orthopedic Pain Relief
- Economical High Power Laser Therapy Machine for Rehabilitation
- Class 4 Laser Therapy Device for Muscle and Joint Pain Treatment
- Class IV Laser Therapy Equipment for Sports Rehabilitation
- Economical Model Class IV Laser Therapy Machine for Pain Relief
- High Power Physiotherapy Laser Therapy Device Class 4
- Economical Class 4 Laser Therapy Device for Pain Relief Clinics
- Class IV High Power Laser Therapy Equipment for Physiotherapy
- Professional Class 4 Laser Machine for Physiotherapy Rehabilitation
- Advanced Class IV Laser Therapy Equipment for Pain Treatment
- Phymaster Shockwave Physiotherapy Machine for Pain Relief Therapy
- Smart Neuro Stimulation System tDCS tACS tVNS tPCS
- UltraPrecision Focus Shockwave System for Professional Rehabilitation Use
- Rechargeable Dual Wavelength Laser Therapy Device 650nm + 808nm with Cooling Fan
- Advanced Pelvic Muscle Re-Education Chair for Physiotherapy Clinics
- 4 in 1 Digital Electrotherapy Unit for Muscle Recovery
- Exoskeleton-Based Physiotherapy for Neuromuscular Control
- Professional Physio Laser Therapy 910nm Laser Pain Therapy Device
- Portable & Robust PEMT Loop Therapy Device for Clinics & Rehab Centers
- Portable Laser Therapy Device-Fan-Cooled, Adjustable Timing, Protection Goggles Included
- 4 in 1 Digital Combo Unit for Rehabilitation & Pain Therapy
- Phymaster Shockwave Pain Therapy Equipment for Rehab & Physiotherapy
- Dual Probe Laser Healing Device Laser Pain Relief Apparatus 750mw
- High Performance tDCS tACS tVNS tPCS Brain Stimulator
- PEMT Loop Therapy Equipment with Large Single Ring & Small Double Ring Handles
- Advanced Neurological Therapy And Modulation System
- Professional Hospital Use Transcranial And Vagus Stimulator
- High Power LLLT Diode Laser Therapy System for Physical Therapy Clinics
- Pedal Bike Automatic Legs And Arm Physiotherapy Device
- Advanced PEMT Loop Therapy Equipment for Clinics
- Phymaster Shockwave Physiotherapy Device for Musculoskeletal Disorders
- High-Power 850W Electromagnetic Therapy Device for Professional Physiotherapy
- Digital Neuro Therapy Machine tDCS tPCS tACS tVNS Device
- Phymaster Shockwave Physiotherapy Machine for Tendon & Muscle Therapy
- Phymaster Shockwave Physiotherapy Equipment for Muscle & Joint Pain
- Digital Laser Therapy Machine with Adjustable Timing, Cooling Fan & Protection Goggles
- Elbow CPM Continuous Passive Motion Machine
- Compact Neuro Stimulation Equipment tDCS tPCS tACS tVNS
- Advanced Exoskeleton Therapy Solutions for Rehabilitation Clinics
- Medical-Grade Exoskeleton Technology for Therapy Centers
- Smart Wearable Exoskeletons for Assisted Rehabilitation Programs
- Back, Leg and Chest Dynamometer for Reliable Clinical Documentation
- Smart Neuro Stimulation Device tDCS tPCS tACS tVNS Machine
- Hospital Grade Neuromodulation And Brain Therapy Device
- Pelvic Muscle Conditioning Chair for Long-Term Physiotherapy Programs
- Digital Brain Therapy Device tDCS tACS tVNS tPCS System
- Back Leg Chest Dynamometer for Post-Injury Strength Assessment
- Medical-Grade Exoskeletons for Advanced Rehabilitation Clinics
- Therapist Recommended Ankle CPM Machine For Home & Clinic Use
- Shock Wave Therapy Machine, For Hospital
- Cryopush Intermittent Pressure Cold Compression Unit
- UniGym Portable Exercise System Home Gym for Post-Surgery Recovery
- Imported Ankle Therapy CPM With Remote For Easy Operation
- Infrared Heat Therapy Knee Massager for Arthritis Pain Relief with Vibration and Air Compression
- Smart Multi Therapy Brain And Nerve Stimulation System
- Phymaster Shockwave Advanced Pain Relief Shockwave Therapy Equipment
- Multi-Function Home Gym Equipment for Strength Training & Wellness Compact Uni Gym
- Tecar Therapy Machine for Chronic Pain and Sports Injuries
- Advanced tDCS tACS tVNS tPCS Brain Therapy Equipment
- 750N Chiropractic Adjusting Tool Instrument Electric Spine Chiropractor Electric Impulse Gun Actuator Massager Professional Massage Equipment for Men Women Adjust Vertebration and Thoracic Scoliosis
- Exoskeleton Rehabilitation Technology for Strength and Mobility
- Advanced Neuro Rehab System tDCS tACS tVNS tPCS
- Advanced Brain And Nerve Modulation For Clinical Use
- Magnetic Pulse Therapy Machine-Deep Oscillation & Muscle Recovery Solution
- Pneumatic Radial Shockwave Therapy Device 7 Heads, 10 Modes
- KM521 Medical Grade Pelvic Muscle Strengthening Device for Clinics
- Konmed KM-560 Advanced Biofeedback Device with EMG Monitoring
- Konmed KM-560 EMG Biofeedback Device with Multi Channel Therapy
- Konmed KM-560 Clinical EMG Biofeedback Device with Therapy Modes
- Exoskeleton-Assisted Rehabilitation for Strength and Balance
- Portable Brain Rehab Device tDCS tACS tVNS tPCS System
- Clinical Pneumatic Shockwave 7 Head Radial ESWT Machine
- Konmed KM-560 EMG Biofeedback Stimulator with Multi Therapy
- Exoskeleton Technology for Precision Motor Recovery Programs
- Konmed KM-560 EMG Biofeedback System with Clinical Programs
- Konmed KM-560 Biofeedback Device with Triggered Stimulation
- Konmed KM-560 Biofeedback Equipment with Rehab Programs
- Konmed KM-560 Clinical EMG Biofeedback Device with NMES
- High Intensity Class 4 Laser Therapy System for Pain and Rehabilitation
- Shockwave therapy for joint lubrication Pneumatic shockwave therapy
- Shockwave therapy for spinal stenosis Pneumatic shockwave therapy for nerve entrapments
- Exoskeleton Technology for Consistent and Measurable Recovery
- Pedo Cycle Top-rated motorized cycle for physical therapy
- Phymaster Shockwave Pain Relief Machine for Sports Injury Clinics
- Professional Clinical Shockwave Therapy Machine-7 Applicator Heads, 10 Treatment Modes
- Medical Grade High Intensity Class 4 Laser Therapy Equipment
- Digital Neuro Rehabilitation System tDCS tACS tVNS tPCS
- 7 Head Pneumatic Shockwave Radial ESWT Device 10 Modes
- High Power Class IV Laser Therapy Equipment for Rehabilitation Clinics
- Phymaster Shockwave Physiotherapy Equipment for Sports Injury Management
- Biotronix Pain Relief Laser Therapy Device Pointed and Cluster Probe Physiotherapy
- Clinical Use High Power Class 4 Laser Therapy System
- Robotic Exoskeleton Rehabilitation for Safe Functional Training
- TENS auto mode ultrasound therapy combo electrotherapy professional physiotherapy
- Class IV Laser Therapy Machine for Physiotherapy Pain Management
- Clinical Brainwave Regulation And Stroke Recovery Equipment
- Medical Grade Brain Plasticity Enhancement And Therapy System
- Integrated Brain Activation And Autonomic Regulation System
- PEMT Loop Therapy-Large & Small Loop Handles
- Phymaster Shockwave Advanced Shockwave Pain Therapy System
- Premium Ankle CPM Machine With Smooth, Noise-Free Operation
- Phymaster Shockwave Professional Pain Relief Therapy Equipment
- Medical Exoskeleton Solutions for Functional Movement Therapy
- Compact UniGym Physiotherapy Device for Muscle Strength and Mobility
- Konmed KM-560 Professional Biofeedback Equipment for Clinics
- 50 Hz ABS Plastic Handheld Pain Relief Laser Therapy Device Handheld Physiotherapy LASER
- Class 4 Laser Therapy Equipment for Orthopedic Pain Management
- Pstics ABS Electric pedal cycle for upper and lower body exercises Fitness Equipment
- Advanced Multi Current Brain And Cognitive Therapy System
- tDCS tPCS tACS tVNS Neurostimulator for Physiotherapy
- Medical Neuromodulation And Neural Therapy Machine
- Robotic Exoskeleton Therapy for Structured Rehabilitation Programs
- Konmed KM-560 EMG Biofeedback Stimulator with Rehab Programs
- Compact Clinical Brain Stimulation Console
- Medical Exoskeleton Systems for Controlled Functional Training
- Advanced Class IV Laser Therapy Machine for Joint Pain Treatment
- Portable PEST Loop Rehabilitation Machine for Clinic, Home, and Hospital Use
- Exoskeleton Therapy Systems Supporting Motor Skill Recovery
- Comfort Strap Ankle CPM Machine For Clinical And Home Rehabilitation
- 4 Channel Electrotherapy Unit TENS EMS IFT MS Physiotherapy Device
- Compact Brain Stimulation Machine tDCS tPCS tACS tVNS
- Exoskeleton Rehabilitation Technology for Functional Improvement
- High Intensity Class 4 Laser Therapy Machine for Orthopedic Rehabilitation
- Clinical Class 4 Laser Physiotherapy Equipment for Pain Relief Clinics
- Cold Therapy System for Back, Hip & Joint Rehabilitation
- Phymaster Shockwave Professional Shockwave Therapy Equipment for Clinics
- Phymaster Shockwave Therapy Machine for Joint & Tendon Rehabilitation
- Phymaster Shockwave Physiotherapy Machine for Advanced Clinical Therapy
- Exoskeleton Solutions Supporting Progressive Movement Therapy
- Phymaster Shockwave Therapy Machine for Sports Injury Pain Relief
- Phymaster Shockwave Physiotherapy System for Chronic Pain Treatment
- Phymaster Shockwave Advanced Pain Relief Shockwave Equipment
- Class IV Laser Therapy Device for Pain Management and Recovery
- Advanced Class IV Laser Therapy Equipment for Joint Pain Treatment
- Electro Brain Stimulator tDCS tPCS tACS tVNS Equipment
- motorized cycling bike for physiotherapy bike for home pedal exerciser for seniors
- Phymaster Shockwave Advanced Pain Relief Physiotherapy Equipment
- Phymaster Shockwave Therapy System for Non-Invasive Pain Relief
- Phymaster Shockwave Pain Relief Shockwave Therapy Equipment
- Economical Model Class 4 Laser Therapy System for Pain Clinics
- Professional Pelvic Floor Strengthening Stimulator-KM521 Model
- Professional Pneumatic Shockwave Therapy System-1-22Hz, 0.5-12 Bar, 7 Heads
- Phymaster Shockwave Physiotherapy System for Professional Use
- Phymaster Shockwave Advanced Pain Management Physiotherapy Machine
- Konmed KM-560 Biofeedback Machine with EMG Multi Mode Therapy
- Dual Function Cold Compression Machine for Orthopedic and Post-Op Recovery
- UniGym Active Passive Resistive Training Device
- Combi 2 in One Ultrasound Therapy 1 Mhz and TENS 4 ch Auto Mode Digital
- Clinical Class IV Laser Therapy Equipment for Pain Relief
- Phymaster Shockwave Advanced Physiotherapy Pain Relief Machine
- Economical Class 4 Laser Therapy Machine for Pain Treatment Clinics
- Phymaster Shockwave Physiotherapy Machine for Chronic Pain Therapy
- PowerPulse Focus Shockwave Machine for Deep Tissue Regeneration Therapy
- Phymaster Shockwave Shockwave Therapy Device for Clinical Use
- Phymaster Shockwave Shockwave Therapy Machine for Orthopedic Pain
- Phymaster Shockwave Pain Relief Therapy Device for Rehabilitation
- Phymaster Shockwave Professional Pain Therapy Physiotherapy Machine
- Phymaster Shockwave Physiotherapy Pain Therapy Device
- Phymaster Shockwave Advanced Pain Relief Shockwave Therapy System
- Pelvic Floor Strengthening & Muscle Re-Education Therapy Chair
- Phymaster Shockwave Therapy System for Sports & Injury Rehabilitation
- Pneumatic Shockwave 7 Head Clinical Radial ESWT Machine Extracorporeal Therapy
- Compact Brain Rehab Device tDCS tACS tVNS tPCS
- Motorized Cryo Therapy Equipment for Knee, Shoulder & Back Care
- Motorized Cryotherapy Unit Designed for Joint Pain and Inflammation Relief
- Phymaster Shockwave Physiotherapy Machine for Professional Rehab Centers
- Pelvic Floor Muscle Activation Chair for Non-Invasive Physiotherapy
- Konmed KM-560 EMG Biofeedback Device with Multi Therapy Modes
- Konmed KM-560 Biofeedback Equipment with Therapy Programs
- Konmed KM-560 Biofeedback Machine with Electrical Stimulator
- Digital Clinical Pelvic Muscle Therapy Chair for Rehabilitation Clinics
- Phymaster Shockwave Therapy Machine for Physiotherapy Pain Relief
- Phymaster Shockwave Advanced Shockwave Therapy Device
- Electric pedal cycle for enhancing physical recovery
- Phymaster Shockwave Pain Management Machine for Clinics & Hospitals
- Radial shockwave therapy for chronic tendinopathies and physical therapy
- Medical Neuromodulation And Brain Activation Therapy System
- Smart Pelvic Muscle Stimulator Chair for Urinary Control Therapy
- Class 4 Laser Pain Therapy Machine for Physiotherapy Centers
- Hospital Grade Pelvic Floor Rehabilitation Stimulation Chair
- Ultra-Accurate Focus Shockwave Device for Deep Joint & Spine Pain Relief
- Non-Invasive Pelvic Floor Therapy Chair for Men & Women
- Robotic Wearable Exoskeletons for Professional Physiotherapy
- Konmed KM-560 4 Channel Biofeedback Machine for Neuro Rehabilitation
- Back, Leg and Chest Strength Evaluation System for Professionals
- Low Level Laser Therapy
- Back Leg Chest Dynamometer for Functional Strength Evaluation
- Dual Probe LASER Therapy Pain Relief Device Equipment Class 3 B Laser Pointed Cluster Probe
- Professional Back Leg Chest Strength Testing Device for Clinics
- Portable 1470nm/980nm Diode Laser Device for Minimally Invasive Piles and Vascular Treatment
- Pulse Electromagnetic Therapy Device for Rehabilitation Therapy
- Compact & Durable Back Leg Chest Dynamometer for Clinics and Gyms
- Back, Leg and Chest Dynamometer for Scientific Muscle Strength Testing
- Medical & Sports Back Leg Chest Dynamometer for Strength Analysis
- Back, Leg and Chest Dynamometer for Clinical Strength Documentation
- Physiotherapy Muscle Testing Dynamometer for Back, Legs & Chest
- Professional Pelvic Muscle Stimulation Chair for Rehabilitation Clinics
- tDCS tACS tVNS tPCS Brain Therapy Device for Neuro Rehabilitation
- Back Leg Chest Dynamometer for Objective Muscle Power Assessment
- Class 4 Laser Therapy Device Economical Model for Physiotherapy
- Electric pedo bike Motorized mini cycle Electric therapy pedal Motorized
- Clinical Muscle Strength Analyzer Back, Leg & Chest Dynamometer
- Konmed KM-560 EMG Biofeedback System for Muscle
- DVT Compression Corded Electric Body Massager Therapy For Legs Pain Relief, Black
- Pain Physiotherapy Laser Therapy Machine Portable Handheld Model
- PEMT Loop Therapy Physiotherapy Device Premium Quality
- Clinical Pelvic Floor Muscle Therapy Chair for Advanced Rehabilitation
- Exoskeleton-Assisted Physiotherapy for Strength and Coordination
- Konmed KM-560 Advanced Biofeedback Equipment
- Pstics ABS Electric pedal cycle for upper and lower body exercises
- Konmed KM-560 Biofeedback Device with NMES Therapy Modes
- Pulse Electromagnetic Therapy Device Premium Clinical Model
- Professional PEMT Loop Therapy Device for Physiotherapy Treatment
- Top-Quality Digital Remote-Controlled Physiotherapy Pedal Device Motorized Electrical
- Konmed KM-560 Physiotherapy EMG Biofeedback
- Konmed KM-560 EMG Biofeedback System with Rehab Function
- Pelvic Floor Muscle Rehabilitation Chair for Womens Health Clinics
- Electromagnetic Loop Therapy Device Premium Physiotherapy Equipment
- Advanced Pelvic Muscle Therapy Chair for Physiotherapy & Recovery
- Digital Neuro Therapy System tDCS tACS tVNS tPCS
- Premium PEMF Loop Therapy Machine for Pain Relief Therapy
- Inclinometer for Physical Therapy PT Range of Motion (ROM) for Students and Professionals
- Digital Brain Stimulation Machine tDCS tACS tVNS tPCS
- PEMF Loop Therapy Physiotherapy Equipment for Pain Management
- PEMF Loop Therapy Device for Pain Relief and Rehabilitation Clinics
- Pulse Electromagnetic Therapy Loop Applicator Physiotherapy Device
- Pulse Electromagnetic Therapy Device for Muscle and Joint Pain
- KM521 Clinical Pelvic Muscle Rehab Device with Adjustable Intensity
- PEMF Loop Therapy Device for Rehabilitation Therapy Clinics
- Electro Brain Therapy Device tDCS tPCS tACS tVNS Equipment
- Clinical Brain Stimulation Machine tDCS tACS tVNS tPCS
- Advanced Neuro Modulation Device tDCS tPCS tACS tVNS
- Neuro Rehab Stimulation Device tDCS tACS tVNS tPCS
- Portable Brain Therapy Machine tDCS tPCS tACS tVNS Device
- Smart Neuro Therapy Device tDCS tPCS tACS tVNS System
- Smart Brain Stimulator Device tDCS tACS tVNS tPCS
- PEMF Loop Therapy Device for Sports Injury Recovery
- Electrical Pedal Cycle For Stroke Recovery Aid Motorized Digital Rehabilitation Pedal Physiotherapy equipment
- Portable Brain Stimulation System tDCS tACS tVNS tPCS
- Digital PEMT Loop Therapy Device for Pain Management Therapy
- Electro Brain Therapy Equipment tDCS tPCS tACS tVNS Device
- Multi Functional Brain Therapy Device tDCS tPCS tACS tVNS
- Multi Functional Brain and Neuro Device tDCS tACS tVNS tPCS
- Portable Handy Laser Therapy Device-Rechargeable with Cooling Fan & Adjustable Stand
- Smart Brain Therapy Machine tDCS tACS tVNS tPCS System
- tDCS tACS tVNS tPCS Neuro Stimulation Device for Brain Therapy
- Robotic Exoskeleton Therapy for Progressive Clinical Recovery
- Motorized Ankle Rehabilitation CPM With Adjustable Speed & Angle Levels
- PEMT Loop Therapy-Multi-Pulse Frequency 2-8Hz
- Professional Electromagnetic Shockwave Therapy Machine-Multi-Mode, Portable Design
- Professional Neuro Rehab Therapy Machine tDCS tPCS tACS tVNS
- Digital PEMT Loop Therapy Machine Premium Clinical Model
- Professional Cold Therapy System for Inflammation Control
- Exoskeleton-Based Training for Functional Movement Restoration
- Pulse Electromagnetic Field Therapy Equipment for Physiotherapy
- PEMF Loop Therapy Device for Physiotherapy Pain Relief
- Advanced PEMF Loop Therapy Machine for Physiotherapy Clinics
- Exoskeleton Systems Enhancing Clinical Rehabilitation Outcomes
- Pulse Electromagnetic Therapy Device for Orthopedic Rehabilitation
- PEMT Loop Therapy Machine for Muscle and Joint Pain Relief
- Portable Brain Stimulation Equipment tDCS tPCS tACS tVNS
- PEMF Loop Therapy Machine Professional Physiotherapy Equipment
- PEMT Loop Therapy Device for Orthopedic Pain Treatment
- Tecar Therapy Diathermy Machine, Tecar Therapy Machine For Body Pain
- Tecar Therapy Diathermy Machine, CET RET For Sports Rehabilitator Sport Therapist, 4 Modes and 10 Body Parts To Choose From, 300~448KHz Therapy Machine For Body Pain
- Advanced Digital Ankle CPM Equipment For Faster Recovery
- Professional PEMF Loop Therapy Device for Orthopedic Therapy
- Advanced PEMT Loop Therapy Machine for Physiotherapy Centers
- Premium PEMT Loop Therapy Machine for Physiotherapy Treatment
- Physiotherapy laser equipment with LCD Pre-programmed laser physiotherapy device
- Pulse Electromagnetic Therapy Machine for Muscle and Joint Pain
- Digital PEMF Loop Therapy Device for Physiotherapy Treatment
- Premium Pulse Electromagnetic Therapy Machine for Physiotherapy Clinics
- PEMF Loop Therapy Device for Pain Relief and Rehabilitation
- Clinical Exoskeleton Devices for Modern Physiotherapy Practice
- tDCS tACS tVNS tPCS Brain Stimulation Therapy Device for Rehab
- Precision Brain And Neural Recovery Equipment
- High Intensity Magnetic Therapy Emtt Magnetotherapy for Pain Relief Physical Equipment
- Advanced Pulse & Magnetic Therapy Device for Muscle Recovery & Pain Relief
- Digital PEMT Loop Therapy Machine for Physiotherapy Centers
- Cryopush Digital Cold Compression Machine for Joint Pain Management
- PEMT Loop Therapy-4500Hz Oscillation System
- Pulse Electromagnetic Therapy Loop Machine Premium Model
- PEMF Loop Therapy Machine for Rehabilitation Treatment
- Physiotherapy Grade Back Leg Chest Dynamometer for Daily Practice
- Clinical Back, Leg and Chest Dynamometer for Objective Strength Evaluation
- Reliable Back, Leg and Chest Dynamometer for Daily Clinical Use
- UniGym All-in-One Physio & Fitness Rehabilitation Equipment
- Advanced Brainwave Regulation And Vagus Nerve Therapy System
- Medical Brainwave And Vagus Nerve Neuromodulation Equipment
- Professional Brainwave And Vagus Nerve Activation Device
- PEMT Loop Therapy Physiotherapy Machine for Pain Relief
- Clinical Brainwave And Vagus Nerve Therapy Machine
- Compact Brain Therapy Equipment tDCS tACS tVNS tPCS Machine
- Konmed KM-560 4 Channel Physiotherapy Biofeedback
- Konmed KM-560 EMG Biofeedback Machine with Therapy Modes
- Konmed KM-560 Advanced Biofeedback Device with Multi Channel
- Konmed KM-560 Biofeedback Machine for Pelvic Floor Therapy
- Konmed KM-560 4 Channel EMG Biofeedback Device for Rehab Use
- Konmed KM-560 Professional Biofeedback Machine for Rehab
- Konmed KM-560 4 Channel EMG Biofeedback System for Clinics
- Konmed KM-560 EMG Biofeedback Machine for Sports Therapy
- Konmed KM-560 EMG Biofeedback Device with Therapy Control
- Konmed KM-560 4 Channel Physiotherapy Biofeedback Equipment
- Konmed KM-560 EMG Biofeedback Machine with Gaming Interface
- Konmed KM-560 Advanced Biofeedback Device with EMG Output
- Automatic Motorized Wrist CPM Equipment for Smooth and Controlled Exercise
- Konmed KM-560 4 Channel EMG Biofeedback Device for Sports Rehab
- Konmed KM-560 Physiotherapy EMG Biofeedback with Gaming Feature
- Konmed KM-560 4 Channel EMG Biofeedback Device with ETS FES NMES
- Konmed KM-560 EMG Biofeedback Equipment with 4 Channel Output
- Konmed KM-560 Physiotherapy Biofeedback Device with ETS NMES FES
- Konmed KM-560 Pelvic Floor EMG Biofeedback Machine
- Konmed KM-560 EMG Biofeedback Stimulator for Rehabilitation
- Konmed KM-560 Rehab Biofeedback Device with Electrical Stimulation
- Konmed KM-560 EMG Biofeedback System for Neuro Rehabilitation
- Compact Brain Neuro Therapy Equipment tDCS tPCS tACS tVNS
- Smart Neuro Rehab Therapy Machine tDCS tACS tVNS tPCS
- Digital Neuro Therapy System tDCS tACS tVNS tPCS Machine
- Professional Neuro Stimulation Therapy Equipment tDCS tPCS tACS tVNS
- Brain Neuro Rehab Therapy Device tDCS tACS tVNS tPCS
- Electro Brain Stimulation Therapy Machine tDCS tPCS tACS tVNS
- Multi Functional Neuro Therapy System tDCS tACS tVNS tPCS Device
- Electro Neuro Modulation Therapy Device tDCS tACS tVNS tPCS
- Konmed KM-560 Professional Biofeedback Machine for Hospitals
- Konmed KM-560 EMG Biofeedback Device with Therapy Interface
- Konmed KM-560 EMG Biofeedback Machine for Clinical Rehab
- Konmed KM-560 Physiotherapy Biofeedback Device with EMG Training
- Konmed KM-560 EMG Biofeedback System for Recovery Therapy
- Konmed KM-560 Biofeedback Machine with Stimulator Function
- Konmed KM-560 Rehab Biofeedback Machine with EMG Training
- Konmed KM-560 Clinical Biofeedback Device with Multi Modes
- Konmed KM-560 4 Channel Biofeedback System with FES Therapy
- Konmed KM-560 Biofeedback Machine with EMG and Gaming Therapy
- Konmed KM-560 4 Channel EMG Biofeedback Device with Therapy Modes
- Konmed KM-560 Clinical Biofeedback Device with Rehab Features
- Konmed KM-560 4 Channel Biofeedback System with Muscle Training
- Konmed KM-560 Pelvic Floor EMG Biofeedback Machine for Therapy
- Robotic Exoskeleton Rehabilitation for Controlled Joint Movement
- Medical PEST Loop Device for Stroke Rehabilitation and Joint Mobility Training
- Electrical Wrist Rehabilitation CPM Unit-Smooth Motorized Operation
- Clinic Use 980nm + 1470nm Diode Laser Machine for Fistula, Hemorrhoids and Vascular Surgery
- Professional Diode Laser 980nm 1470nm for Anorectal, Varicose Vein and Piles Treatment
- 1470nm + 980nm Diode Laser for Proctology, Hemorrhoid Surgery, Vascular Lesion Therapy
- Medical-Grade Robotic Exoskeletons for Rehabilitation Centers
- Complete Multi Mode Brain Rehabilitation And Therapy System
- All-In-One 4-Channel Electro-Magnetic Rehabilitation System (4T + EMT2)
- Cold Laser Device Home Laser Therapy Pain Relief Therapy Device,Healing &Reducing Pain and Inflammation
- Cold Laser, 660 Mw Pain Therapy Device For Knee, Shoulder, Back, Joint & Muscle Pain
- Mini UV Gel Nail Lamp, UV Nail Dryer, Handheld 5W 3pcs Lamp Chips Fast Curing Portable USB Nail Dryer
- Hand-held Physical Treatment Low-level Infrared Light Cold Laser Therapy
- Konmed KM-560 EMG Biofeedback System for Muscle Activation
- Konmed KM-560 Physiotherapy Biofeedback Device with ETS Mode
- Medical Pelvic Floor Rehabilitation Chair for Incontinence Treatment
- Advanced High Power Laser Therapy Machine Class 4 for Clinics
- Digital Pelvic Muscle Rehab Chair with Automatic Therapy Programs
- High Power Class 4 Laser Physiotherapy Device for Clinics
- Advanced Brain Plasticity Enhancement And Cognitive Therapy Equipment
- UniGym Interactive Rehab & Recovery Equipment for Clinics and Home Use
- Economical Model Class IV Laser Therapy Equipment for Clinics
- Inclinometer for Physical Therapy No Leak PT Inclinometer for Range of Motion (ROM) Measurement
- PEMF Loop Therapy Device Premium Physiotherapy Equipment
- Konmed KM-560 Professional Biofeedback Machine
- Class 4 Laser Therapy Device for Sports Injury Treatment Clinics
- 7-Head Pneumatic ESWT Device-Energy 0.5-1.2 Bar
- Best DVT Pump for Circulation and Swelling Relief
- Electrical Wrist CPM Machine-Continuous Passive Motion Device for Rehabilitation
- High Power Laser Therapy Machine Class IV for Physiotherapy Centers
- Konmed KM-560 Clinical Biofeedback Device for Rehabilitation
- Konmed KM-560 4 Channel Biofeedback System with EMG Output
- Konmed KM-560 Biofeedback Machine with EMG Therapy System
- Advanced Wrist CPM Therapy Equipment for Orthopedic and Neuro Rehabilitation
- Integrated Brain And Nerve Therapy Equipment
- Motorized Ankle CPM Device For Smooth Plantar-Dorsiflexion Movement
- Precision Controlled Brain And Neural Activation System
- Electromagnetic Loop Therapy Device for Pain Relief Therapy
- Chiropractor Gun Electric 400N Spine Adjustor 4 Levels
- Complete TDCS TPCS TACS TVNS Neuro Rehabilitation Machine
- Adjustable Ankle CPM Machine With 1-9 Speed And Intensity Settings
- Konmed KM-560 Biofeedback Equipment with ETS Modes
- PEMT Loop Therapy Machine Premium Touch Screen Model
- Exoskeleton Solutions for Modern Neuro Rehabilitation
- Advanced Multi Current Brain And Neural Therapy Machine
- Advanced Electrical Cortical And Neural Therapy Machine
- Radial Pneumatic ESWT Device-Adjustable Frequency 1-22Hz, 0.5-12 Bar Intensity
- Phymaster Shockwave Physiotherapy Machine for Sports & Rehab Use
- Professional Pelvic Muscle Activation Chair for Clinical Treatment
- Low Level Laser Therapy Cold and Hot Laser Dual Probe 750 Mw 910nm Biotronix Physiotherapy Laser
- Pulse Electromagnetic Therapy Equipment Premium Model
- Pulse Electromagnetic Therapy Loop Device Professional Model
- PEMF Loop Therapy Equipment for Rehabilitation Clinics
- Cryopush Cold Therapy Machine with Long 1.5 Meter Hose
- Ankle CPM Device for Post-Surgery Recovery Electric Passive Motion Machine
- Economical Model Class IV Laser Physiotherapy Device
- Adjustable Infrared Knee Therapy Massager Heat & Vibration Pain Relief Machine for Arthritis and Sports Injury
- Pulse Electromagnetic Field Therapy Loop Machine for Physiotherapy
- Portable Knee Massager with Heat Kneading Infrared Vibration Therapy for Joint and Muscle Pain Relief
- Electric Heated Knee Massager for Joint Pain Relief with Infrared Vibration and Kneading Physiotherapy
- Pulse Electromagnetic Therapy Loop Machine Clinical Physiotherapy Model
- Pulse Electromagnetic Therapy Device Premium Touch Screen Model
- Medical Grade Brain And Autonomic Nerve Activation System
- Advanced Brain Plasticity And Cognitive Recovery Machine
- Pulse Electromagnetic Therapy Loop Machine for Pain Management
- Smart Brainwave And Neural Control Equipment
- Pulse Electromagnetic Therapy Machine for Rehabilitation Therapy
- Back, Leg and Chest Dynamometer for Recovery & Performance Monitoring
- PEMT Loop Therapy Device for Pain Relief Physiotherapy
- Multi Waveform Brain And Neural Stimulation Equipment
- Advanced Medical Class 4 Laser Therapy Equipment for Rehabilitation Clinics
- Professional Brain And Vagus Nerve Activation System
- Advanced Multi Current Brain Rehabilitation System
- Advanced Electrical Brain And Nerve Stimulation Equipment
- KM-580 Biofeedback Stimulator with EMG, ETS, NMES, FES, and TENS Therapy
- Focused Shockwave Therapy Machine for Tendon and Nerve Pain Treatment
- Medical Grade Cortical And Autonomic Nervous System Stimulator
- Focused Shockwave Therapy Equipment for Physiotherapy and Rehabilitation
- 1 MHz ultrasound therapy 4 channel TENS machine auto mode TENS unit
- Phymaster Shockwave Advanced Shockwave Therapy Machine for Rehab
- All-In-One 4-Channel Electro-Magnetic Rehabilitation System (4T + EMT2)
- High Power Class 4 Laser Therapy Machine for Sports Physiotherapy
- Konmed KM-560 Physiotherapy EMG Biofeedback with ETS
- Cryopush Professional Cold Compression Machine with 9 Modes
- KM521 Clinical Pelvic Muscle Strengthening System for Rehab Centers
- best self-adhesive electrode pads for TENS units
- Ankle Passive Motion Machine For Smooth Plantar-Dorsiflexion Movement
- Advanced Brainwave Regulation Therapy Unit
- Shockwave Therapy Machine for Plantar Fasciitis, Shoulder & Elbow Pain
- Tecar therapy Diathermy Physiotherapy Equipment physio spine machine shoulder pain relief
- Konmed KM-560 Physiotherapy Biofeedback Device with Gaming
- Exoskeleton-Based Therapy for Structured Rehabilitation Programs
- Phymaster Shockwave Shockwave Therapy System for Clinical Rehabilitation
- High Power Class 4 Laser Therapy Machine for Advanced Pain Management Clinics
- Wrist Joint CPM Device-Digital, Remote Controlled, and Speed Adjustable
- Pulse Electromagnetic Therapy Device for Sports Injury Treatment
- Digital Brain Rehab Equipment tDCS tACS tVNS tPCS
- Pelvic Muscle Conditioning Chair for Long-Term Pelvic Health Support
- Premium Wrist CPM Device-Adjustable Speed & Angle for Physiotherapy Rehabilitation
- Metal Physio Magneto Extracorporeal Machine NIRS Therapy
- Exoskeleton Technology for Professional Rehabilitation Centers
- Professional Class 4 Laser Therapy Machine for Advanced Rehab Clinics
- Phymaster Shockwave Pain Management Therapy Equipment for Clinics
- Wrist CPM Physiotherapy Device with 90 Motion Range and Adjustable Controls
- Versatile Handheld Laser: Ideal For Back, Neck, And Shoulder Pain Rechargeable Laser Therapy Device:
- Robotic Exoskeletons in Post-Stroke Physiotherapy Programs
- Advanced Class IV Laser Therapy Equipment for Pain Relief Clinics
- Smart Rehabilitation Exoskeletons for Hands Arms and Legs
- Advanced Multi Channel Brain And Neural Therapy Machine
- Konmed KM-560 EMG Biofeedback Device
- Exoskeleton Therapy Enhancing Neuroplasticity and Motor Control
- Phymaster Shockwave Physiotherapy Machine for Deep Tissue Pain Therapy
- High-Frequency Electromagnetic Radial Shockwave Therapy Machine 5-250 Hz, 10 Modes
- High Power Class IV Laser Therapy System for Pain Relief Treatment
- Portable grip strength dynamometer Digital dynamometer grip tester
- Phymaster Shockwave Shockwave Therapy Equipment for Pain Management
- Advanced Class 4 Laser Therapy Machine for Modern Rehab Centers
- Focused ESWT Therapy Device for Deep Tissue Muscle and Tendon Recovery
- Phymaster Shockwave Physiotherapy Machine for Advanced Pain Management
- Portable Laser Therapy Machine for Pain Relief & Rehabilitation, 670mW Power
- Professional Class 4 Laser Therapy Machine for Modern Rehab Clinics
- Professional Class 4 Laser Therapy System for Pain Relief Clinics
- High Intensity Class 4 Laser Therapy Equipment for Physiotherapy Practice
- Hospital Use Multi Mode Neuro Stimulation Machine
- Electric Hair Growth Massage Treatment Anti Hair Loss Head Brush
- Medical Grade Cold and Compression Therapy Unit for Rehabilitation Clinics
- Phymaster Shockwave Shockwave Therapy Machine for Clinical Pain Care
- Phymaster Shockwave Advanced Shockwave Therapy Machine for Clinics
- Phymaster Shockwave Therapy Machine for Deep Tissue Pain Relief
- Exoskeleton-Based Training for Neurological Movement Disorders
- Smart Wearable Exoskeletons for Targeted Movement Training
- Smart Exoskeleton Devices for Precision Rehabilitation Therapy
- Clinical Pelvic Floor Muscle Therapy Chair for Comprehensive Rehabilitation
- Medical Class 4 Laser Therapy Device for Physiotherapy and Pain Relief
- Konmed KM-560 Physiotherapy EMG Biofeedback with Clinical Mode
- Grip strength tester digital Hand strength gauge
- Portable digital hand grip strength tester
- Digital Physiotherapy Machine LASER therapy Pain Relief Dual Probe 750mw 910nm
- Rehabilitation Cryo Cold Therapy System with Universal Joint Pads
- Exoskeleton Systems Supporting Safe and Controlled Therapy
- Phymaster Shockwave Shockwave Therapy System for Professional Clinics
- Advanced Brain Stimulation Device tDCS tPCS tACS tVNS
- Clinical-Grade Exoskeletons for Physiotherapy and Recovery
- Smart Brain And Autonomic Nerve Stimulator
- High Performance Brain and Nerve Stimulator tDCS tACS tVNS tPCS
- Professional Neuro Stimulator tDCS tACS tVNS tPCS
- Konmed KM-560 Clinical Biofeedback Machine with ETS Stimulation
- Clinical Grade Neuro Recovery Stimulation Unit
- Phymaster Shockwave Shockwave Therapy Equipment for Clinical Treatment
- Rechargeable Handy Laser Machine with Adjustable Stand & Fan
- Advanced 808nm/980nm Diode Laser Device for Pain Relief and Rehabilitation
- 810nm+980nm ABS Plastic Laser Therapy Unit Class 4, For Clinical
- Wearable Robotics Exoskeletons for Progressive Motor Training
- Smart Brain And Vagus Nerve Stimulation Equipment
- Portable Neuro Therapy Device tDCS tPCS tACS tVNS
- Robotic Exoskeleton Devices for Precision Rehab Training
- High-Power PEMT Loop Machine for Sports Injury & Rehabilitation Centers
- Electrotherapy Physiotherapy Device 4 in 1 Portable Model
- Phymaster Shockwave Shockwave Therapy Device for Sports & Ortho Clinics
- Professional 4 in 1 Combo Therapy Unit with Dual Output
- PEMT Loop Therapy Device for Sports Injury Rehabilitation
- Precision Controlled Transcranial Therapy Device
- Motorized pedal exercise machine Electrical Pedal Cycle For Stroke Recovery
- Professional Brain Therapy Machine tDCS tACS tVNS tPCS
- Multi Mode Neuro Stimulator tDCS tPCS tACS tVNS
- remote-controlled Top-Grade Electric Stroke Recovery Pedal Machine for Home Rehabilitation
- PEMF Loop Therapy Device Professional Physiotherapy Equipment
- Multi Functional Brain Stimulator tDCS tACS tVNS tPCS
- Digital Remote Controlled Wrist CPM Device-Physiotherapy Equipment for Clinics
- Digital Pelvic Floor Therapy Chair for Urinary & Pelvic Muscle Control
- Phymaster Shockwave Physiotherapy Equipment for Orthopedic Pain Therapy
- Phymaster Shockwave Shockwave Therapy Machine for Clinical Rehabilitation
- Electro Neuro Therapy Equipment tDCS tACS tVNS tPCS Device
- Konmed KM-560 4 Channel EMG Biofeedback System with Monitoring
- Robotic Gait Rehabilitation Exoskeleton with Smart Control System
- Exoskeleton Therapy for Structured Recovery Programs
- 4 in 1 Digital Therapy Device for Nerve & Muscle Stimulation
- Professional Class 4 Laser Therapy Device for Modern Physiotherapy Setup
- Exoskeleton Therapy Technology for Consistent Recovery Outcomes
- Exoskeleton-Assisted Rehabilitation for Controlled Motor Practice
- Phymaster Shockwave Shockwave Therapy System for Rehabilitation Clinics
- Deep Oscillation PEMT Loop Therapy Equipment for Sports Injury Recovery
- Professional 4 in 1 Physiotherapy Device for Clinic Use
- Advanced Digital Neuromodulation And Rehab System
- Advanced 980nm Class 4 Diode Laser for Physiotherapy & Pain Relief
- Konmed KM-560 EMG Biofeedback System for Sports Injury Rehabilitation
- High Quality Neuro Therapy Device tDCS tACS tVNS tPCS
- Brain Therapy Equipment tDCS tPCS tACS tVNS for Clinics
- Compact Focused Shockwave Therapy Device for Physiotherapy Applications
- Bubble inclinometer for physiotherapy
- Clinical Brain Rehab Device tDCS tACS tVNS tPCS
- Compact Brain Stimulation Equipment tDCS tACS tVNS tPCS
- Premium PEMT Loop Therapy Equipment for Physiotherapy Centers
- Phymaster Shockwave Pain Relief Therapy Machine for Professionals
- Red & Infrared Laser Therapy Device with Digital Display and USB-C Charging
- Inclinometer for physical therapy Inclinometer for rehabilitation
- Portable Physiotherapy LASER Therapy Device Handy Model Pain Relief LASER Machine
- Plastic Digital Pedal Cycle For Stroke Recovery
- Stroke Recovery Pedal with Digital Remote Control with Leg Support Model
- Motorized pedal cycle for comprehensive physical therapy
- 4T TENS EMS + EMT2 PEMF Dual-Mode Physio & Magnet Therapy Machine
- 7 Head Pneumatic Shockwave Clinical Radial ESWT System for Pain Relief
- Physio exerciser Equipment Pedo Cycle Pedal Exerciser Leg Exerciser Pedal Device, Automatic
- Bubble inclinometer for physiotherapy Swivel inclinometer for therapists
- Plastic 360 Degree Physiotherapy Biotronix Bubble Inclinometer, Size: Compact, Packaging Type: Box
- Portable pain relief LASER wand Handheld LASER therapy for pain relief
- Back, Leg and Chest Dynamometer for Post-Surgery & Injury Rehabilitation
- Wearable Robotics Exoskeletons for Progressive Therapy Sessions
- Radial shockwave therapy for chronic disease management and quality of life improvement
- Physiotherapy Device Electrical Pedal Cycle For Rehabilitation
- Rechargeable Infrared Hot Compress Knee Massager Physiotherapy Device
- Premium Handheld Laser Device - Effective Pain Relief Anywhere, Anytime
- Pain Relief Magnetic Therapy Physio Magneto massage Extracorporeal Magnetic Transduction
- Radial shockwave therapy for costochondritis for shin pain
- LCD laser pain relief equipment Dual probe laser for therapy
- dual probe laser therapy 910nm pain relief laser physiotherapy
- Physiotherapy Laser Pain Relief Equipment with Dual Probes Digital LCD Based Biotronix
- Cryo Compression Therapy System with Ice Wrap
- Pedo Cycle Innovative Motorized Electrical Pedal Exerciser for Home Use
- Biotronix Shockwave Therapy Clinical Touch Screen 7Heads Foot Switch Pre Program Physiotherapy & ED
- Adjustable motorized pedal exerciser for rehab
- Konmed KM-560 Biofeedback Machine with ETS Triggered Stimulation Mode
- Biotronix Clinical Professional Pain Relief Physiotherapy and ED Treatment Shockwave Therapy Machine
- Electric rehab cycle Physiotherapy pedal machine Motorized therapy bike
- 2 in 1 pmst physio magneto laser physiotherapy equipment EMS21
- Pedo Cycle Exercise Cycle Motorized With Leg Support
- PEMF Physiotherapy Device Pmst Loop For Shoulder Pain Relief With Mats And Pads
- Motorized exercise cycle for elderly therapy
- Compact Pain Relief Handheld Laser with Protection google Wireless Handheld LASER Therapy
- Automatic Rehabilitation Machine Electrical Leg and Arm trainer Precision Pedal Cycle Therapy
- Capacitive Resistive Tecar Therapy Machine for Deep Tissue Physiotherapy
- Knee Massager Rheumatoid Knee Joint Physiotherapy Relieve Knee Arthritis Leg Pain
- Shockwave Therapy Machine Touch Panel White Stylish Model with 5 Heads 10 Treatments
- Leg Pedal Automatic Electrical Digital with Digital Remote Control and Leg Support Pair
- Physical Therapy Professional Easy To Read Range Of Motion Test ROM Rehabilitation Inclinometer
- Pain Relief Pemf Magnetic Therapy Device Physio Magneto Human Pulse Electromagnetic Field Physiotherapy Pmst Loop Machine
- Handheld LASER physiotherapy wand Compact LASER therapy
- Pelvic Muscle Stimulator Chair for Advanced Pelvic Floor Rehabilitation
- Magnetotherapy for Pain Relief Electromagnetic
- Inclinometer for therapists Precision bubble inclinometer
- Computerized Laser Therapy Equipment Advance Pre Program Dual Probe Laser Pointed and Cluster
- Shockwave Machine Professional Clinical Touch Screen for Physiotherapy & ED
- laser Physiotherapy pain relief laser machine Digital display laser therapy device
- Advanced 3B Class Laser Therapy Portable Fast Pain Relief for Joints and Muscles
- 4-Channel 4T TENS EMS Physiotherapy Machine Digital Muscle Stimulator for Pain Relief
- Electric Cold Compression Therapy Unit for Sports Rehabilitation and Therapy
- Electromagnetic PEMT Therapy Machine for Joints, Muscles & Sports Recovery
- Clinical Brain Recovery And Neurological Therapy Machine
- KM-580 Biofeedback and Stimulation Device for Swallowing & Pelvic Therapy
- Biotronix Dual Probe Laser Red visible Pointed probe and Cluster probe 750 mw 910 nm
- 808nm Infrared + 650nm Red Light Laser Therapy Machine for Pain Relief & Rehabilitation
- Inclinometer with swivel head Leak-resistant
- Exoskeleton Rehabilitation Systems for Progressive Motor Training
- KM521 Dual Channel Pelvic Muscle Stimulator with Adjustable Intensity
- Physio Magneto Plus NIRS Therapy Pmst Neo Super Transduction Physio Magneto Extracorporeal Machine
- Best Electric Stroke Recovery Pedal Machine for Digital Electrical Motorized Home Rehabilitation
- Sequential Compression Therapy DVT Device Digital 6 Chamber for Lymphedema Relief
- Wearable Exoskeleton Devices for Assisted Mobility Training
- Phymaster Shockwave Pain Relief Equipment for Rehab Clinics
- Rechargeable Dual-Wavelength Laser Therapy Device Digital Display, Cooling Fan & USB-C Charging
- Clinical Brain Therapy Machine tDCS tPCS tACS tVNS
- High Power Class 4 Laser Therapy Device for Modern Physiotherapy Clinics
- Phymaster Shockwave Pain Relief Physiotherapy Device
- Multi Therapy Neuro Rehabilitation Stimulation Unit
- Medical-Grade Exoskeleton Systems for Rehabilitation Clinics
- Professional Class 4 Laser Physiotherapy System for Pain Relief
- Class 4 Laser Pain Therapy Equipment for Rehabilitation Centers
- Handheld Laser Therapy Equipment for Joint Pain, Arthritis & Rehabilitation
- Professional Class 4 Laser Therapy Device for Physiotherapy Centers
- View Similar IASTM 304 SS Gua Sha Scraping Massage Tool 5 Pc for Soft Tissue Mobilization Physical Therapy
- Economical Model Class IV Laser Therapy Machine for Clinics
- High Power Class IV Laser Physiotherapy Machine for Rehabilitation
- Professional Class 4 Laser Therapy System for Clinical Rehabilitation
- Medical Grade Class IV Laser Therapy System for Pain Management
- Professional PEMT Loop Therapy Machine Clinical Physiotherapy Model
- Advanced Pelvic Muscle Rehab Chair for Womens & Mens Health
- Air Compression Based Clinical Shockwave Therapy Device-1-22Hz, 0.5-12 Bar
- 750mw laser therapy device 910nm laser for physiotherapists
- Pulse Electromagnetic Therapy Machine with Loop Applicator
- Clinical TDCS TPCS TACS TVNS Neuro Rehabilitation And Recovery Machine
- Konmed KM-560 EMG Biofeedback System for Muscle Rehab
- Advanced Pelvic Muscle Rehabilitation Chair for Complete Pelvic Care
- Multi Protocol Brainwave Regulation And Therapy Device
- Pulse Electromagnetic Therapy Equipment for Physiotherapy Centers
- Konmed KM-560 EMG Biofeedback Equipment with ETS Trigger Mode
- Physiotherapy measurement tool Office use inclinometer
- Phymaster Shockwave Physiotherapy Machine for Advanced Pain Relief
- Phymaster Shockwave Therapy Equipment for Sports & Musculoskeletal Pain
- Konmed KM-560 4 Channel EMG Biofeedback Physiotherapy Machine
- Clinical Grade Focus Shockwave Pain Relief & Tissue Regeneration Equipment
- Konmed KM-560 Physiotherapy Biofeedback Device with NME
- Phymaster Shockwave Shockwave Therapy System for Pain Relief Clinics
- Konmed KM-560 Biofeedback Machine with Electrical Therapys
- Phymaster Shockwave Physiotherapy Machine for Advanced Pain Therapy
- Portable Neuro Stimulation Device tDCS tPCS tACS tVNS Machine
- Motorized Digital Rehabilitation Pedal Bike Pedo Cycle Digital Electrical Motorized Stroke rehab bike
- Exoskeleton Rehabilitation Systems for Clinical Performance
- Digital Brainwave Control And Neurological Therapy Equipment
- Intelligent Exoskeleton Robot for Gait Training & Limb Rehabilitation Smart Robotic Physiotherapy Device
- Rechargeable Laser Therapy Device for Pain Relief & Rehabilitation-Adjustable Timing & Stand
- Professional Class IV Laser Physiotherapy System for Rehabilitation
- Phymaster Shockwave Clinical Physiotherapy Machine for Pain Therapy
- Digital Brain Activation And Cognitive Enhancement Machine
- Hospital-Grade Focus Shockwave Therapy System for Long-Term Pain Healing
- PEMT Magnet Therapy Device for Physiotherapy, Muscle Recovery & Wellness
- Konmed KM-560 Multi Therapy EMG Biofeedback Rehabilitation System
- Robotic Exoskeleton Systems for Motor Function Enhancement
- Handheld Laser Physiotherapy Equipment with Adjustable Timer and Cooling System
- 750mw Laser Pain Relief Treatment Infrared Laser Therapy 910nm
- Konmed KM-560 Smart Physiotherapy Equipment with Biofeedback Training
- Motorized UniGym Physiotherapy Trainer with App-Based Interaction
- Advanced DVT Compression Therapy Pump for Enhanced Blood Circulation
- Advanced Digital Remote-Controlled Leg Exerciser for Stroke Recovery Motorized Pedo Cycle
- Smart Brain Plasticity Enhancement Equipment
- Class 4 Laser Physiotherapy Machine for Muscle Pain Treatment
- Motorized pedal cycle Electric leg cycle Motorized exercise equipment Electric home bike Motorized compact exerciser
- 650+808nm Laser Physiotherapy Handy Pain Therapy Handheld Device For Home Use
- Automatic Electrical Rehabilitation Cycle Electrical Pedal Cycle For Stroke Recovery
- Knee pain relief device Electric heating knee massager
- Motorized Electric Mini Pedal Exercise Bike
- Electrical Pedal Cycle For Stroke Recovery Aid Motorized Digital Rehabilitation Pedal Bike
- Motorized therapy bike Plastic Digital Pedal Cycle For Stroke Recovery
- Biotronix Combination Therapy 2 in One Ultrasound UST 1 Mhz TENS 4 ch Digital Electrotherapy
- Brain Stimulation Therapy Machine tDCS tACS tVNS tPCS
- Compact UniGym Rehab Trainer for Arms and Legs
- Phymaster Shockwave Physiotherapy Device for Deep Pain Management
- Portable LASER therapy for physio Handheld pain management LASER
- TensCare X-PR13 probe for electrostimulators with universal connection to 2 mm plug
- Chiropractor Device 750N Spine Correction 50N -750 N Adjustable Force
- Bubble Inclinometer Leak Free 360 degree ROM Physical Therapy Measuring Tool
- Pneumatic shockwave therapy for immune system support Radial and metabolic health
- UniGym Post-Surgery & Injury Recovery Training Device
- Shockwave therapy for migraine relief Pneumatic shockwave therapy for tension headaches
- Pneumatic shockwave therapy for plantar fasciitis and physiotherapy for shock treatment
- Pain Therapy System Shockwave Machine for Pain Relief ED Physiotherapy, For Hospital, Model Name/Number: Renewable Energy
- shockwave therapy for keloid scars blood flow enhancement
- Knee Massager Vibration Heating Joint Physiotherapy Electric Massage Pain Relief Rehabilitation
- Inclinometer for orthopedic use Bubble rehabilitation Therapy
- Handy LASER therapy device Pain relief LASER
- Low Level Laser Therapy LASER Machine for Physiotherapy Cold Laser Therapy Equipment
- Air Pressure Pneumatic Dvt Pump Compression Therapy System Leg Massager Sports Recovery Boots
- Advance Laser Therapy Unit (Hot & Cold) with 2 Probes Digital LCD Display Pre Programmed
- self-adhesive electrode pads for electronic muscle stimulators
- TENS therapy pads EMS therapy pads pulse massager pads
- reusable electrode pads replacement electrode pads
- pulse massager electrode pads IFT electrode pads
- pedal machine Home physiotherapy equipment Pedal exerciser for seniors
- Physio Laser Pain Therapy Device Dual Probe Infrared Laser 910nm
- 4 Heads Manual Chiropractic Adjustment Tool Deep Tissue Spine Massager
- Pneumatic Radial ESWT Device-7 Heads, 10 Modes Shockwave Therapy Machine
- Versatile inclinometer High accuracy inclinometer
- Best motorized electrical pedal exerciser for physiotherapy
- Automatic Leg Rehabilitation Pedal Device Electrical Pedal Exercise Leg Therapy with Leg Support
- UST 1 MHz TENS combo electrotherapy physiotherapy device TENS 4 channel auto mode TENS dual therapy machine
- Advanced Neuro Therapy System tDCS tACS tVNS tPCS Device
- Advanced Dual Probe Laser for Pain Therapy Pointed and Cluster Probe Pre Programmed
- laser pain management Digital laser treatment for physiotherapy
- Advanced Neuro Stimulation Machine tDCS tACS tVNS tPCS
- UniGym Multifunction Physiotherapy & Rehabilitation Exercise Device
- Phymaster Shockwave Physiotherapy Equipment for Joint Pain Treatment
- laser Professional laser therapy machine LCD display laser pain relief device
- Laser therapy device for pain management Digital laser treatment for physiotherapy
- UniGym All-in-One Multifunction Rehabilitation Exercise Device
- Advanced Brain Stimulator tDCS tPCS tACS tVNS System
- Smart Neuro Rehab Device tDCS tACS tVNS tPCS
- Pelvic Muscle Conditioning & Recovery Chair for Physiotherapy Clinics
- Biotronix DVT Pump Air Compression Therapy Machine Digital Remote Controlled
- Multi Mode Brain Stimulation Device TDCS TPCS TACS TVNS
- Phymaster Shockwave Shockwave Therapy Device for Orthopedic Clinics
- tDCS tPCS tACS tVNS Neuro Rehab Therapy Device for Brain Stimulation
- Konmed KM-560 Rehab Biofeedback Equipment with EMG Analysis
- High Performance Neuro Rehab Device tDCS tACS tVNS tPCS
- Konmed KM-560 Biofeedback Machine for Pelvic Muscle Training
- Hospital Grade Brain Stimulation System With TVNS
- Phymaster Shockwave Shockwave Therapy Device for Muscle Pain Relief
- Konmed KM-560 EMG Biofeedback Device with Therapy Modes
- Advanced Transcranial Direct And Alternating Current Stimulation System
- Professional Brain Stimulation System tDCS tACS tVNS tPCS
- Konmed KM-560 EMG Biofeedback Machine with Gaming Mode
- Konmed KM-560 Professional Biofeedback Equipment with EMG
- Konmed KM-560 EMG Biofeedback System with Multi Mode
- Konmed KM-560 EMG Biofeedback Equipment for Physiotherapy
- Konmed KM-560 Rehab Biofeedback Machine with ETS
- Advanced Brain Therapy System tDCS tACS tVNS tPCS Equipment
- Konmed KM-560 EMG Biofeedback Stimulator with ETS Technology
- Advanced Neuro Therapy Machine tDCS tACS tVNS tPCS
- Konmed KM-560 4 Channel EMG Biofeedback System for Therapys
- Portable Brain Therapy System tDCS tACS tVNS tPCS
- Konmed KM-560 Biofeedback Machine with Electrical Stimulations
- Portable Magnetic Magneto Physiotherapy Equipment for Pain Relief Sport Injuiry
- Professional Neuro Rehab Equipment tDCS tACS tVNS tPCS
- Best motorized pedaling machine for therapy
- Advanced Pedo Cycle Pedal Motorized Electrical Exerciser with Automatic Settings
- Konmed KM-560 Physiotherapy EMG Biofeedback for Clinics
- Professional Grade Physiotherapy Laser Device for Chiropractic and Rehabilitation Centers
- Konmed KM-560 Rehab Biofeedback Equipment with Stimulator
- Exoskeleton-Based Rehabilitation for Controlled Joint Movement
- Electric physiotherapy Rehabilitation Device for Improved Mobility With Leg Protection Support
- DVT pump for travel Portable lymphedema solution Home lymphedema compression therapy
- Precision Controlled Brainwave Stimulation Equipment
- Compact Neuro Rehab Device tDCS tACS tVNS tPCS
- Pedo Cycle Adjustable motorized pedal exerciser for rehab physical therapy machine
- Professional Neuro Stimulation Machine tDCS tPCS tACS tVNS
- Pelvic Muscle Conditioning Chair for Long-Term Pelvic Health
- Compact Neuro Stimulation Device tDCS tPCS tACS tVNS
- Clinical Neuro Modulation Device tDCS tPCS tACS tVNS Equipment
- Neuro Modulation Device tDCS tPCS tACS tVNS
- Exoskeleton Therapy Devices for Progressive Recovery Programs
- Clinical Pelvic Floor Muscle Therapy Chair for Advanced Patient Care
- Konmed KM-560 EMG Biofeedback Machine for Clinical Use
- Konmed KM-560 Rehab Biofeedback Machine with Therapy Programs
- Konmed KM-560 Physiotherapy EMG Biofeedback with FES
- Professional Brain Stimulator tDCS tPCS tACS tVNS Equipment
- Advanced Electrotherapy Combo Unit 4 in 1 for Pain Management
- Laser Pain Management Device 750mw Physiotherapy Laser Treatment 910nm
- Konmed KM-560 Rehab Biofeedback Machine with EMG Feedback
- Digital Neuro Rehab Device tDCS tPCS tACS tVNS Equipment
- Clinical Brain Therapy System tDCS tACS tVNS tPCS
- High Performance Class 4 Laser Therapy Machine for Advanced Rehab
- Class IV Laser Therapy Device for Physiotherapy Hospitals
- Class IV Laser Therapy Device for Joint Pain and Muscle Recovery
- Electro Brain Rehab Machine tDCS tPCS tACS tVNS
- Konmed KM-560 EMG Biofeedback System with Therapy Programs
- Clinical Class 4 Laser Physiotherapy Equipment for Pain Treatment
- Konmed KM-560 Rehab Biofeedback Machine with Smart Interface
- Advanced Medical Class 4 Laser Therapy Equipment for Rehabilitation
- Smart Neuro Rehab Equipment tDCS tPCS tACS tVNS
- Clinical Neuro Stimulation System tDCS tPCS tACS tVNS
- Konmed KM-560 Pelvic Therapy Biofeedback Machine with EMG
- Compact Neuro Therapy Equipment tDCS tPCS tACS tVNS
- Digital Neuro Stimulation Device tDCS tPCS tACS tVNS
- Smart Brain Rehab Equipment tDCS tACS tVNS tPCS
- Cognitive And Psychiatric Therapy Neuromodulation System
- Smart Brain Stimulation Device tDCS tACS tVNS tPCS Machine
- Professional Multi Mode Neuro Stimulation Device
- Professional Neuro Modulation Therapy System For Clinics
- Economical Class IV Laser Therapy Machine for Orthopedic Rehabilitation
- Integrated Neurological Therapy And Brain Modulation Device
- Digital 4 in 1 Combo Therapy Machine with Multiple Modes
- Advanced Class IV Laser Therapy System for Physiotherapy Clinics
- Advanced Neuro Stimulation Equipment tDCS tPCS tACS tVNS Machine
- 4 in 1 Combo Therapy Device for Physiotherapy Pain Relief Treatment
- Professional Class 4 Laser Therapy Equipment for Pain Relief Therapy
- Four In One Brain And Vagus Stimulation Therapy Device
- Konmed KM-560 EMG Biofeedback Equipment with Muscle Activation
- Economical Model Class 4 Laser Therapy Equipment for Rehabilitation Clinics
- Professional Class 4 Laser Physiotherapy Equipment for Pain Management
- Advanced Cortical And Vagus Nerve Therapy System
- Professional Class IV Laser Physiotherapy Device for Rehabilitation Centers
- Konmed KM-560 Biofeedback Equipment with Gaming Rehabilitation
- 4 in 1 Electrotherapy Unit for Sports Injury & Muscle Recovery
- Multi Mode Brain Therapy Machine tDCS tPCS tACS tVNS Device
- Clinical TDCS TPCS TACS TVNS Neuro Rehabilitation Machine
- Electrotherapy Combo Machine 4 in 1 with Carry Case Portable
- Konmed KM-560 Pelvic Floor Biofeedback Machine with Rehab Features
- Digital 4 in 1 Physiotherapy Machine for Clinic & Home Therapy
- Non Invasive Transcranial Brain Stimulation Equipment
- Multi Current Brainwave Modulation System
- High Intensity Class 4 Laser Therapy Device for Pain Treatment
- Clinical Neurological Recovery And Stimulation Unit
- Stroke Rehabilitation Brain Stimulation Device
- High Power Class IV Laser Therapy Machine for Physiotherapy Clinics
- 4 in 1 Physiotherapy Combo Unit with TENS EMS IFT MS Therapy
- High Intensity Class 4 Laser Physiotherapy Equipment for Rehabilitation Clinics
- Semiconductor LASER Light Therapy Instrument Portable Compact Handheld Model Digital
- Clinical Brain and Neuro Rehab Machine tDCS tACS tVNS tPCS
- Exoskeleton Solutions for Precision-Guided Rehabilitation
- Digital Brain Therapy Equipment tDCS tACS tVNS tPCS Machine
- Phymaster Shockwave Pain Relief Machine for Joint & Muscle Therapy
- Professional Pelvic Muscle Rehabilitation Chair for Medical Facilities
- Physiotherapy Laser Pain Relief with Dual Probe Imported Probe Pre Program with 2 Year Warranty
- Back, Leg and Chest Strength Measurement Device for Physiotherapy Use
- Back, Leg and Chest Dynamometer for Accurate Muscle Strength Assessment
- Class IV Laser Therapy System for Orthopedic
- KM521 Advanced Pelvic Floor Muscle Rehab Equipment for Clinics
- Physio Tecar Physiothearpy Machine Massage
- Portable Combo Therapy Machine 4 in 1 for Muscle Therapy
- Advanced 4 in 1 Therapy Machine for Muscle Stimulation & Pain Relief
- Compact tDCS tPCS tACS tVNS Neuro Rehab Machine
- Compact 4 in 1 Therapy Machine for Physiotherapy Clinics
- Advanced Physiotherapy Device 4 in 1 for Nerve Stimulation
- Multifunction Electric Knee Massager with Heating Vibration and Kneading for Pain Relief and Recovery
- Economical Class IV Laser Therapy Machine for Pain Relief
- PEMT Pulse Electromagnetic Therapy Machine for Pain Relief
- Digital PEMT Loop Therapy Device for Physiotherapy Treatment
- Pulse Electromagnetic Therapy Machine Professional Model
- PEMT Loop Therapy Device Advanced Physiotherapy Equipment
- Wrist Continuous Passive Motion Device-Adjustable Range for Therapy Sessions
- Phymaster Shockwave Physiotherapy System for Orthopedic Rehabilitation
- Phymaster Shockwave Advanced Physiotherapy Machine for Clinics
- Phymaster Shockwave Shockwave Therapy Equipment for Physiotherapy Clinics
- Phymaster Shockwave Physiotherapy Device for Musculoskeletal Pain
- Phymaster Shockwave Physiotherapy System for Sports Injury Treatment
- Phymaster Shockwave Physiotherapy Machine for Muscle & Tendon Therapy
- Phymaster Shockwave Shockwave Therapy Device for Advanced Physiotherapy
- Phymaster Shockwave Shockwave Therapy Device for Chronic Pain Relief
- Phymaster Shockwave Physiotherapy Therapy Machine for Clinical Pain Relief
- Phymaster Shockwave Therapy Machine for Orthopedic Physiotherapy
- Phymaster Shockwave Shockwave Therapy Equipment for Sports Clinics
- Advanced Magnetic Pulse Therapy Device for Muscle, Joint & Sports Rehab
- Portable Neuro Modulation Machine tDCS tACS tVNS tPCS
- Smart Brain Therapy Equipment tDCS tPCS tACS tVNS
- Professional Shockwave Machine Therapy Pain Relief & ED Treatment Extracorporeal Shock Wave Therapy
- Cold Therapy Machine for Shoulder, Knee, Ankle & Back Treatment
- Neuro Rehabilitation Device tDCS tACS tVNS tPCS
- Digital Brain and Vagus Nerve Stimulator tDCS tACS tVNS tPCS
- Multi Mode Neuro Therapy Machine tDCS tACS tVNS tPCS
- Advanced Neuro Modulation Machine tDCS tPCS tACS tVNS for Rehab
- Brain Rehab Therapy Device tDCS tPCS tACS tVNS Equipment
- Advanced Brain Rehab Equipment tDCS tPCS tACS tVNS
- Electro Brain and Nerve Therapy Device tDCS tACS tVNS tPCS
- Multi Functional Neuro Therapy Equipment tDCS tPCS tACS tVNS
- Electro Neuro Therapy Machine tDCS tACS tVNS tPCS Device
- Economical Physiotherapy Laser Therapy Equipment Class IV
- Economical High Power Laser Therapy Machine Class IV
- PEMT Loop Therapy Device for Rehabilitation and Pain Treatment
- Economical Class IV Laser Therapy Equipment for Medical Use
- Professional Brainwave And Neural Activation Device
- Portable Neuro Therapy Machine tDCS tACS tPCS tVNS
- Hospital Approved Pelvic Floor Muscle Therapy Chair for Medical Use
- Dynamic Motion Wrist Therapy - Adjustable Post-Surgery Device for Flexibility & Strength
- Electric Cold Therapy Unit for Pain Management Clinics
- Phymaster Shockwave Therapy Device for Orthopedic & Sports Pain
- Economical Class IV Laser Therapy Device for Physiotherapy Hospitals
- Compact 4 in 1 Physiotherapy Machine with LCD Display & Dual Channel
- UniGym Interactive Rehabilitation & Recovery Device
- Digital UniGym Rehab Machine for Post-Surgery and Stroke Patients
- Class IV Laser Therapy Device for Muscle Pain Rehabilitation
- Economical Class 4 Laser Therapy Equipment for Sports Injury Clinics
- High Intensity Class 4 Laser Therapy Device for Muscle Pain Treatment
- Class 4 Laser Therapy Device for Orthopedic Clinics
- Stroke Rehabilitation And Cognitive Therapy Device
- KM521 Pelvic Muscle Training Device with Colour Screen & Voice Alerts
- Clinical Class 4 Laser Therapy Equipment for Pain Management
- Phymaster Shockwave Physiotherapy Equipment for Sports Injury Therapy
- High Intensity Class 4 Laser Therapy Machine for Rehabilitation
- Cold Therapy Machine with Custom-Fit Shoulder, Knee, Ankle Pads
- Economical Class IV Laser Therapy Equipment for Rehabilitation Clinics
- High Intensity Class 4 Laser Therapy Device for Physiotherapy Centers
- Advanced Class IV Laser Therapy Equipment for Muscle Pain Relief
- Electrotherapy 4 in 1 Machine for Physiotherapy Pain Management
- Economical High Power Class 4 Laser Therapy System
- 4 in 1 Combo Therapy Machine for Back Pain & Joint Relief
- Wrist CPM Physiotherapy Machine-Electrical Motorized Model for Rehabilitation Centers
- Professional Electrotherapy Unit 4 in 1 with LCD Display
- 4 Mode Electrotherapy Combo Machine for Pain Therapy & Recovery
- Multi Mode 4 in 1 Physiotherapy Device for Pain Treatment
- 4 Channel Electrotherapy Combo Machine for Clinic & Home Use
- Portable Physiotherapy Combo Device 4 in 1 with Accessories
- High Power Class 4 Laser Therapy System for Rehabilitation Centers
- 4 in 1 Therapy Device for Physiotherapy Muscle Stimulation
- Economical Model Class IV Laser Therapy Machine for Pain Therapy
- Class IV Laser Therapy Device for Physiotherapy Clinics
- High Power Class IV Laser Therapy System for Physiotherapy Treatment
- Electromagnetic Radial ESWT Device Clinic-Grade Shockwave Therapy with Adjustable Frequency
- Medical Pelvic Floor Muscle Stimulation Chair for Physiotherapy Practice
- High Intensity Class 4 Laser Therapy Equipment
- Portable Electrotherapy Unit 4 in 1 with Multi Therapy Modes
- Digital Pelvic Muscle Training Chair for Incontinence & Rehabilitation
- Precision Controlled Transcranial Brain Therapy And Modulation System
- Multi Mode 4 in 1 Combo Machine for Physiotherapy Clinics
- Economical Class 4 Laser Therapy Machine for Physiotherapy Pain Clinics
- Class 4 Laser Therapy Machine for Muscle and Joint Pain Management
- Class 4 Laser Physiotherapy Equipment for Joint Pain Therapy
- Advanced Class IV Laser Therapy Device for Physiotherapy Pain Relief
- Professional Pelvic Floor Muscle Conditioning Chair for Hospitals
- PEMF Loop Therapy Device Premium Pain Relief Equipment
- High Intensity Class 4 Laser Physiotherapy Device for Clinics
- Revolutionizing Rehabilitation with Exoskeleton Technology
- Brain Neuro Stimulation Device tDCS tACS tVNS tPCS Machine
- Class IV Laser Therapy Equipment for Sports Injury Treatment
- KM-580 Advanced Biofeedback & Electrotherapy Device for Rehabilitation
- Class 4 Laser Therapy Equipment for Orthopedic Rehabilitation
- Cryopush Cold Compression Device for Faster Post-Surgery Healing
- Digital Focused Acoustic Shockwave Generator for Chronic Pain Management
- Class 4 Laser Therapy System for Muscle Pain Rehabilitation
- High Power Class 4 Laser Therapy Equipment for Muscle Pain Treatment
- Economical Class IV Laser Therapy Machine for Pain Relief Clinics
- Class 4 Laser Therapy Machine for Orthopedic Pain Management Clinics
- Biotronix Solution Forever Pedo Cycle Electrical Motorized Digital Remote Controlled Automatic
- Advanced Medical Class 4 Laser Therapy Device for Pain Management
- Chiropractic Adjusting Tool Spine and Back Massage Device
- Medical Grade Class IV Laser Therapy Machine
- Professional Class IV Laser Therapy System for Physiotherapy Centers
- Advanced Clinical Pelvic Muscle Stimulator Chair for Rehabilitation Therapy
- KM521 Pelvic Floor Muscle Therapy Device for Women & Men
- Exoskeleton Technology Advancing Patient-Centered Rehabilitation
- Ultra Precision Focus Shockwave Therapy Machine for Deep Orthopedic Recovery
- Back Leg Chest Strength Testing System for Rehab Professionals
- Pulse Electromagnetic Therapy Loop Machine Professional Model
- PEMF Loop Therapy Machine for Muscle Pain Rehabilitation
- From Support to Independence with Rehabilitation Exoskeletons
- PEMT Physiotherapy Loop Therapy Machine Premium Equipment
- PEMF Pulse Electromagnetic Therapy Equipment for Clinics
- Phymaster Shockwave Therapy Equipment for Rehab Treatment
- Orthopedic & Sports Back, Leg and Chest Muscle Testing Dynamometer
- Motorized Cold Compression Therapy System for Pain and Swelling Control
- Continuous Passive Motion Wrist Device-Advanced Model for Rehabilitation Centers
- Professional Neuro Stimulation Equipment tDCS tACS tVNS tPCS
- Professional Class 4 Laser Therapy Equipment for Physiotherapy Centers
- Phymaster Shockwave Physiotherapy Machine for Advanced Rehabilitation Care
- Phymaster Pain Management Physiotherapy Equipment
- Phymaster Shockwave Shockwave Therapy Machine for Musculoskeletal Pain
- Phymaster Shockwave Advanced Shockwave Therapy Equipment for Clinics
- Phymaster Shockwave Physiotherapy Device for Sports Injury Rehabilitation
- Phymaster Shockwave Physiotherapy Machine for Clinical Pain Management
- Phymaster Shockwave Pain Relief Therapy Machine for Physiotherapy Use
- Phymaster Shockwave Physiotherapy Equipment for Orthopedic Rehabilitation
- Phymaster Shockwave Physiotherapy Device for Advanced Pain Therapy
- Phymaster Shockwave Shockwave Therapy Device for Muscle & Joint Pain
- Phymaster Shockwave Advanced Physiotherapy Equipment for Rehabilitation
- Phymaster Shockwave Physiotherapy Equipment for Orthopedic Therapy
- Phymaster Shockwave Advanced Physiotherapy Pain Relief Equipment
- Digital Multi Current Brain Stimulation Machine
- Neuro Cognitive Enhancement Therapy Device
- Clinical Neuromodulation And Rehabilitation Equipment
- Advanced Neuro Electrical Stimulation Equipment
- Bio VNS-1 Digital Brain And Vagus Therapy System
- Professional Neurological Therapy And Modulation Machine
- Multi Current Transcranial Therapy Machine
- Digital TDCS TPCS TACS TVNS Neuromodulation Machine
- Stroke And Cognitive Rehabilitation Brain Device
- Precision Controlled Transcranial Therapy Machine
- Neuro Recovery And Cognitive Enhancement System
- Advanced Electrical Brain And Vagus Therapy Machine
- Integrated Brainwave Regulation Therapy Equipment
- Digital Focused Shockwave Therapy Equipment for Physiotherapy Clinics
- Medical Grade Brain And Vagus Stimulator
- Multi Protocol Transcranial Stimulation Equipment
- Wrist Rehabilitation CPM Device for Post-Surgery and Injury Recovery
- Professional Wrist CPM Therapy Machine for Hospitals and Rehab Clinics
- Digital Brainwave Synchronization And Vagus Nerve Therapy Device
- Advanced Wrist Joint CPM Machine-Digital and Motorized for Physiotherapy
- Medical Grade Brain And Autonomic Nervous System Stimulator
- Professional Ankle CPM Equipment With Adjustable Motion Intensity
- Ankle Rehabilitation CPM With Controlled Range Of Motion Training
- Digital Brain Activation And Neuro Recovery Machine
- Medical Cold Therapy Device for Post-Surgery Recovery
- Digital Cooling System with Motorized Compression for Joint Care
- Cryopush Motorized Cold Compression Machine for Clinic Use
- RegenPlus Focus Shockwave Therapy Device for Accelerated Tissue Healing
- KM521 Digital Pelvic Floor Muscle Therapy Equipment for Rehabilitation
- KM521 Advanced Pelvic Floor Muscle Therapy Device for Rehab Use
- KM521 Pelvic Muscle Stimulator with TFT Colour Display for Therapy
- KM521 Rechargeable Pelvic Floor Muscle Therapy Machine for Clinics
- Back Leg Chest Muscle Strength Analyzer for Rehabilitation Assessment
- Clinical Pelvic Muscle Stimulation Chair for Post-Surgery Recovery
- Clinical Pelvic Muscle Strength Training Chair for Rehabilitation Clinics
- Professional Pelvic Muscle Rehabilitation Chair for Hospitals & Clinics
- Clinical Pelvic Muscle Strengthening Chair for Incontinence Management
- Pelvic Floor Therapy Chair for Safe, Non-Invasive Muscle Strengthening
- Medical Pelvic Muscle Stimulation Chair for Complete Pelvic Care
- Medical Grade Pelvic Muscle Training Chair for Physiotherapy Use
- Smart Pelvic Floor Muscle Training Chair for Long-Term Clinical Use
- Medical Pelvic Muscle Therapy Chair for Post-Surgical Rehabilitation
- Advanced Pelvic Floor Muscle Rehab Chair for Hospital Physiotherapy
- Pelvic Floor Muscle Strength & Control Chair for Medical Centers
- Digital Pelvic Floor Muscle Activation Chair for Pelvic Health Therapy
- Clinical Pelvic Floor Muscle Rehab Chair for Post-Treatment Recovery
- Non-Invasive Pelvic Floor Muscle Training Chair for Patient Recovery
- Exoskeleton Technology Advancing Modern Rehabilitation Care
- Exoskeleton-Assisted Therapy for Faster Functional Recovery
- Precision Rehabilitation Using Medical Exoskeleton Devices
- Exoskeleton Solutions for Motor Recovery and Muscle Re-education
- Wearable Exoskeleton Technology for Assisted and Active Therapy
- Exoskeleton-Assisted Physiotherapy for Stroke and Spinal Injuries
- Medical-Grade Exoskeletons for Professional Physiotherapy Use
- Robotic Exoskeleton Therapy for Advanced Neurological Rehabilitation
- Clinical-Grade Exoskeletons for Modern Physiotherapy Practice
- Phymaster Shockwave Physiotherapy System for Orthopedic Pain Care
- Phymaster Shockwave Shockwave Therapy Equipment for Muscle Recovery
- High Intensity Class 4 Laser Therapy Equipment for Modern Rehab Clinics
- High Power Class 4 Laser Therapy Equipment for Advanced Treatment
- Medical Class 4 Laser Therapy System for Pain and Rehab Care
- Advanced Class 4 Laser Therapy Equipment for Professional Rehabilitation
- High Intensity Class 4 Laser Therapy Machine for Deep Tissue Recovery
- 7 Head ESWT Machine-Pneumatic Radial Shockwave Therapy with Adjustable Energy
- User-Friendly DVT 6 Chamber System with Touch Controls and Remote Access
- Dual Probe Laser Pain Treatment Laser Pain Relief Therapist Tool
- Radial ESWT Therapy System-7 Heads, Adjustable Frequency
- Compact Radial ESWT Device-Digital Touch Screen, Adjustable Energy & Frequency
- Ankle CPM Equipment With Speed Control And Angle Indication Display
- Medical Exoskeleton Systems for Structured Therapy Programs
- KM521 Digital Pelvic Floor Muscle Strengthening System for Rehab
- Clinical Class 4 Laser Physiotherapy Machine for Rehabilitation Clinics
- High Intensity Class IV Laser Therapy Equipment for Pain Management
- Class 4 Laser Therapy Equipment for Sports Injury Pain Relief
- High Power Class IV Laser Therapy Machine for Orthopedic Therapy
- Class IV Laser Therapy Device for Physiotherapy Treatment
- Phymaster Shockwave Physiotherapy Device for Muscle Recovery Treatment
- Phymaster Shockwave Physiotherapy Equipment for Professional Clinics
- High Intensity Class IV Laser Therapy Machine for Pain Relief Clinics
- Class 4 Laser Therapy Machine for Joint Pain and Rehabilitation Clinics
- Economical Model Class IV Laser Therapy Equipment
- Advanced Class IV Laser Therapy Device for Muscle Pain Treatment
- Class IV Laser Therapy Equipment for Physiotherapy and Pain Relief Centers
- Economical Class 4 Laser Therapy Machine for Physiotherapy Pain Relief Centers
- High Power Class IV Laser Therapy Equipment for Orthopedic Rehabilitation Clinics
- Class IV Laser Therapy System for Sports Injury and Muscle Recovery
- Economical Model Class 4 Laser Therapy Equipment for Pain Management Clinics
- Advanced Class IV Laser Therapy Machine for Physiotherapy Rehabilitation
- High Intensity Class 4 Laser Therapy Device for Joint Pain Treatment
- Class 4 Laser Therapy Machine for Orthopedic and Sports Rehabilitation
- Economical Class IV Laser Therapy Equipment for Pain Relief Therapy
- Professional Class 4 Laser Therapy Equipment for Physiotherapy Clinics
- High Power Class IV Laser Therapy System for Muscle Pain Relief
- Economical Model Class IV Laser Therapy Machine for Physiotherapy Centers
- Advanced Class IV Laser Therapy Equipment for Sports Injury Rehabilitation
- Class 4 Laser Therapy Machine for Physiotherapy Clinics
- Professional Class IV Laser Therapy Machine for Orthopedic Clinics
- High Power Class 4 Laser Therapy Equipment for Pain Relief Centers
- Economical Class 4 Laser Therapy Equipment for Rehabilitation Therapy
- Class 4 Laser Therapy Machine for Physiotherapy and Rehabilitation Clinics
- High Power Class 4 Laser Therapy Equipment for Daily Physiotherapy Use
- Economical Model Class IV Laser Therapy Device for Pain Treatment Clinics
- Class IV Laser Therapy Equipment for Pain Relief and Physiotherapy Use
- Economical Class 4 Laser Therapy Machine for Pain Relief and Physiotherapy
- Professional Class 4 Laser Physiotherapy Device for Orthopedic Pain Treatment
- Economical Model Class 4 Laser Therapy Equipment for Physiotherapy Clinics
- Advanced Class IV Laser Therapy System for Pain Relief Therapy
- Phymaster Shockwave Physiotherapy Machine for Deep Pain Relief Therapy
- Class IV Laser Therapy System for Sports Injury and Pain Management
- 4 in 1 Electrotherapy Combo Unit Digital Physiotherapy Device
- Portable Electrotherapy Combo Device 4 in 1 for Rehabilitation
- Multi Function 4 in 1 Combo Therapy Device for Clinic & Home Use
- Digital 4 in 1 Electrotherapy Equipment for Pain Management
- 4 in 1 Combo Electrotherapy Unit with Carry Case & Accessories
- Lightweight 4 in 1 Physiotherapy Unit for Home & Clinical Treatment
- Digital Combo Therapy Device 4 in 1 for Muscle & Nerve Stimulation
- Portable 4 in 1 Physiotherapy Combo Machine with LCD Screen
- 4 in 1 Electrotherapy Device for Muscle Recovery & Rehab Use
- Digital Electrotherapy 4 in 1 Unit with Dual Channel Output
- 4 in 1 Electrotherapy Combo Unit for Professional Rehabilitation
- 4 in 1 Electrotherapy Machine for Joint Pain & Rehabilitation
- Digital tDCS tACS tVNS tPCS Brain Stimulator for Clinics
- Professional tDCS tPCS tACS tVNS Neuro Therapy Device
- Phymaster Shockwave Shockwave Therapy Device for Rehab Applications
- Digital Brain Stimulator tDCS tACS tVNS tPCS Machine
- Professional Neuro Modulation Device tDCS tPCS tACS tVNS
- Portable Brain Therapy Device tDCS tACS tVNS tPCS
- 4 Mode tDCS tACS tPCS tVNS Brain Stimulator Machine
- tDCS tPCS tACS tVNS Neuro Therapy Device for Brain Rehabilitation
- Brain Rehab Device tDCS tPCS tACS tVNS
- Digital Brain Therapy Device tDCS tPCS tACS tVNS
- Portable Neuro Stimulator tDCS tACS tVNS tPCS
- High Efficiency Brain Therapy Device tDCS tACS tVNS tPCS
- Clinical Brain Stimulation Device tDCS tPCS tACS tVNS
- Digital Brain Therapy System tDCS tPCS tACS tVNS
- Clinical Neuro Stimulator Device tDCS tACS tVNS tPCS
- Clinical Neuro Rehab Equipment tDCS tPCS tACS tVNS Device
- Motorized pedal device Electric therapy bike Motorized recovery bike Electric physio pedal Motorized pedal tool
- All-in-One UniGym Rehabilitation Equipment for Clinics and Home
- Smart Pelvic Floor Trainer with Biofeedback Support
- Class 4 Laser Therapy Machine for Orthopedic Pain Management
- High Intensity Class 4 Laser Physiotherapy Machine for Pain Relief
- Economical Class 4 Laser Therapy Equipment for Physiotherapy Hospitals
- Professional Class IV Laser Therapy Machine for Pain Clinics
- Class IV Laser Therapy Device for Sports Injury Rehabilitation
- Economical Model Class 4 Laser Therapy Device for Medical Clinics
- Class 4 Laser Therapy Equipment for Muscle Injury Treatment
- Economical Model Class IV Laser Therapy Device for Pain Therapy
- Clinical Class 4 Laser Physiotherapy Equipment for Pain Relief
- Advanced Class IV Laser Therapy Machine for Physiotherapy Treatment
- High Intensity Class IV Laser Therapy System for Orthopedic Clinics
- Economical Class 4 Laser Therapy Device for Rehabilitation Therapy
- Professional Class IV Laser Therapy Equipment for Physiotherapy Clinics
- Class 4 Laser Therapy Machine for Sports Medicine Clinics
- High Power Class IV Laser Therapy Device for Pain Management
- Economical Model Class 4 Laser Therapy Machine for Clinics
- High Power Class IV Laser Therapy Machine for Sports Injury Clinics
- Economical Class 4 Laser Therapy Device for Physiotherapy Pain Relief
- Class IV Laser Therapy Machine for Physiotherapy Rehabilitation
- Economical Model Class 4 Laser Physiotherapy System for Clinics
- Class IV Laser Therapy System for Orthopedic Pain Treatment
- Class IV Laser Therapy Equipment for Muscle Pain Relief
- Class 4 Laser Therapy Machine for Joint Pain Management
- Professional Class 4 Laser Therapy Device for Pain Management
- Class 4 Laser Therapy Equipment for Sports Medicine Clinics
- Economical Model Class IV Laser Therapy Device for Pain Relief Therapy
- Clinical Class 4 Laser Physiotherapy Machine for Rehabilitation Centers
- Class IV Laser Therapy Device for Physiotherapy and Rehabilitation
- Professional Class IV Laser Therapy Equipment for Physiotherapy Hospitals
- High Power Class 4 Laser Therapy Device for Muscle Injury Treatment
- Class 4 Laser Therapy System for Orthopedic Rehabilitation
- Advanced Class IV Laser Therapy Equipment for Physiotherapy Centers
- High Intensity Class 4 Laser Physiotherapy Device for Pain Management
- Class IV Laser Therapy Machine for Pain Relief and Rehabilitation Clinics
- Economical Class IV Laser Therapy Equipment for Physiotherapy Clinics
- High Power Class 4 Laser Therapy Machine for Pain Relief Treatment
- Economical Model Class 4 Laser Therapy System for Pain Relief Clinics
- Class IV Laser Therapy Machine for Physiotherapy Treatment
- High Intensity Class 4 Laser Therapy Equipment for Sports Injury Clinics
- High Intensity Class 4 Laser Therapy Device for Orthopedic Clinics
- Economical Class 4 Laser Therapy Machine for Pain Relief Clinics
- High Power Class IV Laser Therapy Equipment for Physiotherapy Hospitals
- Professional Class 4 Laser Physiotherapy Machine for Rehabilitation Therapy
- Class IV Laser Therapy Device for Orthopedic Pain Relief Treatment
- Advanced Class IV Laser Therapy System for Sports Injury Rehabilitation
- High Intensity Class 4 Laser Therapy Machine for Physiotherapy Centers
- Class IV Laser Therapy Machine for Physiotherapy and Pain Treatment
- Phymaster Shockwave Shockwave Therapy Machine for Clinical Applications
- Clinical Class 4 Laser Physiotherapy Equipment for Rehabilitation
- Class 4 Laser Therapy Device for Sports Injury Treatment
- High Power Class IV Laser Therapy Machine for Rehabilitation Centers
- Class 4 Laser Therapy Equipment for Muscle and Joint Pain Relief
- Economical Model Class IV Laser Therapy Equipment for Pain Treatment
- Class 4 Laser Therapy Device for Rehabilitation Clinics
- Professional Class 4 Laser Therapy System for Pain Management Clinics
- High Intensity Class 4 Laser Physiotherapy Device for Rehabilitation Therapy
- Class IV Laser Therapy Equipment for Pain Relief and Recovery Clinics
- Economical Model Class IV Laser Therapy Equipment for Sports Injury Clinics
- Class 4 Laser Therapy System for Orthopedic Rehabilitation Clinics
- High Power Class 4 Laser Therapy Device for Joint Pain Treatment
- Professional Class IV Laser Therapy Machine for Physiotherapy Clinics
- Economical Class 4 Laser Therapy Equipment for Rehabilitation Centers
- Advanced Class IV Laser Therapy Equipment for Physiotherapy Treatment
- High Intensity Class 4 Laser Therapy Machine for Muscle Injury Treatment
- Class IV Laser Therapy Device for Physiotherapy Rehabilitation Centers
- Economical Model Class IV Laser Therapy System for Hospitals
- Medical Class 4 Laser Therapy Machine for Physiotherapy Treatment
- Economical Class IV Laser Therapy Machine for Sports Injury Recovery
- Advanced Class IV Laser Therapy Device for Orthopedic Clinics
- Economical Physiotherapy Laser Therapy Machine Class 4
- Class 4 Laser Therapy Device for Physiotherapy and Sports Rehab
- Phymaster Shockwave Physiotherapy Machine for Sports Injury Rehab
- Phymaster Shockwave Physiotherapy Equipment for Advanced Rehab
- High Power Class IV Laser Machine for Rehabilitation Therapy
- Economical Model Class 4 Laser Therapy Device for Clinics
- High Intensity Class IV Laser Therapy Machine for Pain Treatment
- High Power Class 4 Laser Therapy Equipment for Pain Relief
- Professional Class IV Laser Physiotherapy Machine for Rehabilitation
- Economical Class IV Laser Therapy Device for Physiotherapy Clinics
- Class 4 Laser Therapy Machine for Sports Injury Rehabilitation
- Economical Model Class IV Laser Therapy Equipment for Pain Relief
- Class IV Laser Therapy Equipment for Orthopedic Pain Treatment
- Clinical Class IV Laser Therapy Equipment for Rehabilitation Therapy
- Economical Model Class 4 Laser Therapy System for Hospitals
- Advanced Class IV Laser Therapy Equipment for Joint Pain Relief
- Economical Class IV Laser Therapy Equipment for Pain Treatment
- Economical Model Class 4 Laser Therapy System for Physiotherapy Clinics
- Class IV Laser Therapy Equipment for Physiotherapy Rehabilitation
- High Power Class 4 Laser Physiotherapy Machine for Clinics
- Professional Class IV Laser Physiotherapy Machine for Clinics
- Professional Class IV Laser Therapy Device for Rehabilitation Centers
- Economical Class 4 Laser Therapy Device for Pain Relief Therapy
- Advanced Class IV Laser Therapy Equipment for Pain Management
- Class 4 Laser Therapy System for Physiotherapy Pain Management
- Economical Class IV Laser Therapy Equipment for Medical Rehabilitation
- Clinical Class 4 Laser Therapy Machine for Physiotherapy Centers
- Advanced Class IV Laser Therapy Device for Joint Pain Treatment
- High Power Class 4 Laser Physiotherapy System for Pain Relief
- Class 4 Laser Therapy Equipment for Orthopedic Rehabilitation Clinics
- Class 4 Laser Therapy Machine for Muscle and Joint Pain Relief
- Phymaster Shockwave Physiotherapy Equipment for Joint Pain Relief
- Advanced Class IV Laser Therapy Equipment for Sports Medicine Clinics
- Economical Class IV Laser Therapy Machine for Rehabilitation Centers
- Economical Class 4 Laser Therapy Equipment for Pain Relief Clinics
- Professional Class IV Laser Therapy Equipment for Pain Management
- Economical Model Class 4 Laser Therapy Machine for Physiotherapy Clinics
- Advanced Class IV Laser Therapy System for Orthopedic Pain Relief
- High Intensity Class IV Laser Therapy Device for Medical Clinics
- Economical Class 4 Laser Therapy Machine for Rehabilitation Therapy
- Class IV Laser Therapy Equipment for Physiotherapy Pain Relief
- Economical Class IV Laser Therapy Device for Orthopedic Clinics
- Advanced Class IV Laser Therapy Equipment for Pain Relief Therapy
- Konmed KM-560 EMG Biofeedback Device with Muscle Rehab
- Phymaster Shockwave Physiotherapy Machine for Non-Invasive Treatment
- Konmed KM-560 Rehab Biofeedback Equipment with NMES Stimulation
- Exoskeleton-Based Rehabilitation Supporting Motor Skill Development
- Exoskeleton Technology for Long-Term Rehabilitation Outcomes
- Konmed KM-560 EMG Biofeedback Equipment with FES Stimulator
- Phymaster Shockwave Non-Invasive Pain Relief Therapy Device
- Phymaster Shockwave Physiotherapy Pain Relief Equipment
- Phymaster Shockwave Therapy Device for Deep Tissue Pain Treatment
- Clinical Grade Class 4 Laser Therapy System for Physiotherapy Experts
- Phymaster Shockwave Therapy Device for Orthopedic & Sports Injury Treatment
- Advanced Brain and Neuro Modulation Device tDCS tPCS tACS tVNS
- Advanced Brain Therapy Equipment tDCS tPCS tACS tVNS
- Rotating dial inclinometer Inclinometer with swivel head
- Clinical Brain Rehab System tDCS tACS tVNS tPCS Equipment
- Exoskeleton-Assisted Therapy for Safe Functional Recovery
- DVT 6 Chamber Digital Air Compression Device For Circulation And Recovery
- Professional Focused Shockwave Therapy Machine for Clinic and Hospital Use
- High Power Class 4 Laser Therapy Machine for Pain Relief Physiotherapy Clinics
- Electro Neuro Stimulation Machine tDCS tACS tVNS tPCS
- Multi Mode Brain Therapy Device tDCS tPCS tACS tVNS
- Multi Mode Brain Stimulator tDCS tPCS tACS tVNS Device
- Compact Physiotherapy 4 in 1 Combo Unit for Professional Use
- Pneumatic Compression Device Leg Pump Machine for Lymphedema, Circulation and Swelling Fullset
- Exoskeleton-Based Physiotherapy Supporting Motor Relearning
- Exoskeleton Rehabilitation Systems Supporting Neuroplasticity
- Exoskeleton Solutions for Upper Limb and Lower Limb Recovery
- High Intensity Class 4 Laser Therapy Device for Pain Management Clinics
- High Power Class 4 Laser Therapy Equipment for Pain Management Clinics
- Advanced Focus Shockwave System for Targeted Muscle & Tendon Healing
- Digital DVT Machine 6 Chamber For Lymphedema And Blood Circulation Therapy
- Medical Exoskeleton Devices for Neuromuscular Rehabilitation
- Economical Model Class IV Laser Therapy Machine for Sports Injury Therapy
- High Intensity Class 4 Laser Therapy Equipment for Pain Clinics
- Supreme Focus Shockwave Therapy System for Advanced Pain Solutions
- Magneto Therapy Magnetotherapy for Pain Relief Electromagnetic
- Exoskeleton Technology Supporting Safe Motor Relearning
- High Power Class 4 Laser Therapy Device for Pain Management
- High Intensity Class 4 Laser Therapy Device for Sports Injury Clinics
- Class 4 Laser Therapy Machine for Orthopedic Pain Relief
- Advanced Class 4 Laser Therapy System for Physiotherapy Centers
- Advanced Class 4 Laser Therapy System for Physiotherapy Clinics
- Pain relief machine monopolar tecar therapy RET CET RF diathermy equipment physical device
- Professional RF Tecar Therapy Equipment for Physiotherapy Clinics, For Sports Injury Recovery
- Economical Physiotherapy Laser Therapy Device Class 4
- Hospital Approved Pelvic Muscle Rehabilitation Chair for Medical Use
- PlasticsABS Physiotherapy Extracorporeal Magnetic Therapy Physio Magneto Therapy
- Electromagnetic Pulse Therapy Loop Device for Rehabilitation Centers
- Pulse Electromagnetic Therapy Equipment for Rehabilitation Clinics
- Pulse Electromagnetic Therapy Device Clinical Physiotherapy Model
- Medical Grade Pelvic Muscle Strengthening Chair for Physiotherapy Use
- Portable Handheld Chiropractic Adjusting Tool Manual 4-Head Spine Massager
- Advanced Pelvic Floor Muscle Rehab Chair for Medical Facilities
- Cryopush Digital Cold Therapy Machine for Post-Surgery Rehab
- Pelvic Floor Muscle Activation Chair for Post-Surgical Rehabilitation
- Portable Analog Bubble Inclinometer for Shoulder, Knee, and Spine Evaluation
- Clinical Grade Pelvic Floor Muscle Training Chair for Rehabilitation Centers
- High-Performance Diode Laser Therapy Device for Physiotherapy and Rehabilitation Clinics
- Professional Pelvic Muscle Activation Chair for Clinic
- Electric Cryo Cooling Unit Ideal for Orthopedic and Rehab Use
- Professional Brain And Autonomic Activation Equipment
- Advanced Electrical Transcranial And Vagus Therapy Equipment
- Non-Surgical Pelvic Floor Therapy Chair for Incontinence Management
- Professional Back Leg Chest Dynamometer for Accurate Rehab Monitoring
- Back Leg Chest Strength Assessment Tool for Sports Medicine
- Medical-Grade Pneumatic Clinical Radial ESWT Equipment 7 Heads 10 Modes
- Inclinometer for spinal assessment Clinically approved and Leak-proof therapy inclinometer
- Portable digital hand grip measurement device
- Hand grip dynamometer digital monitor
- Combination Therapy Combi 2 in one UST 1 Mhz Digital Display TENS 4 Ch Digital Auto Mode Metallic
- Digital hand strength tester Portable grip strength tester
- Medical Grade Pelvic Muscle Training Chair for Womens Health Clinics
- 360 Degree Bubble Inclinometer Goniometer for Physical Therapy and Rehabilitation
- Phymaster Shockwave Pain Relief Device for Physiotherapy Treatment
- Stainless Steel IASTM Tools 5 pc 304 Grade Graston Tools
- Konmed KM-560 Biofeedback Stimulator with NMES FES Technology
- Compact Class 4 Laser Therapy Unit for Physiotherapy, Rehabilitation & Sports Injuries
- Konmed KM-560 EMG Biofeedback System with Multi Channel Mode
- Konmed KM-560 Biofeedback Machine with EMG Muscle Training
- Konmed KM-560 EMG Biofeedback Device with Muscle Therapy
- Konmed KM-560 4 Channel EMG Biofeedback Device for Clinic
- Konmed KM-560 4 Channel EMG Biofeedback System for Hospitals
- Konmed KM-560 EMG Biofeedback Machine for Sports Recovery
- physiotherapy UST 1 MHz TENS 4 channel combo therapy machine
- 7 Head Pneumatic Radial ESWT Therapy Machine-Pain Relief & Musculoskeletal Recovery
- Stepper Fitness Motorized Electric Mini Exercise Bike/Pedal Exerciser
- professional physiotherapy advanced physiotherapy device
- PEMT Pulse Electromagnetic Therapy Loop Machine for Physiotherapy
- Ultrasound And Tens Combo Electrotherapy Physiotherapy UST 1 Mhz and TENS 4 ch Auto Mode
- 4 in 1 Combo TENS EMS IF RUSS Digital Therapy Machine for Pain Relief and Muscle Stimulation
- Hospital Grade Pelvic Muscle Rehabilitation Chair for Patient Recovery
- Advanced Elbow CPM Machine-Adjustable Angles for Effective Joint Recovery
- Electric Elbow CPM Therapy Device
- Robotic Exoskeleton Therapy for Structured Recovery Programs
- Wearable Rehabilitation Exoskeletons Supporting Motor Recovery
- Biotronix Shock Wave Machine Shockwave Therapy Machine Extracorporeal Equipment Digital ESWT
- Konmed KM-560 EMG Biofeedback Stimulator with NMES
- Konmed KM-560 EMG Biofeedback System for Muscle Training
- Konmed KM-560 EMG Biofeedback Stimulator for Muscle Therapy
- Konmed KM-560 Rehab Biofeedback Machine with Gaming Therapy
- Konmed KM-560 EMG Biofeedback Stimulator with Therapy Modes
- Konmed KM-560 Pelvic Floor Biofeedback Machine for Rehab
- Konmed KM-560 4 Channel EMG Biofeedback Device for Physiotherapy
- Konmed KM-560 4 Channel EMG Biofeedback Device
- Konmed KM-560 Rehab Biofeedback Machine with EMG Output
- Konmed KM-560 EMG Biofeedback System for Physiotherapy Clinics
- Konmed KM-560 Rehab Biofeedback Equipment
- Konmed KM-560 Physiotherapy EMG Biofeedback with Gaming
- Konmed KM-560 Biofeedback Equipment with Gaming Rehab
- Konmed KM-560 4 Channel EMG Biofeedback System
- Konmed KM-560 Biofeedback Machine with Electrical Stimulation
- Konmed KM-560 Clinical EMG Biofeedback Device
- Konmed KM-560 Physiotherapy Biofeedback Device with EMG
- Konmed KM-560 EMG Biofeedback System for Muscle Recovery
- Konmed KM-560 Advanced Biofeedback Device for Clinics
- Konmed KM-560 Biofeedback Machine for Pelvic Therapy
- Konmed KM-560 4 Channel EMG Biofeedback Device for Rehab
- Konmed KM-560 Rehab Biofeedback Equipment with Therapy
- Konmed KM-560 Professional Biofeedback Machine for Clinics
- Konmed KM-560 Biofeedback Device with FES NMES
- Konmed KM-560 EMG Biofeedback Device for Swallow Therapy Training
- Konmed KM-560 Biofeedback System with Gaming Based Therapy Interface
- Konmed KM-560 EMG Biofeedback Machine for Pelvic Floor Rehabilitation
- Konmed KM-560 Physiotherapy Biofeedback Device with Multi Therapy Options
- Konmed KM-560 4 Channel EMG Biofeedback Equipment with Rehab Programs
- Konmed KM-560 EMG Biofeedback Device with 4 Channel Therapy System
- Konmed KM-560 EMG Biofeedback System for Recovery
- Konmed KM-560 EMG Biofeedback Device with Muscle Rehabs
- Konmed KM-560 4 Channel EMG Biofeedback System for Rehabs
- Digital Biofeedback Pelvic Trainer for Physiotherapy Use
- Clinical Pelvic Floor Trainer for Incontinence and Women Therapy
- Professional Pelvic Muscle Trainer for Postpartum Recovery and Strength
- 4 in 1 Electrotherapy Combo Unit TENS EMS IFT Muscle Stimulator Machine
- Medical Grade Pelvic Floor Trainer KM533-4 with Smart Biofeedback Technology
- Digital 4 Channel Electrotherapy Machine for Pain Relief & Rehabilitation
- Konmed KM-560 Physiotherapy Rehab Machine with EMG Monitoring
- Konmed KM-560 4 Channel EMG Biofeedback System for Physiotherapy Clinics
- Konmed KM-560 4 Channel Rehab Device with Biofeedback Training Modes
- Konmed KM-560 Multi Function Physiotherapy Machine with EMG Feedback
- Konmed KM-560 EMG Biofeedback Device for Orthopedic Physiotherapy
- Konmed KM-560 Rehabilitation Machine with Biofeedback and EMS
- Konmed KM-560 4 Channel Physiotherapy Biofeedback System with EMS
- Konmed KM-560 EMG Rehab Device for Muscle Strength & Recovery
- Konmed KM-560 Digital EMG Biofeedback Equipment with Therapy Programs
- Konmed KM-560 Multi Channel Physiotherapy Machine with EMG Feedback
- Konmed KM-560 4 Channel EMG Biofeedback Unit with Therapy Functions
- Konmed KM-560 Professional Rehab Device with EMG Signal Monitoring
- Konmed KM-560 EMG Biofeedback Machine for Neurological Rehabilitation
- Konmed KM-560 Advanced Rehab System with EMG Controlled Therapy
- Konmed KM-560 EMG Muscle Activation Device for Physiotherapy Clinics
- Konmed KM-560 4 Channel Electrotherapy Machine with Biofeedback System
- Konmed KM-560 EMG Biofeedback Equipment for Hospitals and Clinics
- Konmed KM-560 Physiotherapy System for Muscle Recovery and Strength
- Advanced Pelvic Floor Trainer with Biofeedback for Women Rehabilitation
- Pelvic Muscle Rehabilitation Device for Women Health Therapy
- Physiotherapy Pelvic Floor Trainer for Clinic and Home Use
- Portable Pelvic Trainer for Kegel Exercises and Daily Therapy
- Pelvic Floor Strengthening Device for Core Stability and Recovery
- Pelvic Muscle Strength & Recovery Chair for Medical Facilities
- Clinical use inclinometer Portable therapy inclinometer
- Phymaster Shockwave Shockwave Therapy System for Clinical Pain Relief
- Phymaster Shockwave Therapy Device for Muscle & Tendon Pain
- Phymaster Shockwave Professional Shockwave Therapy Equipment for Hospitals
- Phymaster Shockwave Physiotherapy Machine for Deep Pain Therapy
- Phymaster Shockwave Therapy Machine for Professional Pain Care
- Phymaster Shockwave Advanced Shockwave Pain Therapy Device
- Phymaster Physiotherapy Device for Chronic Pain Treatment
- Phymaster Shockwave Physiotherapy System for Joint Pain Treatment
- Phymaster Shockwave Therapy System for Pain Management
- Motorized pedal Motorized Leg Rehabilitation Device Pedo Cycle with Remote Control
- Portable motorized pedal exerciser for rehab exercises
- Advanced Exoskeleton Rehabilitation Devices for Clinical Use
- Exoskeleton Solutions for Neurological and Functional Recovery
- Smart Wearable Exoskeletons for Clinical Rehab Applications
- Advanced Exoskeleton Solutions for Clinical Physiotherapy
- Exoskeleton-Assisted Rehabilitation
- Cryopush Motorized Cooling and Compression Therapy Machine for Injury and Rehab Support
- KM521 Pelvic Floor Therapy Device with Colour Screen & Voice Alerts
- Imported Motorized Pedocycle Physiotherapy and Rehabilitation Equipment
- Pneumatic Shockwave Therapy Machine for Pain Relief and Rehabilitation
- Arm Exerciser with Digital Ergometer for Upper Limb Physiotherapy, For Home, Fitness Equipment
- Medical Rehabilitation Exoskeleton Robot for Neuro and Motor Recovery
- Shockwave Therapy Machine, Zinnor Extracorporeal ED Shock Wave Machine
- Automatic Legs And Arm Physiotherapy Device Motorized pedo cycle
- Advanced Wearable Exoskeleton Technology for Rehabilitation
- Pedo Cycle Electric pedal exerciser for rehabilitation exercises
- Medical-Grade DVT Compression Device for Hospital and Home Use
- Post-Surgery Elbow CPM Recovery Machine
- Professional Cold Compression Therapy Machine for Post-Surgery and Sports Injuries
- Phymaster Shockwave Pain Relief Equipment for Rehab Centers
- Digital Pelvic Muscle Therapy Chair for Pelvic Strength & Control
- Air Compression Therapy system DVT 8 chamber, Disposable
- Electric Massage Gun, 3-Level Chiropractic Adjustment Adjustment Tool, Complete Muscle
- UniGym Multifunction Physiotherapy and Rehabilitation Device
- Pneumatic ESWT System -7 Heads, Frequency 1-22 Hz, 10 Modes
- Radial ESWT Machine-7 Heads, 10 Clinical Modes
- Portable Focused Shockwave Therapy Machine for Orthopedic Care
- Knee Massager for joint pain relief- Physiotherapy for Arthritis Pain, Cramps, Knee Pain Relief
- Digital Clinical Pelvic Floor Rehabilitation Chair with Smart Control
- Radial ESWT Therapy System- Adjustable 1-16 mJ Intensity, 10 Modes
- T43 Radial ESWT Machine 10 Modes, 5-250Hz, 7 Heads
- T43 Electromagnetic Shockwave Therapy Device & Touch Screen
- Clinical-Grade PEMT Loop Therapy Machine-Adjustable Frequency & Deep Oscillation
- PEMT Loop Therapy Device-Pulse Electromagnetic Therapy with Deep Oscillation & Magnet Therapy
- Professional PEMT Loop Therapy Machine-Deep Tissue Penetration, Electromagnetic Pulse Therapy
- PEMT Loop Therapy Machine-Pulsed Electromagnetic Therapy Device for Pain Relief & Rehabilitation
- Home and Clinic Use DVT Air Compression Pump 4 Chamber Digital System
- 980nm 10W Class 4 Laser Therapy Physiotherapy Device With Touch Screen
- Laser Therapy Equipment Handy Model Portable Compact Home use and Clinical Pain Relief
- Multi-Function UniGym Motorized Therapy Device for Hospitals and Clinics
- Handheld Laser Therapy for Relieve Neck Back Arthritis Pain Rechargeable
- Wireless Physiotherapy Professional Pain Relief Ergonomic Handheld Laser Design
- Digital Focused Shockwave Therapy Equipment for Rehabilitation and Clinic Use
- Portable LASER therapy machine for pain relief Handheld LASER therapy tool
- Focused Shockwave Therapy System for Non-Invasive Pain Relief Treatment
- 10 Hz To 5 Khz ABS Plastic Physiotherapy Pain Relief Laser Therapy Machine, For Hospital
- Class 4 Diode Laser Therapy Machine 980nm 10W for Physiotherapy and Pain Relief
- Physiotherapy Laser Therapy Unit Dual Probe Advance Computerized Pre Program Dual Probe 750mw
- PEMT Loop Therapy Device for Muscle Recovery
- Phymaster Shockwave Therapy Machine for Musculoskeletal Disorders
- Clinical Pneumatic Shockwave Therapy Device 7 Heads
- Exoskeleton Rehabilitation Technology for Strength and Stability
- Digital laser pain relief equipment Physiotherapy laser treatment device Dual probe digital laser therapy
- Advanced Pelvic Floor Muscle Rehab Chair for Physiotherapy Clinicals
- LCD display laser therapy pain relief Physiotherapy laser with 46 pre-set programs
- physiotherapy laser therapy 750mw professional laser pain relief
- Professional 980nm Class 4 Diode Laser Therapy Machine for Effective Pain Relief, Tissue Regeneration, and Post-Surgery Rehabilitation
- Professional Elbow Rehabilitation CPM Device Imported Remote Controlled
- Back, Leg and Chest Dynamometer for Orthopedic & Neuro Rehab
- Medical Exoskeleton Technology for Assisted Rehabilitation
- Smart Wearable Exoskeletons for Functional Therapy Programs
- PEMT Loop Therapy 4500Hz Oscillation Equipment
- Radial Shockwave Therapy Equipment Accelerates Healing & Reduces Pain
- Heavy Duty Back Leg Chest Dynamometer for Rehab & Fitness Evaluation
- KM521 Clinical Pelvic Floor Muscle Training Equipment
- Durable Elbow Continuous Passive Motion Device for Post-Surgery Rehabilitation
- Home Use Elbow CPM Device for Faster Recovery
- High Performance Class 4 Laser Therapy System for Sports Rehabilitation
- Medical Pelvic Muscle Training Chair for Post-Treatment Recovery
- Infrared Diode Laser Therapy Machine for Deep Tissue Pain Relief and Healing
- Adjustable Elbow CPM Rehab Equipment
- Phymaster Shockwave Pain Management Device for Clinical Use
- PEMT Loop Therapy Machine-18cm Penetration, 1000-6000 Gauss Magnetic Intensity
- Pulse Electromagnetic Therapy Device-Large Single & Small Double Ring Handles, 850W Power
- Advanced Clinical Pelvic Floor Muscle Training Chair for Rehabilitation
- Imported Elbow CPM Machine Remote Controlled for Smooth Flexion & Extension
- Pneumatic Shockwave Clinical Radial ESWT Machine 7 Heads
- Professional 10W 980nm Diode Laser Therapy Machine
- Advanced Pelvic Floor Muscle Rehab Chair for Physiotherapy Clinics
- Electric Chiropractic Massager 400N, 4 Levels Force Spine Correction Massage, 10pcs Multifuction Massage Heads for Adjusting Chiropractic, Soothing Bones, Massage Acupuncture Points
- Pelvic Floor Muscle Strengthening Chair For Comprehensive Care
- Multi-Head Radial ESWT-7 Heads, Clinical Shockwave Therapy
- UniGym Complete Rehabilitation & Recovery Training System
- 7 Head Pneumatic Shockwave Clinical Radial ESWT Machine
- Precise ROM Evaluation with Advanced Bubble Inclinometer Physiotherapy Joint Angle Measurement
- Touch Screen Diode Laser Therapy Equipment 10W
- PMST NEO Plus Shockwave Therapy Machine for Pain Relief and Rehabilitation
- Shock Wave Therapy Device ED Shockwave Machine Physical Pain Therapy System Acoustic
- Bubble Inclinometer Physiotherapy Goniometer Measuring Tool Portable Joint Angle Measuring
- KM521 Digital Pelvic Muscle Stimulation Equipment for Rehabilitation
- 980nm Class IV Laser Therapy Device For Muscle Pain, Joint Pain, And Rehab
- Portable UniGym Motorized Exercise Therapy System
- Inclinometer for accurate measurement with easy-to-read scale Compact inclinometer for therapy
- Portable Motorised Cryo Cooling Unit for Home & Hospital Use
- Konmed KM-560 Biofeedback Machine for Pelvic Muscle Rehabs
- Radial ESWT Device -7 Heads, 0.5-1.2 Bar Energy, 10 Modes
- Konmed KM-560 4 Channel Physiotherapy Biofeedback Equipments
- Robotic Rehabilitation Tools for Stroke, Paralysis, and Injury Management
- Pneumatic shockwave therapy for muscle recovery after exercise
- Shockwave Therapy Pneumatic Digital Machine ESWT for Sports Injuries Physiotherapy & ED
- UniGym Multifunction Digital Physio Training Machine
- Shock Wave Machine Shockwave Therapy Machine Extracorporeal Equipment Digital ESWT
- Advanced PEMT Loop Machine-Deep Penetration & Multi-Pulse Therapy
- IASTM Stainless Steel Gua Sha Tools 5 Pieces Set for Myofascial Release
- Professional Myofascial Release Tools IASTM, Gua Sha Scraping Massage Tools, 5 Piece Medical Grade
- Silver Stainless Steel Quality Physical Therapy IASTM 5 pc Instruments for Rehabilitation, For Therapeutic Recovery Tools
- High-Quality Physical Therapy IASTM Imported 5 Pc Instruments for Clinics
- ultrasound TENS electrotherapy advanced physiotherapy equipment
- pain relief electrotherapy muscle recovery device ultrasound therapy unit
- ultrasound therapy machine TENS unit 4 channel combo electrotherapy unit
- combo therapy machine UST and TENS for pain relief
- Physical Therapy Bullet Based Pneumatic Shock Wave Therapy Equipment, For Clinical Purpose
- Shockwave Clinical Professional
- Bubble Inclinometer for clinical settings Therapy tool
- Portable Inclinometer 0-360 Degree Physiotherapy Inclinometer High Accuracy Measuring Tool
- Professional physiotherapy inclinometer Reliable bubble inclinometer
- Electric Knee Massager Infrared Heated Vibration Physiotherapy Osteoarthritis Rheumatoid Arthritis
- Shockwave Therapy Pneumatic ESWT For Plantar Fasciitis, For Hospital
- Bubble Inclinometer for Comprehensive ROM Testing Physiotherapy Rehabilitation PHD Students
- Adjustable swivel dial inclinometer High precision
- Shockwave therapy for chronic ankle instability and electrotherapy
- Professional physiotherapy Bubble inclinometer
- Pneumatic shockwave therapy for greater trochanteric pain syndrome
- Pneumatic shockwave therapy for calcific shoulder tendinitis
- Pneumatic shockwave therapy for shoulder calcific deposits
- Shockwave therapy for osteopathy Pneumatic and Sports Injury Recovery with ESWT Shockwave Therapy"19. "Precision
- Kneading Magnetic Knee Massager With Heat Vibration Laser Digital touch Panel Imported, 5000 V
- Radial shockwave therapy for chronic pain management
- Radial shockwave therapy for hamstring injuries
- Pedo Cycle High-quality motorized pedal exerciser for recovery
- Motorized electrical pedal cycle for improving muscle tone
- Lymphedema Dvt Machine Lymphatic Drainage Pump
- Electric therapy machine Motorized rehab cycle Electric mini exercise bike Motorized home
- Plastics ABS Portable DVT Prevention Device 4 Chamber Digital Pneumatic Compression Therapy Device
- Electric pedal bike for home rehabilitation
- Electric rehab exerciser Motorized workout cycle Electric pedal therapy bike Motorized mini pedo cycle Electric fitness machine
- Electrical Motorized Rehabilitation Device Pedo Cycle Digital pedo cycle
- Mini Exercise Bike High-tech physiotherapy device Motorized rehab pedal cycle
- Automatic Physiotherapy Device Electrical Pedal Cycle For Rehabilitation
- Digital Remote-Controlled Leg Pedal Automatic Electrical Digital with Leg Support Pair
- Pedal exerciser Best Electric Stroke Recovery Pedal Machine for Digital Electrical Motorized Home Rehabilitation
- Laser Therapy Equipment Class 3B Low LEVEL Cold LASER Physiotherapy Dual Probe Digital
- Motorized Physiotherapy Cycle Digital Electrical Pedo Cycle
- Plastics ABS Exercise Equipment Automatic Motorized Legs And Arm Rehabilitation Electrical Pedal Cycle, Manual
- Motorized pedal bike for physical therapy fitness exerciser
- Electrical Pedal Exerciser for Legs with Motorized Leg Exercise Machine
- Plastics ABS Exercise Equipment Motorized Stroke Recovery Pedal Bike, Manual
- Innovative Pedo Cycle Motorized Electrical Pedal Exerciser for Home Use
- Motorized pedal exercise bike Electrical Pedal Cycle for Rehab Pedo Cycle Electrical
- Lymphedema Machine for Varicose Vein
- Physiotherapy And Rehabilitation Equipment Pedo Cycle Motorized Electrical Digital
- T43 ESWT Device Adjustable 16 mJ Intensity for Tendon & Muscle Therapy
- Motorized Leg Exerciser with Remote Control Automatic Leg Rehabilitation Pedal with Digital
- Premium Quality Pedo Cycle Pedal Exerciser Motorized Eelectrical with Automatic Settings
- Remote-Controlled Pedal Exerciser for Legs Motorized Leg Rehabilitation Device
- Legs And Arm Physiotherapy Device Pedal Cycle Physiotherapy Motorized Electrical, Manual
- Plastics ABS Exercise Equipment Pedo Cycle Pedal Exerciser for Stroke Recovery Therapy, Manual
- Pedal device for automatic rehabilitation Leg Therapy Electrical Pedal Exerciser with Leg Support
- Rehab equipment Motorized Leg Exercise Machine: Stroke Recovery Digital Rehab Exerciser
- Automatic Rehabilitation System Electrical Pedal Cycle for Rehab Pedo Cycle Electrical
- Electric pedal Motorized Electric Mini Pedal Exercise Bike
- Efficient Motorized Leg Rehabilitation Device Pedo Cycle with Remote Control
- Mini Pedal Exercise Cycle Motorized With Leg Support
- Motorized Leg Rehabilitation Device for Improved Mobility With Leg Protection Support
- Shock Wave Therapy Machine for Chronic Pain, For Hospital
- Hospital Grade Pelvic Muscle Training Chair for Physiotherapy Clinics
- Pulse Electromagnetic Therapy Machine-Physiotherapy & Muscle Recovery Equipment
- Clinical Pelvic Floor Muscle Training Chair for Advanced Rehabilitation Care
- 910nm Laser Pain Relief Machine Laser Pain Relief Treatment Tool
- Dual Probe Laser Pain Relief for Physiotherapists Professional Pain Relief 750mw 910nm
- LASER pain relief device Portable physiotherapy LASER
- LCD display physiotherapy laser device Dual probe laser pain relief device
- Dual Probe Infrared Laser Therapy Laser Pain Therapy Equipment 750mw
- dual probe laser pain relief professional pain relief laser
- Compact LASER therapy wand Handy LASER for pain relief device
- Dynamometer for Grip Strength Digital
- Professional Laser Pain Therapy Dual Probe Laser Pain Management
- Dual Probe Laser Pain Relief Physiotherapists Pain Relief Laser
- Ultrasonic Cavitation Slimming Equipment LIPO LASER RF Radio Frequency Skin Vacuum Body Shaper LIPO
- Biotronix RF Lipo LASER Cavitation Slimming Machine 6 in One, For Clinical Purpose
- Physiotherapy bubble Inclinometer Wide Use Therapy Inclinometer Anti-Leak Swivel Dial Precise Office
- Dual Probe Laser Therapy Equipment for Pain Management Cluster Probe and Pointed Probe
- Biotronix Solution Forever Dual Probe Laser Red Visible Pointed Probe & Cluster Probe 750 mw 910 nm
- dual probe laser for pain relief laser pain relief device 910nm
- Compact LASER physio device Handy pain relief LASER
- Professional Laser Therapy Device Physiotherapist Laser Pain Relief
- Red Visible Laser Therapy Dual Probe Pointed & Cluster 750 mw 910 nm Physiotherapy Laser
- Professional Laser Pain Relief 750mw 910nm Laser Pain Relief Device
- Laser Pain Management 750mw Dual Probe Laser Treatment Device
- Digital Hand Dynamometer Grip Strength Tester 265lbs/120kgs for Sport, Home & Clinical Use
- Laser Therapy for Pain Relief 750mw Dual Probe Laser Therapy
- Portable LASER therapy device Pain relief LASER machine
- Digital Hand Grip Dynamometer 120Kg EH-108
- Pre-programmed digital laser therapy LCD laser device for pain relief
- Home Use Portable Body Pain Reduce Low level Cold Laser Therapy Handy Physiotherapy
- Cold Laser Red Light Infrared Physiotherapy Device Therapy Pet Pain Relief LLLT
- Red Light Device for Body Infrared Light Hand Held Device Portable Handy Laser Physiotherapy
- LASER Therapy Device, 650nm&808nm Light Therapy, For Body Arthritis Physiotherapy,
- DVT Machine Digital 6 Chamber Lymphedema Pump
- Laser Physiotherapy 650nm 808nm Handheld Handy Portable Arthritis Knee Cervical Pain Relief
- LASER Therapy Device Wand for Body Pain Therapy Device for Joint and Muscle & Tissue Pain Relief
- Exercise Equipment Pedo Cycle Motorized Physiotherapy Equipments, Manual
- DVT Four-chamber medical compression equipment Pneumatic therapy
- Pedo Cycle Best motorized pedal exerciser for physiotherapy
- Clinical Pelvic Floor Therapy Chair for Post-Natal Rehabilitation
- Professional Pelvic Muscle Training & Stimulation Chair for Hospitals
- Phymaster Shockwave Advanced Physiotherapy Pain Therapy System
- Intermittent Compression Cold Therapy Machine for Pain Relief
- Cryotherapy Ice Compression Wrap for Knee, Shoulder, and Joint Pain Relief
- Cryopush LCD Controlled Cold Compression System for Rehab
- Physical Inclinometer, Accurate Rotary Dial Scratch Proof Bubble Inclinometer
- Ankle Joint CPM Machine for Hospital and Home Use Pain Relief and Mobility Recovery Device
- UniGym Motorized Digital Exercise Therapy Device
- DVT 6 Chamber Digital Air Compression Therapy Device With Touch Panel
- Multifunction pain therapy Dual modality therapy unit
- Veterinary-Grade Class IV Laser for Muscle & Joint Pain Relief in Animals
- Biotronix Combination therapy TENS and Ultrasound Therapy Digital Auto Mode 4 ch TENS and 1 mhz UST
- 2 In 1 Combination Physiotherapy Machines Ultrasound Therapy + TENS 4 ch Digital Auto Mode
- 980nm Class 4 Laser Therapy Device 10W Power for Rehabilitation and Pain Management
- Back Leg Chest Dynamometer for Orthopedic, Neuro & Sports Rehab
- Physiotherapy Shockwave Therapy Machine
- High Power 850W PEMT Loop Therapy Machine with Large & Small Ring Handles
- Compact Pain Relief Handheld Laser Therapy Device
- Remote Controlled Elbow CPM Machine Precision Therapy for Joint Stiffness
- Back Leg Chest Muscle Testing Dynamometer for Rehab & Fitness Centers
- Physical Therapy Pneumatic Shock Wave Therapy Equipment
- Physiotherapy Treatment Shockwave Therapy Machine
- Shockwave Therapy Machine, Extracorporeal Physiotherapy Instrument with 6 Massage Head
- Advanced Class 4 Laser Therapy Machine 980nm For Physiotherapy And Rehab Centers
- Exoskeleton-Based Rehabilitation for Neurological Strength Training
- Pneumatic Radial Shockwave Therapy Device-7 Heads, 10 Modes
- All-in-One Back Leg Chest Dynamometer for Physiotherapy Assessment
- Precision Back Leg Chest Dynamometer for Physiotherapy & Sports Clinics
- Advanced Pelvic Floor Muscle Therapy Chair for Clinical Rehabilitation
- Advanced Pelvic Floor Therapy System Chair for Clinical Rehabilitation
- Kneading Magnetic Intelligent Knee Massager Electric Knee Physiotherapy Jain Pain Relief, 500 V
- Clinical Grade Pelvic Floor Muscle Training Chair for Rehabilitation Therapy
- Pneumatic shockwave therapy machine Pain relief shockwave therapy, For Hospital
- KM521 Pelvic Muscle Therapy System with Dual Channel Output
- KM521 Advanced Pelvic Floor Muscle Training System for Rehabilitation
- KM521 Clinical Pelvic Floor Muscle Rehabilitation Device
- KM521 Pelvic Floor Exercise & Muscle Stimulation Therapy Machine
- KM521 Pelvic Muscle Therapy Device
- KM521 Advanced Pelvic Floor Muscle Training Device for Physiotherapy
- KM521 Clinical Pelvic Muscle Rehabilitation Stimulator for Daily Therapy
- KM521 Smart Pelvic Floor Muscle Trainer for Strength & Control
- Advanced Class 4 Laser Therapy Equipment for Musculoskeletal Rehab
- Clinical Grade High Power Class 4 Laser Therapy Equipment
- KM521 Pelvic Muscle Strength Improvement Device for Daily Therapy
- KM521 Physiotherapy Pelvic Floor Muscle Stimulator with Dual Output
- Professional Class 4 Laser Therapy System for Sports Rehabilitation
- KM521 Rechargeable Pelvic Muscle Training Device with Animation Display
- High Intensity Class 4 Laser Therapy Device for Physiotherapy Experts
- Professional Class 4 Laser Therapy Device for Advanced Treatment Protocols
- Advanced Clinical Class 4 Laser Therapy System for Physiotherapy Experts
- Professional High Power Class 4 Laser Therapy Machine for Treatment Rooms
- High Intensity Class 4 Laser Therapy Machine for Clinical Use
- Advanced Medical Class 4 Laser Therapy Equipment for Rehabilitation Practice
- High Power Class 4 Laser Therapy Machine for Advanced Clinical Physiotherapy
- High Power Clinical Class 4 Laser Therapy System for Pain Relief Clinics
- Clinical Pneumatic Radial Shockwave Therapy Machine-7 Heads, Multi-Mode ESWT
- 10W High Power Class 4 Diode Laser Machine For Pain Relief Therapy
- Multi-Frequency PEMT Loop Therapy Equipment
- Motorized Leg Exercise Machine Electrical Pedal Exerciser for Legs
- Professional PEMT Loop Therapy Machine with Adjustable Magnetic Intensity 10006000 Gauss
- Exoskeleton-Assisted Therapy for Improved Motor Coordination
- High-Performance 7 Head Pneumatic Clinical Radial ESWT Device
- Multi-Pulse PEMT Loop Device for Electromagnetic Therapy & Recovery
- Advanced Pneumatic Clinical Radial ESWT System 7 Heads 10 Modes
- Air Compression Pneumatic Shockwave ESWT Equipment
- Dual-Diode Laser Therapy Device for Muscle Recovery & Deep Tissue Therapy
- Electromagnetic PEMT Loop Therapy Equipment-Multi-Frequency Pulse & Oscillation
- Professional 7 Head Pneumatic Shockwave Clinical Radial ESWT Machine
- Air Compression Pneumatic Clinical Radial ESWT Device 7 Heads 10 Modes
- Physio LASER Device Handheld Portable Pocket Compact Model
- Air Compression Therapy Pump Digital 8 Chamber Pneumatic Device
- Metal Physio Magneto Extracorporeal Machine NIRS Therapy PMST
- Physiotherapy Cold Laser Relieve Pain Magnetotherapy Machine Non-invasive Magneto Therapy Machine
- Medical Diode Laser 980nm/1470nm for Laser Piles Treatment, Fistula, Fissure & Vascular Use
- Diode Laser 980nm + 1470nm (Laser Proctology / Vascular Laser / Laser for Piles)
- 7-Head Pneumatic Radial ESWT- 10 Modes, 1-22 Hz, 0.5-1.2 Bar
- Compact Radial ESWT-Pneumatic, 7 Heads, Multi-Mode
- 7-Head ESWT Machine-Pneumatic, Adjustable Settings
- Clinical Radial ESWT Machine 7 Heads, Adjustable Frequency & Energy
- Magnetic Therapymat Physio Emtt Magneto Laser Infrared Physiotherapy Therapy Machine
- 7-Head Pneumatic ESWT-Adjustable Frequency & Energy
- Air Compression Radial Shockwave Therapy 7 Heads, 10 Modes
- Electric Continuous Passive Motion Elbow Device for Joint Recovery
- Passive Motion PEST Loop Device for Stroke Patient Rehabilitation and Leg Therapy
- Physiotherapy Laser Device for Pain Relief Handy Handheld
- Compact Shockwave Therapy Machine for Clinics & Rehabilitation Centers
- Smart Tecar Therapy Device with Touch Screen for Muscle Recovery & Joint Pain
- Portable EMT-2 Magnetic Pulse Therapy Unit Bone Healing & Tissue Regeneration
- Air Compression Radial Shockwave Therapy Equipment-ESWT for Physiotherapy & Rehab
- Air Compression Radial Shockwave Therapy-7 Heads, 10 Modes
- PEST Loop Physical Therapy Equipment for Range of Motion and Muscle Recovery
- Pneumatic Radial Shockwave Therapy Device-10 Modes, 1-22Hz Frequency, Clinical ESWT
- 980nm 1470nm Diode Laser Proctology Equipment for Hemorrhoid Fistula Laser Surgery
- Compact Elbow CPM Rehab Machine for Hospitals and Clinics
- Adjustable Passive Exercise and Stretch Therapy Loop Machine for Elderly and Post-Surgery Care
- Tecar therapy unit
- PEST Loop Machine Passive Exercise Therapy Equipment for Rehabilitation and Physiotherapy
- Electromagnetic Radial Shockwave Therapy Machine Touch Screen, 7 Heads
- Electric Passive Exercise PEST Loop Machine for Lower Limb Recovery and Physical Therapy
- Advanced Laser Proctology Machine 980nm & 1470nm Diode Laser for Fissure & Hemorrhoids
- Electromagnetic Compact Radial Shockwave Therapy Machine 7 Heads, 10 Modes
- Portable Radial ESWT-Pneumatic, 7 Heads, Clinical Use
- Pain Relief Treatment Physio Magneto Machine
- 7-Head Air Compression ESWT -10 Treatment Modes, 1-22 Hz
- 750N Electric Spine Chiropractor Electric Massager
- PMST Physio Magneto 808nm Laser Physiotherpay Pemf Machine Pain Relief
- Advanced Uni Gym Digital Therapy Equipment for Recovery
- Physio Magneto Replaceable Handle Magnetic Therapy Machine Pain Relief
- Magnetic therapy unit MBS-EM002-1
- Tecar Therapy Machine For Body Pain, CET RET For Sports Rehabilitator Sports Therapist, 4 Modes And 10 Body Parts To Choose From
- Extracorporeal Shockwave Therapy (ESWT) Machine-Pneumatic, Multi-Mode, Clinical Use
- 7-Head Radial Shockwave Therapy Device Adjustable Settings for Pain Relief & Rehabilitation
- Portable Tecar Therapy Machine for Physiotherapy Pain Relief and Rehabilitation
- Digital Sequential Compression Pump with 4 Chamber Digital Lymphedema Pump for Lymphatic Drainage Lymphedema Pump for Lypmhatic Drainage
- Iron Exercise Equipment Physio Magneto Plus Nirs Therapy Pmst Neo for Back Pain Relief
- Professional Magnetic Therapy Device for Pain Relief and Rehabilitation
- Dvt Device Scd Lymphedema Pump Sequential Intermettint Pneumatic Air Compression Therapy
- Magnetotherapy Machine EMTT Physio Magneto Therapy
- Magnetotherapy Machine EMTT Physio Magneto Therapy for Pain Relief
- Smart Heated Knee Massager with Kneading Function Infrared Light for Joint Pain and Physiotherapy
- Pneumatic Air Compression Therapy For Lypmhatic Drainage 4 Chamber 3 Modes With 2 Legs Attachment
- Ankle CPM Machine for Physiotherapy
- Class 4 Laser Therapy Device for Fast Pain Relief and Muscular Recovery
- Iron Exercise Equipment Physiotherapy Extracorporeal Magnetic Therapy
- Rechargeable Physiotherapy Knee Massager Wrap for Arthritis Joint Pain Infrared Heat Therapy Device
- Ankle CPM Machine Portable Rehabilitation Device
- High Frequency Portable Extracorporeal Magneto Therapy Device for Pain Relief
- 750N Electric Spine Chiropractor Electric Impulse Gun Actuator Massager Chiropractic Adjusting Tool Instrument Professional Massage
- DVT 4 Chamber Lymphatic Drainage Device with Digital Control for Leg
- Physio Magneto Plus Advanced Therapy NIRS Transduction Physio Machine
- Physio Magneto Therapy
- Portable Cryo Cold Compression Therapy Device for Pain Relief and Injury Recovery
- Digital 4T Electrotherapy Machine with Touchscreen-OEM/ODM Available
- Advanced Tecar Therapy Machine for Chronic Pain and Sports Injuries
- 4T Physiotherapy Machine Airotor Control Box for Dental Clinic and Training Dental Elevator
- Focused Shockwave Therapy Equipment for Physiotherapy and Muscle Pain Relief
- Robotic Exoskeleton Physiotherapy Rehabilitation Device for Neuro and Stroke Recovery
- Next-Gen Exoskeleton Rehabilitation Robot for Clinical Neuro Therapy
- Robotic Digital Rehabilitation Exoskeleton for Improved Mobility Assistance
- Phymaster Shockwave Physiotherapy Machine for Rehab & Recovery
- Phymaster Shockwave Clinical Pain Management Therapy Equipment
- Phymaster Shockwave Physiotherapy Machine for Clinical Pain Treatment
- Phymaster Shockwave Advanced Pain Relief Machine for Physiotherapy
- Phymaster Shockwave Orthopedic Physiotherapy Therapy Equipment
- 6 Chamber DVT Therapy Machine Digital Display Type for Rehabilitation Treatment
- Exoskeleton-Based Therapy for Improved Motor Coordination
- Exoskeleton-Assisted Physiotherapy for Neurological Recovery
- Phymaster Shockwave Pain Relief Machine for Physiotherapy Clinics
- Phymaster Shockwave Physiotherapy Device for Sports Injury Recovery
- Phymaster Shockwave Pain Relief Equipment for Orthopedic Clinics
- Phymaster Shockwave Advanced Physiotherapy Treatment Equipment
- Medical Exoskeleton Solutions Supporting Safe Rehabilitation
- Phymaster Shockwave Therapy Machine for Orthopedic Rehabilitation
- Phymaster Shockwave Physiotherapy System for Hospital & Clinic Use
- Phymaster Shockwave Therapy Equipment for Chronic Pain Management
- Robotic Exoskeleton Systems for Assisted Physiotherapy Training
- Phymaster Shockwave Therapy Equipment for Chronic Muscle Pain
- Phymaster Shockwave Shockwave Therapy Machine for Physiotherapy
- Phymaster Shockwave Therapy Machine for Physiotherapy & Rehabilitation
- Phymaster Shockwave Therapy Machine for Rehabilitation Centers
- Phymaster Shockwave Physiotherapy Equipment for Muscle Pain Relief
- Phymaster Shockwave Therapy Device for Muscle Stiffness & Joint Pain
- Phymaster Shockwave Therapy Machine for Joint Pain & Tendon Healing
- Exoskeleton Technology Supporting Safe Movement Relearning
- Clinical Exoskeleton Devices for Targeted Motor Training
- Focused Shockwave Therapy Equipment for Sports Injury and Muscle Recovery
- DVT 6 Chamber Digital Sequential Therapy Machine for Swelling and Post-Surgery Care
- Phymaster Shockwave Physiotherapy Machine for Sports Injury Treatment
- Exoskeleton Rehabilitation Systems for Clinical Excellence
- Phymaster Shockwave Non-Invasive Therapy Machine for Pain Relief
- Phymaster Shockwave Non-Surgical Therapy Machine for Pain Relief
- Advanced Wearable Exoskeleton Devices for Rehabilitation Therapy
- Phymaster Shockwave Physiotherapy Equipment for Fast Pain Recovery
- Exoskeleton Therapy Solutions for Functional Motor Recovery
- Phymaster Shockwave Physiotherapy Therapy Machine for Pain Relief
- Phymaster Shockwave Therapy System for Musculoskeletal Pain Relief
- Phymaster Shockwave Advanced Physiotherapy Equipment for Clinics
- Phymaster Shockwave Therapy Machine for Muscle, Joint & Tendon Pain
- Phymaster Shockwave Physiotherapy Machine for Joint & Muscle Pain
- Phymaster Shockwave Shockwave Therapy Device for Tendon Pain Treatment
- Phymaster Shockwave Therapy Equipment for Musculoskeletal Pain
- Phymaster Shockwave Physiotherapy Device for Orthopedic Pain Relief
- Phymaster Shockwave Therapy Machine for Sports Physiotherapy Clinics
- Phymaster Shockwave Therapy System for Physiotherapy & Rehab Centers
- Phymaster Shockwave Physiotherapy System for Pain & Mobility Improvement
- Phymaster Shockwave Advanced Shockwave Pain Relief Machine
- Phymaster Shockwave Therapy Device for Muscle Stiffness Treatment
- Phymaster Shockwave Physiotherapy Therapy Equipment for Pain Relief
- Phymaster Shockwave Physiotherapy Equipment for Deep Pain Treatment
- Phymaster Shockwave Physiotherapy Equipment for Hospital Use
- Phymaster Shockwave Shockwave Therapy Equipment for Hospitals & Clinics
- Phymaster Shockwave Clinical Shockwave Therapy System
- Phymaster Shockwave Therapy Equipment for Muscle & Tendon Recovery
- Phymaster Shockwave Shockwave Therapy Machine for Hospitals
- Phymaster Shockwave Shockwave Therapy Machine for Professional Use
- Phymaster Shockwave Physiotherapy Equipment for Muscle Recovery Treatment
- Orthopedic Ankle CPM Machine For Injury & Fracture Recovery
- Ankle Continuous Passive Motion Device For Plantar & Dorsiflexion Therapy
- OrthoMax Focus Shockwave Therapy System for Complete Pain Management
- CorePrecision Focus Shockwave System for Advanced Physiotherapy Clinics
- Digital Cold Compression Device Designed for Effective Rehabilitation
- Exoskeleton Technology Enhancing Coordination and Balance
- Exoskeleton Therapy Solutions for Long-Term Functional Recovery
- Exoskeleton-Based Rehabilitation for Neurological Recovery
- Medical Exoskeleton Systems Supporting Functional Movement
- Advanced Medical Exoskeletons for Professional Rehabilitation
- Robotic Exoskeleton Rehabilitation for Progressive Recovery
- Medical Wearable Exoskeletons for Advanced Therapy Sessions
- Smart Robotic Exoskeletons for Modern Rehab Clinics
- Robotic Wearable Exoskeletons for Consistent Rehabilitation
- Wearable Exoskeleton Devices for Upper and Lower Limb Rehab
- Exoskeleton-Assisted Rehabilitation for Functional Independence
- Exoskeleton-Based Rehabilitation for Strength and Mobility Training
- Exoskeleton-Based Therapy for Functional Movement Improvement
- Smart Medical Exoskeletons for Motor Function Training
- Exoskeleton Therapy Solutions for Modern Physiotherapy Practice
- Exoskeleton Therapy Systems Designed for Clinical Excellence
- Smart Exoskeleton Therapy Devices for Clinical Rehabilitation
- Wearable Medical Exoskeletons for Long-Term Therapy Programs
- Exoskeleton Solutions for Neurological and Orthopedic Recovery
- Advanced Wearable Exoskeleton Systems for Clinical Rehabilitation
- Wearable Medical Exoskeletons for Assisted Movement Therapy
- Smart Exoskeleton Solutions for Clinical Therapy Applications
- Robotic Exoskeleton Systems for Precision Movement Training
- Advanced Wearable Exoskeletons for Rehabilitation Therapy
- Medical Exoskeleton Therapy for Neuro and Orthopedic Rehabilitation
- Medical-Grade Wearable Exoskeletons for Physiotherapy Clinics
- Wearable Medical Exoskeletons for Structured Physiotherapy
- Exoskeleton Rehabilitation Systems for Strength and Control
- Robotic Exoskeleton Devices Supporting Motor Function Recovery
- Exoskeleton-Based Therapy for Neuromuscular Rehabilitation
- Exoskeleton Innovation Redefining Recovery and Mobility
- Exoskeleton Technology Supporting Progressive Functional Recovery
- Advanced Medical Exoskeletons for Physiotherapy and Rehabilitation
- High-Precision Exoskeleton Therapy for Neuro and Ortho Recovery
- Advanced Wearable Robotics for Physiotherapy and Neuro Rehab
- Smart Wearable Exoskeletons for Clinical Movement Training
- Smart Exoskeleton Rehabilitation for Hands Arms and Lower Limbs
- Precision-Guided Exoskeleton Therapy for Functional Independence
- Wearable Exoskeleton Devices for Assisted Functional Recovery
- Exoskeleton-Based Rehabilitation for Improved Movement Control
- Exoskeleton-Based Physiotherapy for Motor Skill Restoration
- Clinical Exoskeleton Solutions for Modern Rehabilitation Centers
- Medical Exoskeleton Technology for Controlled Functional Training
- Smart Medical Exoskeletons for Precision Movement Therapy
- Exoskeleton Solutions for Post-Stroke and Paralysis Recovery
- Next-Generation Exoskeleton Devices for Clinical Rehabilitation
- Exoskeleton-Based Movement Therapy for Clinical Excellence
- Clinical Applications of Exoskeletons in Modern Rehab Centers
- Exoskeleton Rehabilitation Technology for Motor Skill Development
- Smart Pelvic Floor Muscle Therapy Chair for Comprehensive Rehabilitation
- Smart Pelvic Floor Muscle Conditioning Chair for Long-Term Therapy
- Professional Pelvic Muscle Therapy Chair for Advanced Clinical Care
- Hospital Approved Pelvic Muscle Therapy Chair for Physiotherapy Clinics
- Medical Grade Pelvic Muscle Conditioning Chair for Physiotherapy Clinics
- Hand and Finger Exoskeletons for Fine Motor Skill Training
- Digital Clinical Pelvic Muscle Rehabilitation Chair with Preset Therapy
- Professional Pelvic Muscle Activation Chair for Rehabilitation Centers
- Hospital Approved Pelvic Muscle Rehabilitation Chair for Medical Practice
- Digital Pelvic Floor Muscle Activation Chair for Pelvic Care Programs
- Medical Pelvic Muscle Therapy Chair for Post-Operative Care
- Professional Pelvic Muscle Strength Training Chair for Clinical Settings
- Advanced Pelvic Floor Muscle Conditioning Chair for Physiotherapy Practice
- Smart Pelvic Floor Muscle Rehabilitation Chair for Medical Use
- Non-Invasive Pelvic Floor Muscle Strengthening Chair for Clinical Use
- Professional Pelvic Muscle Therapy Chair for Rehabilitation Programs
- Advanced Pelvic Floor Muscle Rehabilitation Chair for Hospitals
- Pelvic Floor Muscle Strengthening Chair for Complete Pelvic Care
- Pelvic Muscle Strength & Recovery Chair for Physiotherapy Centers
- Professional Pelvic Muscle Activation Chair for Physiotherapy Practice
- Medical Grade Pelvic Muscle Therapy Chair for Physiotherapy & Recovery
- Professional Pelvic Muscle Training Chair for Comprehensive Rehabilitation
- Non-Surgical Pelvic Floor Muscle Training Chair for Clinical Care
- Non-Invasive Pelvic Floor Rehabilitation Chair for Muscle Recovery
- Non-Surgical Pelvic Floor Muscle Therapy Chair for Clinical Rehabilitation
- Smart Pelvic Floor Muscle Therapy Chair for Long-Term Rehabilitation
- Advanced Pelvic Floor Muscle Rehab Chair for Rehabilitation Hospitals
- Professional Pelvic Muscle Strengthening Chair for Clinical Use
- Pelvic Floor Muscle Strength & Stability Chair for Medical Rehabilitation
- Medical Pelvic Muscle Activation Chair for Post-Treatment Therapy
- Hospital Approved Pelvic Muscle Therapy Chair for Physiotherapy Practice
- Professional Pelvic Muscle Training Chair for Advanced Rehabilitation
- Professional Pelvic Muscle Rehab Chair for Long-Term Clinical Care
- Smart Pelvic Floor Muscle Rehabilitation Chair for Advanced Care
- Pelvic Muscle Conditioning & Recovery Chair for Long-Term Therapy
- Medical Grade Pelvic Muscle Rehab Chair for Hospital Use
- Digital Clinical Pelvic Muscle Therapy Chair with Intelligent Control
- Non-Invasive Pelvic Floor Rehabilitation Chair for Incontinence Care
- Pelvic Floor Muscle Rehabilitation Chair for Womens & Mens Clinics
- Digital Pelvic Floor Muscle Activation Chair for Pelvic Health Clinics
- Pelvic Muscle Strengthening & Recovery Chair for Pelvic Health Care
- Professional Pelvic Muscle Training Chair for Hospital Rehabilitation
- Advanced Pelvic Floor Muscle Training Chair for Physiotherapy Practice
- Deep Tissue PEMT Magnet Therapy Device
- Wireless Rechargeable Knee Massage Device with Heat Infrared and Vibration Therapy for Joint Pain
- Wearable Exoskeleton Technology for Assisted Physiotherapy
- Pneumatic shockwave therapy for pain-free movement Shockwave therapy for muscle stiffness relief
- Shock Wave Therapy Machine with 10 Bar for Joint and Muscle and Bone Tissue Regeneration, Non-Invasive, No Side Effects
- Portable shockwave therapy unit Shockwave therapy machine for home use, For Hospital
- High Power PEMT Loop Therapy Equipment-4500Hz Oscillation, Multi-Frequency 2-8Hz
- UniGym App-Based Rehab Exercise Machine with Sensor Feedback
- UniGym Advanced Rehabilitation Trainer for Clinics & Home
- Compact DVT Air Compression Pump for Post-Surgery Recovery
- Digital DVT Air Compression Therapy Machine with Remote Control
- KM521 Smart Pelvic Muscle Rehabilitation Device with Guided Programs
- Computerized Laser Therapy Equipment Dual Probe Pointed and Cluster 750mw 2 Year Warranty
- Handheld Physiotherapy Laser Therapy
- Professional Brain And Autonomic Nervous System Modulation Device
- Shockwave Therapy Machine Pneumatic Shock Wave Physiotherapy Instrument Deep Muscle Massager Relax
- Pneumatic Shockwave Therapy Machine (10 Bar)
- Compact Laser Therapy Device with Cooling Fan, Digital Display & Rechargeable Battery
- Focused Shockwave Therapy Equipment for Shoulder and Knee Pain Treatment
- Hospital Grade Elbow CPM Device Reliable Post-Surgery Recovery Tool
- CE Approved Passive Exercise PEST Loop Machine for Orthopedic Rehabilitation
- Cold compression Therapy
- Adjustable Angle Elbow CPM Machine Post-Surgery and Injury Recovery Solution
- Konmed KM-560 EMG Biofeedback Equipment with ETS Function
- Motorised Cryo Cold Therapy System for Fast Pain Relief
- Compact physiotherapy LASER Portable pain management LASER
- Wearable Medical Exoskeletons for Functional Mobility Support
- Physiotherapy Robotic Exoskeleton for Assisted Mobility Training
- Tecar Therapy Machine Feet Back Knee Shoulder Pain
- Motorized rehab exerciser for elderly Electric pedal machine for physiotherapy
- Pelvic Floor Muscle Strengthening Chair for Comprehensive Pelvic Care
- Imported Professional Elbow CPM Device Smooth Motion for Effective Rehabilitation
- Digital Ergometer Arm Exerciser Physiotherapy for Upper Limb, For Home, Fitness Equipment
- Sequential Air Compression Device for Deep Vein Thrombosis Management
- Portable Intermittent Pneumatic Compression Device for DVT Prevention
- pain relief ultrasound TENS muscle stimulation device
- T43 Touch Screen Shockwave Therapy Equipment Non-Invasive Treatment Solution
- Portable Elbow CPM Machine with Digital Control Panel
- Digital Laser Therapy Machine with Protective Goggles & Adjustable Stand
- Infrared Laser Therapy Device 910nm Laser Pain Relief Device 750mw
- Rechargeable Pain Relief Laser Device with 14 Red+4 Infrared Diodes
- UniGym Smart Rehab Trainer with Active, Passive, Resistive & Assistive Modes
- Air Compression Based Radial ESWT Device-10 Treatment Modes, Adjustable Intensity
- Robotic Exoskeleton Devices for Precision Physiotherapy Training
- Shockwave therapy for scar tissue breakdown and tissue regeneration
- UniGym Robotized Arm and Leg Rehab Device
- Konmed KM-560 4 Channel EMG Biofeedback Device with ETS Mode
- Compact Pneumatic Radial ESWT 10 Modes, 1-22 Hz
- Pain Relief Physiotherapy LASER Device Digital LCD Based Dual Probe 750mw 910nm
- Compact Elbow CPM Device Home & Clinic Use for Flexion and Extension Therapy
- Pedo Cycle Motorized Electrical Digital Physiotherapy Rehabilitation Device
- Dual probe laser pain relief device Physiotherapy laser pain relief
- Motorized DVT Compression Pump with Adjustable Pressure Settings
- PEMT Pulse Electromagnetic Therapy Device for Physiotherapy Clinics
- DVT therapy pump Digital DVT system 4 chamber compression pump
- Hospital Grade Pelvic Muscle Strengthening Chair for Clinical Use
- Smart Pelvic Floor Muscle Therapy Chair for Rehabilitation Programs
- Pelvic Muscle Recovery & Strength Training Chair for Medical Centers
- Digital Pelvic Floor Muscle Conditioning Chair for Pelvic Health Care
- Non-Invasive Pelvic Floor Muscle Therapy Chair for Patient Recovery
- Pelvic Floor Muscle Strength & Stability Chair for Rehabilitation Centers
- Electromagnetic Focus Shockwave Device for Precision Tissue Regeneration
- Digital Clinical Pelvic Muscle Training Chair with Advanced Therapy Modes
- Hospital Use Pelvic Muscle Rehabilitation Chair for Physiotherapy
- Pulse Electromagnetic Field Therapy Device with Loop Applicator
- Digital Stroke Recovery Pedal Bike Automatic Legs And Arm Physiotherapy Device
- Motor-assisted rehab bike System Electrical Pedal Cycle for Rehab Pedo Cycle Electrical
- Chiropractor Electrical Device 750N Spine Correction 50N -750 N Adjustable Force
- Motorized Digital Rehabilitation Pedal Bike Pedo Cycle
- Advanced PEMT Loop Therapy Equipment with 18cm Penetration Depth & Multiple Frequencies
- PlasticsABS Rectangular Smart Tecar Wave RF Therapy Machine for Physical Therapy, For Sports Injury Recovery
- Rehabilitation PEST Loop Machine with Digital Control for Knee and Hip Mobility
- Laser Pain Relief Sport Injury Physio Magneto Machine
- Technology Body Massage Equipment Pain Relief Physical Therapy Instrument
- KM521 Pelvic Floor Muscle Training Machine with Voice Guidance
- KM521 Advanced Pelvic Muscle Rehab Machine for Clinical Use
- Professional Pelvic Muscle Activation Therapy Chair for Rehabilitation
- Digital Clinical Pelvic Muscle Rehabilitation Chair with Preset Modes
- Pelvic Floor Muscle Strength & Control Chair for Physiotherapy Centers
- Advanced Clinical Pelvic Floor Therapy Chair for Muscle Strengthening
- Medical Grade Pelvic Muscle Rehabilitation Chair for Physiotherapy Clinics
- Rechargeable Pelvic Floor Muscle Stimulator KM521 for Clinical & Home Use
- Konmed KM-560 Physiotherapy Biofeedback Device
- EMT2 Advanced Magnetotherapy System Adjustable Field Strength for Arthritis
- Complete Neurological Stimulation And Recovery Machine
- Electromagnetic PEMT Therapy Equipment-Multi-Frequency Pulse & Deep Tissue Stimulation
- Non-Invasive Pelvic Floor Muscle Conditioning Chair for Patient Care
- Physiotherapy Laser Therapy Unit 750 Mw 910 nm Dual Probe LCD Baced Computerized LASER Device
- Bio VNS-1 Advanced Brainwave And Vagus Nerve Neuromodulation Therapy System
- Shockwave Head Therapy Machine Physical Therapy Pain Relief
- Non-Invasive Portable Extracorporeal Magnetic Therapy Device
- Electromagnetic PEMT Loop Therapy System for Pain Management & Healing
- Portable Cryo Cold Therapy Device for Home Use Joint and Muscle Recovery
- Digital Pelvic Muscle Strength Training Chair for Incontinence Care
- Konmed KM-560 Biofeedback Equipment with ETS Mode
- Konmed KM-560 EMG Biofeedback Device with FES Therapy
- Magneto Physiotherapy Pain Relief Equipment in Physiotherapy Physio Machine with Air Cooling System
- Phymaster Shockwave Physiotherapy Machine for Muscle Pain Recovery
- Phymaster Shockwave Advanced Physiotherapy System for Rehabilitation
- Phymaster Shockwave Physiotherapy Equipment for Muscle Pain Management
- Phymaster Shockwave Physiotherapy Device for Chronic Muscle Pain Treatment
- Konmed KM-560 EMG Biofeedback Device for Pelvic Floor Therapy
- Konmed KM-560 Advanced Physiotherapy Biofeedback Equipment
- Konmed KM-560 EMG Biofeedback Machine with Multi Therapy Modes
- Konmed KM-560 4 Channel EMG Biofeedback Unit for Rehabilitation
- Konmed KM-560 Biofeedback Machine with Electrical Therapy
- Konmed KM-560 Advanced Biofeedback Equipment with EMG
- Konmed KM-560 Professional Biofeedback Device for Clinics
- Konmed KM-560 Biofeedback Machine for Pelvic Muscle Rehab
- Konmed KM-560 Physiotherapy Biofeedback Device with EMG Analysis
- Konmed KM-560 EMG Biofeedback Equipment with ETS Rehab Mode
- Konmed KM-560 Biofeedback Machine with 4 Channel EMG Output
- Konmed KM-560 EMG Biofeedback Device for Sports Therapy
- Konmed KM-560 EMG Biofeedback Machine with Advanced Features
- Konmed KM-560 Clinical Biofeedback Device with Gaming Therapy
- Konmed KM-560 EMG Biofeedback Machine for Muscle Training
- Konmed KM-560 4 Channel Biofeedback Device with FES NMES
- Konmed KM-560 EMG Biofeedback System with Triggered Stimulation
- Konmed KM-560 Clinical EMG Biofeedback Device with ETS
- Konmed KM-560 4 Channel EMG Biofeedback System for Therapy
- Konmed KM-560 Professional Biofeedback Machine for Physiotherapy
- Konmed KM-560 Physiotherapy Biofeedback Device with NMES
- Konmed KM-560 Clinical EMG Biofeedback Device with Gaming
- Konmed KM-560 4 Channel EMG Biofeedback System for Rehab
- Konmed KM-560 Rehab Biofeedback Machine with Gaming Interface
- Advanced Brain Rehab Device tDCS tPCS tACS tVNS Machine
- 4 in 1 Electrotherapy Combo Device for Pain Relief & Muscle Rehab
- Smart tDCS tACS tVNS tPCS Brain Therapy System
- Electro Neuro Stimulation Therapy Machine tDCS tPCS tACS tVNS
- Smart Brain and Neuro Stimulation Equipment tDCS tACS tVNS tPCS
- Konmed KM-560 EMG Biofeedback Equipment with FES NMES Support
- Konmed KM-560 4 Channel EMG Biofeedback Device with Rehab Therapy
- Konmed KM-560 Professional Biofeedback Machine for Physiotherapys
- Smart Neuro Therapy Machine tDCS tACS tVNS tPCS
- Advanced Neuro Stimulator tDCS tPCS tACS tVNS for Clinics
- Konmed KM-560 EMG Biofeedback Stimulator with Clinical Modes
- Konmed KM-560 Rehab Biofeedback Equipment with ETS Mode
- Bubble Inclinometer for Physical Therapy ROM Assessment Reliable Range of Motion Measurement
- Phymaster Shockwave Physiotherapy Equipment for Rehab & Recovery
- Konmed KM-560 Physiotherapy Biofeedback Device with FES
- Konmed KM-560 Biofeedback Machine with EMG Stimulator
- Konmed KM-560 Clinical Biofeedback Device with EMG Output
- Phymaster Shockwave Advanced Shockwave Therapy Machine for Pain Relief
- Konmed KM-560 4 Channel EMG Biofeedback Device with ETS Therapy
- Konmed KM-560 Professional Biofeedback Machine for Therapy
- Phymaster Shockwave Professional Shockwave Therapy Equipment
- Konmed KM-560 Biofeedback Machine with EMG NMES FES Therapy
- IASTM Tools Instrument-Assisted Soft Tissue Mobilization Soft Tissue Mobilization Tools
- Physiotherapy Combo Machines Combination US and TENS UST 1 Mhz TENS 4 ch Auto Mode Premium
- Metal Silver Professional IASTM Tools for Effective Soft Tissue Mobilization"
- Traditional Guasha Tools for Holistic Therapeutic Massage IASTM Physiotherapy Tools
- 5 pc Set IASTM 304 Stainless Steel Gua Sha Tools For GuaSha Physiotherapy Massage Tools
- Therapeutic Massage IASTM Physiotherapy Tools
- IASTM 5 pc Instruments for Rehabilitation, For Therapeutic Recovery Tools
- Physiotherapy Massaging Tools For Fascia Tools Set For Soft Tissue Iastm Kit
- Scraping Guasha IASTM Physiotherapy Massaging Tools For Fascia Tools Set For Soft Tissue Iastm Kit
- Konmed KM-560 4 Channel Biofeedback System with Gaming Therapy
- pain relief device muscle recovery tool dual therapy unit physiotherapy equipment
- electrotherapy device physiotherapy UST 1 MHz TENS 4 channel combo therapy machine
- TENS auto mode ultrasound therapy combo electrotherapy
- Electrotherapy Digital Combi 2 in one TENS 4 ch Auto Mode and Ultrasound therapy 1 Mhz
- Combination Physiotherapy Equipment Combi UST 1 Mhz and TENS 4 ch, 4- Channel, Analogue ( Manual )
- Combination Combi US + TENS 4 ch Physiotherapy Electrotherapy Device, 4- Channel, Analogue ( Manual )
- Physiotherapy laser with dual probes Advanced laser therapy equipment
- Combination Therapy Unit TENS 4 channel with Ultrasound Therapy 1 Mhz Digital Physiotherapy
- Combi 2 in one UST Ultrasound Therapy 1 Mhz and TENS 4 channel Digital Auto mode Electrotherapy
- Combination therapy device 2 in 1 ultrasound TENS machine
- Professional Brain and Neuro Stimulation Device tDCS tACS tVNS tPCS
- Portable LASER for pain relief wand Handheld LASER physiotherapy wand
- KM-580 EMG Triggered Stimulation (ETS) & Biofeedback Physiotherapy Equipment
- Medical Exoskeleton Devices Enhancing Therapy Precision
- Exoskeleton Rehabilitation Systems for Controlled Mobility Training
- Exoskeleton Rehabilitation Technology for Safe Movement Practice
- Clinical Exoskeleton Devices for Targeted Therapy Sessions
- Physiotherapy knee massager Pain relief knee massager
- Pneumatic shockwave therapy for tendonitis Radial shockwave therapy for hamstring tendinopathy
- shockwave Knee pain relief Lower back pain therapy Sciatica pain treatment
- Non-invasive pain treatment Joint pain therapy
- Shockwave therapy for postural correction Pneumatic shockwave therapy for fascia adhesions
- Shockwave therapy for post-surgical scarring Pneumatic shockwave therapy for chronic pain conditions
- bubble Professional physiotherapy inclinometer
- Shockwave therapy for muscle atrophy Radial shockwave therapy for tennis elbow
- Pneumatic Shockwave Therapy Physiotherapy Radial shock Wave Therapy
- Physiotherapy bubble inclinometer Therapy
- Inclinometer for body mechanics with swivel dial
- Plastic 0-360 Degree Bubble Inclinometer Physiotherapy, Size: Portable, Packaging Type: Box
- Bubble Inclinometer Glass Tube Inclinometer Therapy Inclinometer
- Advanced Rom Assessment Tool Bubble Inclinometer Accurate Joint Flexibility Testing Physiotherapy
- Clinical use bubble inclinometer Portable therapy
- Inclinometer for physical therapy and rehabilitation
- Physiotherapy Inclinometer Wide Use Therapy Inclinometer Anti-Leak Swivel Dial Precise Office
- Cavitation Slimming Machine 6 in one Digital Touch Screen With RF Vacuum LASER Weigh Loss Slimming
- Ultrasonic Cavitation Machine 6 in one with RF Vacuum and LIPO LASER Touch Screen Digital
- conductive self-adhesive electrode pads for physiotherapy
- premium self-adhesive electrode pads for muscle stimulation
- pain relief pads muscle stim pads electrotherapy pads stimulator pads
- reusable TENS pads reusable EMS pads durable TENS pads durable EMS pads
- adhesive pads TENS adhesive pads EMS TENS unit pads EMS device pads
- durable self-adhesive electrode pads for pulse massagers
- adhesive electro pads pain relief therapy pads stick-on TENS pads
- replacement electrode pads stick-on electrode pads pain relief pads
- IFT electrode pads pulse massager pads reusable electrode pads
- self adhesive electrode pads TENS electrode pads EMS electrode pads
- reusable self-adhesive electrode pads for IFT therapy
- long-lasting self-adhesive electrode pads for TENS and EMS units
- medical-grade self-adhesive electrode pads for pain relief
- universal self-adhesive electrode pads for electrotherapy
- high-quality self-adhesive electrode pads for EMS devices
- self stick electrode pads adhesive pads for TENS
- muscle stimulation pads physiotherapy electrode pads
- TENS unit electrode pads EMS electrode pads
- Self Adhesive Electrode Pads for EMS, TENS, IFT (Pin Size 1.5mm) SELF ADHESIVE Electrotherapy
- Self Adhesive ELECTRODE Square pads (Pack of 4) TENS electrode pad Electrotherapy Device
- Electrodes Reusable Sticking Pads For Tens, Ift, Stimulators & Massager Machines One Pair 4 Pieces
- Self Adhesive Electrode Pad for Electronic Stimulators, Ift, Ems and Pulse Massagers
- Sticking TENS Pads Self-Adhesive, for IFT, TENS, Massager Machines Four Pieces Pig-Tail Design
- Self Adhesive Electrode Pads For Electronic Stimulators, Ift, Tens,Ems & Pulse Massagers
- dynamometer Digital hand grip analyzer meter
- Hand grip strength dynamometer digital analyzer
- Digital hand grip strength measurement device
- Hand grip dynamometer digital strength tester
- Digital hand grip gauge Digital grip strength meter
- Hand grip strength dynamometer digital meter
- Digital grip force meter Hand grip strength digital
- Electronic hand grip dynamometer Digital hand grip meter
- Digital grip strength tester Digital hand grip strength tester
- dynamometer Hand strength tester digital Digital grip force dynamometer
- dynamometer Hand grip dynamometer digital tester
- Digital hand grip dynamometer Hand grip dynamometer
- dynamometer Digital handgrip force measurement device
- Digital hand grip strength tester Electronic handgrip dynamometer
- Digital handgrip dynamometer Hand grip dynamometer digital
- Biotronix HandGrip Dynamometer Digital 268Lbs/120Kgs USB Charging Strength Measurement
- Dynamometer for Hand Muscle Strength Digital Hand Strength Gauge
- EASYCARE (EC3060) Digital Hand Dynamometer for Grip Strength Measurement (90Kgs / 198Lbs)
- Digital Handheld Dynamometer Grip Strength Measuring Automatically Captures Grip 268 Lbs/120Kg
- View Similar Digital Adjustable Grip Strength Tester Electronic Hand Dynamometer
- Hand strength digital device Portable hand grip dynamometer
- Digital Hand Grip Strength Dynamometer EH-108 Model Battery and USB Operated
- Grip Strength Tester Digital Hand Dynamometer 265 Lbs/120 Kg LCD Hand Grip Strengthener
- Hand Dynamometer Grip Strength Trainer Electronic Hand Grip Strength Tester
- Digital CAMRY Auto Capturing Digital Hand Grip Dynamometer Strength Measurement Meter
- Digital Hand Grip Dynamometer
- Hand Strength Measurement Tool Dynamometer for Grip Assessment
- Biotronix Digital Hand Grip Dynamometer 120Kg
- Digital Hand Dynamometer Grip Strength Measurement Meter Auto Capturing Hand Grip Hand Grip Tester
- Digital Hand Biotronix Solution Forever Dynamometer Grip Strength Measurement
- Dynamometer Grip strength Assessment Device Hand Grip Strength Testing Equipment
- Hand Grip Dynamometer for Grip Strength Digital
- Dual probe laser therapy for pain relief treatment Advanced laser therapy device
- Class 4 Diode Laser 10W Therapy Unit For Muscle, Joint, And Pain Treatment
- Handy LASER therapy device for pain relief Portable LASER pain management tool
- Laser therapy for joint pain Portable laser pain relief device
- User-Friendly Class 4 Laser Device for Pain Relief & Rehabilitation
- OEM 4T 4-Channel TENS & EMS Physiotherapy Device Touchscreen, Portable
- Advanced Class IV Laser Therapy Device for Sports Injury Treatment
- OEM Custom Physiotherapy Knee Massager Infrared Heat Pain Relief Device with Vibration Function
- Non-invasive EMTT Painless Extracorporeal Magnetic Transduction Magneto Therapy
- Leg compression Therapy pump DVT relief Portable therapy Lymphedema treatment
- Pedo Cycle Leg Rehabilitation Tool Motorized Digital with Remote Control
- Pedal exerciser for physiotherapy Rehabilitation pedal exerciser
- Motorized pedal fitness machine Electric physiotherapy pedals
- Electromagnetic Pulse Therapy Device Loop Model for Clinics
- Pedal exerciser for seniors Motorized pedal exerciser for rehabilitation
- Electric exercise pedals Portable pedal exerciser Motorized pedal fitness machine
- physical therapy Electric Stroke Recovery Pedal Machine for Home Rehabilitation
- Pedo Cycle Pedal exerciser for physical therapy Leg Rehabilitation Tool Motorized Digital with Remote Control
- Electric Stroke Recovery Pedal Machine for Home Rehabilitation
- Pstics ABS Electric pedal cycle for upper and lower body exercises
- Pstics ABS Electric pedal bike for rehab Motorized exercise cycle for therapy
- Inflatable Cryo Compression Wrap with Cold Therapy Pad for Swelling and Pain
- Plastics ABS PEDO CYCLE Best electric pedal exerciser for elderly
- Plastics ABS Exercise Equipment Motorized Stroke Recovery Machine Pedo Cycle Electrical, Manual
- PEDO CYCLE Electric stationary pedal bike for physical therapy
- Electric pedal exerciser for senior exercises Pedo Cycle
- Clinical-Grade PEMT Loop Device-4500Hz Oscillation & Multi-Frequency Pulse
- Exercise Equipment Motorized Stroke Recovery Machine Pedo Cycle Electrical, Manual
- pedal exerciser for rehabilitation Electric mini pedal exerciser for rehabilitation
- Electric pedal exerciser for home use physical therapy
- Portable Manual Chiropractor Tool Four-Head Chiropractic Adjuster for Deep Tissue Massage
- Pedo cycle electric Motorized pedal exerciser for gradual rehabilitation
- Pedo cycle with electric motor Electric pedal exerciser for physical therapy progress
- Biotronix Rehabilitation Electric Pedo Cycle Seniors Physiotherapy Arm & Leg Pedal Training Device
- Electric pedo rehabilitation bike Electric pedal exerciser for professional physiotherapy
- Physio exerciser Electric pedal exerciser for professional rehabilitation
- Exercise Equipment Pedo Cycle Pedal Exerciser Leg Exerciser Pedal Device, Automatic
- electrical Pedo Cycle Pedal Exerciser Leg Exerciser Pedal Device, Automatic
- Biotronix Solution Forever Rehab Trainer Motorized Electrical for Legs and Arms Rehabilitation
- Biotronix Motorized Electric Mini Exercise Pedo Cycle Intelligent Remote Control Mini Ergometer
- Mini Electric Exercise Pedal Bike for Elderly Leg Arm Muscle Recovery Physical Rehabilitation
- Electric pedal exerciser for post-surgery physiotherapy
- Pedo Cycle Electrical with Leg Support for Paralysis and Stroke Recovery PAtinets
- Digital Automatic Leg Exerciser for Stroke Recovery Remote-Controlled Pedal Machine for Leg Exercise
- Pedo Cycle Motorized Rehabilitation Mini Under Exercise Bike Pedal Exercise Bike With Desk
- Physiotherapy Pedal Machine for Effective Rehabilitation with Leg Support
- Pedo Cycle Electrical Physical Therapy Equipment
- Motorized Leg Exercise Machine: Stroke Recovery Digital Rehab Exerciser
- Leg Exerciser with Remote Control and Motorized Function for Rehabilitating Legs
- Plastics ABS Exercise Equipment Imported Motorized Pedo Cycle, Manual
- Plastics ABS Exercise Equipment Pedo Cycle Motorized Electrical With Leg Support Digital Automatic, Manual
- Plastics ABS Digital Stroke Recovery Aid Automatic Rehabilitation Pedal Cycle, Size: Medium
- Plastics ABS Exercise Equipment Digital Legs And Arm Physiotherapy Device PEDO Cycle, Manual
- Pedal Machine for Effective Rehabilitation with Leg Support
- Digital Remote-Controlled Pedal Exerciser with Legs Supports, For Home, Fitness Equipment
- Leg Exercise Machine: Stroke Recovery Digital Rehab Exerciser
- Pedal Exerciser with Legs Supports, For Home, Fitness Equipment
- Motorized Pedo Cycle Digital Remote-Controlled Leg Exerciser for Stroke Recovery, For Household
- Biotronix Pedo Cycle Motorized for Lower Limb ( legs ) Rehabilitation
- Electric Pedal Exerciser for Elderly for Arm/Leg Exercise Cycling Trainer Rehabilitation
- Rehabilitation Electric Pedo Cycle Seniors Physiotherapy Arm & Leg Pedal Training Device
- Electrical Physio Pedo Cycle Motorized Automatic Forward & Reverse Motion Cabled Remote
- Pedo Cycle Motorized Electricl for Lower Limb ( legs ) Rehabilitation Physiotherapy Cycle
- Motorized Electric Mini Exercise Pedo Cycle Intelligent Remote Control Mini Ergometer
- ABS plastic Biotronix Motorized Pedocycle Physiotherapy
- Plastics ABS Exercise Equipment Pedal Cycle Physiotherapy Motorized Electrical, Manual
- Compact Brain and Nerve Rehab Device tDCS tACS tVNS tPCS
- Digital Neuro Modulation Therapy Device tDCS tPCS tACS tVNS
- High Quality Brain Rehab Machine tDCS tACS tVNS tPCS
- Konmed KM-560 Biofeedback Machine with EMG ETS Gaming Stimulator
- 4 in 1 Electrotherapy Device with Dual Channel Output System
- Konmed KM-560 EMG Biofeedback Equipment with Therapy Progress Tracking
- Konmed KM-560 Physiotherapy Biofeedback Machine with Multi Channel EMG
- Konmed KM-560 EMG Biofeedback Device with Clinical Therapy Programs
- Konmed KM-560 Biofeedback System with Real Time EMG Feedback
- KM-560 4 Channel Physiotherapy Biofeedback Equipment
- Konmed KM-560 Biofeedback Device with NMES Therapy Mode
- Konmed KM-560 EMG Biofeedback Machine with ETS Trigger
- Konmed KM-560 Biofeedback Machine with EMG Gaming and Stimulator
- Konmed KM-560 Biofeedback Device with Electrical Muscle Activation Mode
- Konmed KM-560 4 Channel EMG Biofeedback Equipment with Clinical Modes
- Konmed KM-560 Biofeedback Machine with EMG Signal Analysis
- Konmed KM-560 EMG Biofeedback System with Smart Therapy Functions
- Konmed KM-560 Physiotherapy Biofeedback Machine with Rehab Programs
- Konmed KM-560 EMG Biofeedback Device with Therapy Gaming Interface
- Konmed KM-560 4 Channel Biofeedback Equipment with Electrical Therapy
- Konmed KM-560 Biofeedback Equipment with Gaming Rehabs
- Konmed KM-560 Clinical EMG Biofeedback Device with ETSS
- Konmed KM-560 EMG Biofeedback Machine for Sports Rehabs
- Konmed KM-560 EMG Biofeedback System for Muscle Recoverys
- Konmed KM-560 EMG Biofeedback Equipment with NMES FES Modes
- Konmed KM-560 Physiotherapy Biofeedback Device for Rehab Use
- Konmed KM-560 Pelvic Floor Biofeedback Machine with EMG
- Konmed KM-560 4 Channel Biofeedback System with ETS Mode
- Konmed KM-560 Clinical Biofeedback Device with Gaming Feature
- Handy Laser Pain Relief Device erxrHandheld Model
- Multi Mode tDCS tPCS tACS tVNS Neuro Rehab Device
- Konmed KM-560 EMG Biofeedback System with Rehab Functions
- Motorized Ankle CPM Device With Remote Control and Adjustable Speed
- Phymaster Shockwave Physiotherapy Machine for Advanced Pain Treatment
- Reliable Motorized Ankle CPM For Regular Physiotherapy Sessions
- Phymaster Shockwave Professional Shockwave Pain Relief System
- The Role of Exoskeletons in Neurological Rehabilitation
- Exoskeleton Therapy Solutions for Long-Term Rehabilitation Care
- High-Performance 980nm Class IV Diode Laser Therapy Machine with Touch Screen, Air Cooling, and Emergency Switch for Clinical Use
- Electric Cryotherapy Device for Post-Injury and Post-Surgery Healing
- 750N Chiropractic Adjusting Tool Instrument Electric Spine Chiropractor Electric Impulse Gun Actuator Massager Professional Massage Equipment
- Konmed KM-560 Biofeedback Machine for Pelvic Therapy Use
- Phymaster Shockwave Shockwave Therapy Machine for Muscle & Joint Pain
- Advanced DVT Pump for Lymphedema and Varicose Veins Treatment
- Automatic Ankle CPM With Soft Straps For Comfortable Therapy Support
- Konmed KM-560 Professional Biofeedback Equipment with ETS
- Exoskeleton Therapy Devices for Motor Control Development
- Biotronix IASTM Tools kit
- Clinical Ankle CPM System With Long-Press Flexion & Stretch Modes
- Multi Therapy 4 in 1 Physiotherapy Machine for Clinic Equipment
- Back, Leg and Chest Strength Assessment Device for Rehabilitation
- Phymaster Pain Relief Machine for Physiotherapy Clinics
- Non-Surgical Pelvic Floor Muscle Rehab Chair for Clinical Wellness Care
- Phymaster Shockwave Advanced Pain Therapy Physiotherapy System
- Advanced Clinical Class 4 Laser Therapy Equipment for Pain Management
- Shockwave Therapy Device Pain Relief
- Exoskeleton Rehabilitation Systems Advancing Patient Outcomes
- Pneumatic shockwave therapy Radial shockwave therapy
- Advanced Brainwave Control And Cortical Therapy System
- Radial shockwave therapy for achilles tendinopathy
- Phymaster Shockwave Shockwave Therapy Device for Professional Use
- Biotronix Solution Forever Bubble Inclinometer Physiotherapy Equipment for Professionals Students
- Professional Grade Class 4 Laser Therapy Equipment for Rehab Use
- High Power Class 4 Laser Therapy Machine for Musculoskeletal Conditions
- Bio VNS-1 Advanced Multi Mode Brainwave And Vagus Therapy Equipment
- Clinical Brain And Neural Modulation Rehabilitation Machine
- Neuro Recovery And Cognitive Therapy System
- Advanced Neuro Rehabilitation Stimulation Equipment
- High Performance Class 4 Laser Therapy System for Rehabilitation Clinics
- Advanced High Power Class 4 Laser Therapy Machine for Rehab Centers
- View Similar Wireless Handheld Laser for Quick and Targeted Physiotherapy Relief Professional
- Konmed KM-560 EMG Biofeedback Machine with Therapy Monitoring System
- Advanced Class IV Laser Therapy Machine for Rehabilitation Therapy
- High Power Class 4 Laser Therapy Machine for Advanced Pain Relief Clinics
- Digital Brain Stimulation Device tDCS tACS tVNS tPCS Machine
- Professional Neuro Therapy Equipment tDCS tPCS tACS tVNS
- tDCS tACS tVNS tPCS Brain Neuro Therapy Device for Rehab Clinics
- High Efficiency Brain Stimulation Equipment tDCS tPCS tACS tVNS
- Multi Mode Neuro Therapy Equipment tDCS tPCS tACS tVNS Machine
- Portable Brain and Neuro Therapy Machine tDCS tACS tVNS tPCS
- Neuro Rehab Brain Stimulation Device tDCS tPCS tACS tVNS
- Advanced Clinical Class 4 Laser Therapy Device for Physiotherapy Practice
- Clinical Neuro Rehabilitation Therapy Device tDCS tPCS tACS tVNS
- Brain and Nerve Rehabilitation Device tDCS tACS tVNS tPCS
- Professional Neuro Stimulation Therapy System tDCS tPCS tACS tVNS
- Digital Brain and Neuro Therapy Equipment tDCS tACS tVNS tPCS
- Compact Neuro Rehab Therapy Device tDCS tPCS tACS tVNS
- Smart Neuro Modulation Therapy Equipment tDCS tPCS tACS tVNS
- Clinical Brain and Neuro Therapy System tDCS tACS tVNS tPCS
- Digital Neuro Rehab Therapy Machine tDCS tPCS tACS tVNS
- Advanced Neuro Rehab Therapy Equipment tDCS tPCS tACS tVNS
- Portable Neuro Therapy System tDCS tACS tVNS tPCS Device
- Brain and Nerve Stimulation Equipment tDCS tPCS tACS tVNS
- Electro Brain and Neuro Rehab Equipment tDCS tACS tVNS tPCS
- Multi Mode Brain and Neuro Therapy Machine tDCS tPCS tACS tVNS
- High Performance Neuro Rehab Therapy Device tDCS tACS tVNS tPCS
- Professional Brain and Nerve Therapy Equipment tDCS tACS tVNS tPCS
- Smart Neuro Stimulation Therapy Device tDCS tPCS tACS tVNS
- High Efficiency Neuro Therapy Machine tDCS tPCS tACS tVNS
- High Performance Neuro Stimulation System tDCS tACS tVNS tPCS
- Compact Brain Stimulation Therapy Equipment tDCS tPCS tACS tVNS
- Smart Neuro Modulation Device tDCS tACS tVNS tPCS Equipment
- Professional Brain Stimulation Therapy Device tDCS tPCS tACS tVNS
- Portable Neuro Therapy Equipment tDCS tACS tVNS tPCS Machine
- Compact Uni Gym Motorized Rehab Trainer for Arms and Legs
- tDCS tACS tVNS tPCS Neuro Stimulation Therapy Machine for Clinics
- Brain and Neuro Therapy Equipment tDCS tPCS tACS tVNS Machine
- Portable Brain Stimulation Therapy System tDCS tACS tVNS tPCS
- Advanced Neuro Therapy System tDCS tPCS tACS tVNS Equipment
- Clinical Neuro Stimulation Therapy Equipment tDCS tACS tVNS tPCS
- Compact Neuro Stimulation Therapy Machine tDCS tACS tVNS tPCS
- Dual Wavelength Diode Laser 980 1470 nm for Proctology Hemorrhoids Varicose Veins
- High Efficiency Brain Therapy Equipment tDCS tPCS tACS tVNS
- Smart Brain and Neuro Therapy Device tDCS tPCS tACS tVNS
- Digital Brain Stimulation Therapy System tDCS tPCS tACS tVNS
- PEMF Pulse Electromagnetic Therapy Loop Machine Premium Model
- UniGym Active Passive Resistive Training Device for Rehabilitation
- Clinical Brain Neuro Stimulation Device tDCS tPCS tACS tVNS
- Phymaster Shockwave Pain Management Shockwave Therapy Machine
- Portable Neuro Stimulation Therapy Equipment tDCS tPCS tACS tVNS
- Konmed KM-560 4 Channel EMG Biofeedback Device for Clinics
- Konmed KM-560 Biofeedback Machine with EMG and ETS Therapy
- Konmed KM-560 Physiotherapy Biofeedback Equipment with Stimulation
- Konmed KM-560 EMG Biofeedback Device for Muscle Recovery
- Konmed KM-560 Pelvic Floor Biofeedback Machine with NMES FES
- Konmed KM-560 Advanced EMG Biofeedback System for Rehab
- Konmed KM-560 Clinical Biofeedback Equipment for Therapy
- Konmed KM-560 Multi Channel Biofeedback Device for Physiotherapy
- Konmed KM-560 Professional EMG Biofeedback Machine
- Economical Model Class 4 Laser Therapy Equipment for Physiotherapy Use
- Konmed KM-560 4 Channel EMG Biofeedback Device with ETS Stimulator
- Konmed KM-560 EMG Biofeedback System with Gaming Rehab Mode
- Portable Class IV Laser Therapy Machine for Clinics
- Konmed KM-560 Physiotherapy Biofeedback Equipment with 4 Channel
- Konmed KM-560 4 Channel EMG Biofeedback Machine for Clinics
- Konmed KM-560 Rehab Biofeedback System with ETS NMES Modes
- Konmed KM-560 Physiotherapy Biofeedback Device with Therapy Modes
- Konmed KM-560 Biofeedback Machine with Electrical Muscle Stimulator
- Konmed KM-560 Physiotherapy EMG Biofeedback with Rehab Mode
- Class IV High Power Laser Therapy Machine for Pain Management
- Konmed KM-560 Biofeedback Machine for Pelvic Muscle Trainings
- Konmed KM-560 Professional Biofeedback Equipment for Therapy
- Konmed KM-560 EMG Biofeedback Machine with Multi Therapy Mode
- Konmed KM-560 4 Channel Physiotherapy Biofeedback machine Equipments
- Konmed KM-560 4 Channel Biofeedback System with Clinical Functions
- Konmed KM-560 Pelvic Floor Biofeedback Machine with Therapy Programs
- Konmed KM-560 Physiotherapy Biofeedback Device with FES Mode
- Konmed KM-560 4 Channel EMG Biofeedback Device with Advanced Therapy
- Konmed KM-560 Rehab Biofeedback Equipment with Multi Channel
- Exoskeleton Technology Advancing Clinical Recovery Outcomes
- Konmed KM-560 4 Channel EMG Biofeedback Device for Hospitals
- Konmed KM-560 EMG Biofeedback System with Muscle Activation
- Konmed KM-560 Professional Biofeedback Equipment with Therapy Modes
- Konmed KM-560 Advanced Biofeedback Device with Rehab Programs
- Konmed KM-560 EMG Biofeedback Machine with Therapy Control
- Konmed KM-560 Biofeedback Device with NMES Muscle Stimulation
- Konmed KM-560 4 Channel Physiotherapys Biofeedback Equipments
- Konmed KM-560 EMG Biofeedback System for Strength Training
- Konmed KM-560 EMG Biofeedback Equipment with Rehab Modes
- Konmed KM-560 EMG Biofeedback Equipment with Therapy Control
- Air Compression Radial ESWT -7 Heads, 10 Treatment Modes
- Konmed KM-560 Physiotherapy Biofeedback Device with Advanced Modes
- Konmed KM-560 Pelvic Rehab Biofeedback Machine with EMG Monitoring
- Konmed KM-560 Clinical Biofeedback Device with Rehab Programs
- Konmed KM-560 Rehab Biofeedback Machine with Smart Interfaces
- Konmed KM-560 EMG Biofeedback System for Clinical Training
- Konmed KM-560 4 Channel EMG Biofeedback Device with Smart Therapy
- Konmed KM-560 Biofeedback Machine with Electrical Therapy Mode
- Konmed KM-560 EMG Biofeedback System for Rehab Training
- Konmed KM-560 Physiotherapy Biofeedback Device with EMG Monitoring
- Konmed KM-560 EMG Biofeedback Machine for Clinical Therapy
- Konmed KM-560 Clinical EMG Biofeedback Device with Rehab Mode
- Konmed KM-560 4 Channel EMG Biofeedback System with Therapy Control
- Phymaster Shockwave Shockwave Therapy Device for Orthopedic Treatment
- Konmed KM-560 Biofeedback Equipment with Muscle Training Programs
- Konmed KM-560 EMG Biofeedback Device with Clinical Applications
- Konmed KM-560 EMG Biofeedback Device with Muscle Recovery
- Konmed KM-560 Rehab Biofeedback Equipment with EMG Feedback System
- Konmed KM-560 Physiotherapy EMG Biofeedback with Rehab Programs
- Konmed KM-560 Biofeedback Machine for Pelvic Muscle Strengthening
- Konmed KM-560 EMG Biofeedback System with Therapy Monitoring
- Konmed KM-560 Professional Biofeedback Equipment for Rehab Clinics
- Konmed KM-560 EMG Biofeedback Machine with Multi Channel Output
- Konmed KM-560 Biofeedback Device with Electrical Muscle Therapy
- Konmed KM-560 Biofeedback Machine with Stimulator Therapy
- Konmed KM-560 EMG Biofeedback System for Muscle Coordination
- Konmed KM-560 EMG Biofeedback Machine for Rehabilitation Centers
- Konmed KM-560 Clinical EMG Biofeedback Device with Smart Therapy
- 7 Head Pneumatic Radial Shockwave Therapy System-10 Modes for Pain Relief & Rehab
- Konmed KM-560 Clinical Biofeedback Equipment with EMG Monitoring
- Smart Brain And Autonomic Regulation System
- 4 in 1 Physiotherapy Equipment for Pain Relief & Rehabilitation
- Advanced Class 4 Laser Therapy Machine for Musculoskeletal Pain
- Multi Channel Brainwave And Vagus Activation Therapy Equipment
- Four In One Neuro Stimulation Therapy Equipment
- Cryo Therapy Motorised Cooling System for Pain Relief & Rehab
- Motorised Cryo Cooler Unit for Physiotherapy Clinics
- Medical Multi Mode Brain Therapy Equipment
- Hospital Grade Non Invasive Neuromodulation Therapy System
- High Intensity Clinical Class 4 Laser Therapy Equipment
- Digital Transcranial Direct And Alternating Current Stimulation System
- Phymaster Shockwave Physiotherapy Machine for Professional Pain Therapy
- KM521 Advanced Pelvic Floor Muscle Training Machine for Rehabilitation
- Konmed KM-560 EMG Biofeedback Device with Electrical Therapy
- Portable Laser Physiotherapy Machine for Pain Relief & Muscle Recovery
- High Precision Bubble Inclinometer For Physical Therapy Students ,Professionals
- Exoskeleton Rehabilitation Robot for Stroke and Paralysis Recovery
- Deep Precision Focus Shockwave Therapy Machine for Advanced Pain Clearance
- Cryopush Multi-Joint Cold Therapy and Compression Machine
- Integrated Brain Recovery And Neuromodulation Device
- Digital Brainwave Synchronization And Recovery Equipment
- Heavy Duty Motorized Ankle Motion Therapy Device
- Class 4 Laser Therapy Device for Pain Management and Recovery Clinics
- Cold Therapy Circulating System with Compression for ACL and Injury Recovery
- Phymaster Shockwave Pain Management Physiotherapy Equipment
- Advanced Brainwave And Vagus Nerve Neural Activation System
- Phymaster Shockwave Advanced Pain Therapy Equipment for Physiotherapy
- Motorized Rehabilitation Pedo Cycle Motorized, Manual
- Phymaster Shockwave Professional Pain Relief Physiotherapy Device
- Phymaster Shockwave Pain Relief Therapy System for Physiotherapy Centers
- Phymaster Shockwave Physiotherapy Equipment for Deep Tissue Therapy
- Phymaster Shockwave Advanced Shockwave Therapy Equipment for Pain Relief
- Phymaster Shockwave Physiotherapy Machine for Professional Clinics
- Wrist CPM Premium Model-Motorized Electrical Wrist Rehabilitation Device with Remote Control
- Compact Brain Rehab Machine tDCS tACS tVNS tPCS Device
- Remote Controlled Ankle CPM Machine For Safe ROM Development
- Class 4 980nm Diode Laser Therapy Machine for Physiotherapy
- KM521 Rechargeable Pelvic Muscle Stimulator for Women's Health & Rehabilitation
- Phymaster Shockwave Pain Relief Physiotherapy System
- KM-580 Advanced Biofeedback Therapy Device for Stroke and Paralysis Rehabilitation
- Magneto Therapy Unit Electrotherapy Unit
- Cold Compression Therapy Device with Large 3.6L Water Reservoir
- Ultra-Focused Shockwave Treatment System for Deep Musculoskeletal Healing
- Phymaster Shockwave Physiotherapy Device for Clinical Pain Relief
- Advanced Class IV Laser Therapy Machine for Pain Management
- Medical Brainwave Modulation And Vagus Nerve Stimulation Device
- Digital Cold Compression Device for Physiotherapy & Rehabilitation
- Digital Elbow Continuous Passive Motion Trainer
- High-Quality Elbow CPM Machine Gentle Motion for Pain Relief and Recovery
- Imported Elbow CPM Machine with Ergonomic Design for Comfortable Physiotherapy
- Pulse Electromagnetic Therapy Device for Physiotherapy Clinics
- Multi-Function Elbow CPM Machine Adjustable Speed and Angle Settings
- Portable Elbow CPM Physiotherapy Device
- Rechargeable Laser Therapy Device for Pain Relief, Inflammation & Circulation
- Electric pedal machine for senior fitness
- Smart Exoskeleton Systems for Neuro and Orthopedic Rehab
- KM521 Clinical Pelvic Floor Rehabilitation Stimulator for Muscle Recovery
- High Precision Ankle CPM Device For Range of Motion Improvement
- Konmed KM-560 EMG Biofeedback Device with Muscle Activation
- Compact Electromagnetic Radial ESWT Device 10 Modes, 5-250 Hz
- Dual Light Laser Therapy Device with Safety Goggles & Adjustable Stand
- Advanced Multi Channel TDCS TPCS TACS TVNS Brain Stimulation System
- Intelligent Exoskeleton Devices for Controlled Movement Training
- Portable Neuro Rehab Machine tDCS tACS tVNS tPCS Device
- Digital Transcranial And Vagus Nerve Neuromodulation Therapy Equipment
- Physiotherapy Physical Equipment SA-SW17 Physio Magneto
- Hospital-Use Focus Shockwave Apparatus for Trigger Point Release
- Medical Grade Pelvic Floor Rehab Chair for Incontinence Treatment
- Clinical Focus Shockwave Therapy Equipment for Advanced Physiotherapy
- Multi Channel Brain And Vagus Activation System
- Portable Pain Relief Rf Slimming Tecar Therapy Machine High Frequency Fat Burning Device
- Clinical-Grade Pelvic Floor Rehab Stimulator with TFT Display-KM521
- Target-Pro Focus Shockwave Therapy Unit for Deep Muscle Rehabilitation
- Clinical Stroke Recovery And Brain Stimulation Machine
- Exoskeleton-Driven Therapy for Improved Mobility and Balance
- Digital Pelvic Floor Muscle Therapy Chair for Pelvic Health Management
- Digital Clinical Pelvic Floor Therapy Chair with Preset Programs
- Clinical Pelvic Floor Muscle Strength Training Chair for Incontinence Care
- Lower Limb Exoskeletons for Gait and Mobility Rehabilitation
- Digital Clinical Pelvic Muscle Conditioning Chair with Therapy Programs
- Medical Pelvic Floor Muscle Activation Chair for Rehabilitation Programs
- Exoskeleton-Assisted Therapy for Motor Control Improvement
- Portable LASER physiotherapy tool Handheld LASER pain management device
- Phymaster Shockwave Physiotherapy Device for Muscle Pain Treatment
- KM-580 Multi-Function Biofeedback Machine with Dual EMG and Four Channel Stimulation
- Pedo Cycle Motorized fitness exerciser Electrical exercise cycle Rehabilitation
- KM521 Pelvic Muscle Stimulator with Dual Channel Output & TFT Display
- Advanced Physiotherapy Class 4 Laser Therapy System High Output
- High Performance Class 4 Laser Therapy System for Physiotherapy Experts
- Medical-Grade DVT Therapy System with 8 Modes and 4 Chamber Design
- Professional Grade High Power Class 4 Laser
- High Power Class 4 Laser Therapy System for Orthopedic Physiotherapy
- Professional Brain Activation And Neuro Rehab System
- DVT Pneumatic Compression Machine for Medical Use
- Professional DVT Therapy Device for Leg, Waist, and Arm Treatment
- Professional Brain And Cognitive Enhancement Equipment
- Medical Exoskeleton Solutions for Neuro Motor Rehabilitation
- KM-580 EMG ETS NMES FES Therapy Unit for Pelvic, Sports & Neuromuscular Recovery
- Cold Therapy Motorized Cooler with Shoulder, Ankle, Knee & Hip Wraps
- Multi Mode Brain And Vagus Nerve Therapy System
- Medical Exoskeleton Solutions for Precision Physiotherapy
- Automated Exoskeleton Robot for Upper and Lower Limb Rehabilitation
- Exoskeleton Technology Supporting Modern Physiotherapy Solutions
- Hospital Use Ankle CPM System With Digital Angle Control
- Smart Robotic Rehabilitation Exoskeleton for Hospitals and Physio Centres
- Phymaster Shockwave Shockwave Therapy Device for Sports Rehabilitation
- Automatic Wrist Joint CPM Machine-High-Performance Rehabilitation Device
- Phymaster Shockwave Pain Relief Physiotherapy Equipment for Clinics
- Precision Neurological Stimulation And Vagus Therapy System
- 6 Chamber Digital Air Compression DVT Pump For Hospital And Clinic Use
- Clinical Pelvic Floor Muscle Training Chair for Incontinence Management
- Therapeutic Focus Shockwave Treatment Instrument for Rehabilitation
- Clinical Pelvic Floor Muscle Therapy Chair for Complete Rehabilitation
- Advanced Exoskeleton Therapy for Stroke and Paralysis Recovery
- Medical Pelvic Muscle Strength Training Chair for Incontinence Therapy
- Wearable Robotics Driving the Future of Physical Therapy
- High-Intensity Pelvic Muscle Rehabilitation Chair for Physiotherapy Centers
- Digital TDCS TACS TVNS Multi Therapy Brain Device
- Clinical Pelvic Muscle Strength Training Chair for Long-Term Care
- Advanced Biofeedback Therapy Equipment-KM-580 for EMG and ETS Training
- Non-Invasive Pelvic Muscle Strengthening Chair for Clinical Use
- Handheld Laser Physiotherapy Device for Pain Relief, Muscle Recovery & Circulation Improvement
- Advanced Multi Current Brain And Neural Activation Device
- Digital Pelvic Floor Muscle Strength Chair for Pelvic Health Programs
- Professional Multi Therapy Neuro Rehabilitation Equipment
- Hospital Grade Brain Plasticity And Recovery Device
- Motorized Cold Cooling Unit for Post-Surgery & Sports Injury Recovery
- Professional Multi Channel Brain Activation And Recovery Device
- Professional Multi Therapy Brain And Neural Recovery Machine
- Hospital Grade Non Invasive Brainwave And Vagus Therapy System
- Handheld Laser Therapy Device 650nm/808nm-Digital Display, Protective Goggles Included
- Non-Surgical Pelvic Floor Muscle Rehab Chair for Pelvic Wellness
- Hospital Grade Multi Mode Brain And Nerve Device
- Phymaster Shockwave Shockwave Therapy Equipment for Clinical Use
- Digital Multi Channel Transcranial Brain Stimulation Equipment
- Integrated Neuro Stimulation And Brainwave Regulation System
- Medical Neuromodulation And Cognitive Therapy Equipment
- Advanced Brain And Neural Plasticity Therapy System
- Medical Neuromodulation And Vagus Therapy Machine
- Precision Pulse Focus Shockwave Machine for Accelerated Healing Response
- Phymaster Shockwave Shockwave Therapy Machine for Muscle Recovery
- Precision Brain Stimulation And Vagus Therapy Equipment
- Digital Brainwave Enhancement And Neuro Therapy Equipment
- Clinical Pelvic Floor Muscle Rehab Chair for Pelvic Health Treatment
- Wearable Exoskeleton Systems for Precision Movement Control
- Wearable Exoskeletons for Clinical Movement Training
- Pelvic Floor Muscle Training Chair for Post-Natal & Geriatric Care
- UniGym Digital Physiotherapy Machine with App-Based Gaming Interaction
- Handy Laser Therapy Device with Adjustable Stand-Safe, Rechargeable, & Easy to Use
- Integrated Brainwave Regulation And Vagus Stimulation Equipment
- Phymaster Shockwave Physiotherapy Machine for Muscle & Joint Recovery
- Phymaster Shockwave Pain Management Physiotherapy Machine
- Non-Invasive Pelvic Floor Muscle Training Chair for Physiotherapy Use
- Professional Pelvic Floor Stimulation Chair for Hospital & Clinical Use
- Compact Neuro Stimulation Machine tDCS tPCS tACS tVNS
- Wearable Exoskeleton Technology for Assisted Motor Training
- Wearable Medical Exoskeletons for Advanced Rehab Practice
- Wearable Medical Exoskeletons for Progressive Physiotherapy
- Wearable Medical Exoskeletons for Structured Recovery Programs
- Pneumatic Shockwave Clinical Radial ESWT Device 7 Heads 10 Modes
- Professional Brainwave Synchronization And Therapy Machine
- IASTM Tools 5 pc 304 Grade Stainless Steel
- Advanced Exoskeleton Rehabilitation Technology for Clinical Care
- Professional PEMF Loop Therapy Device for Physiotherapy Clinics
- Advanced Wearable Exoskeleton Systems for Physiotherapy Practice
- Portable tDCS tACS tVNS tPCS Brain Stimulation Machine
- Clinical Exoskeleton Devices Supporting Advanced Therapy
- Clinical-Grade Wearable Exoskeletons for Rehab Programs
- Iastm Tools Set of 5 Gua Sha Gloves and Case SS Steel Gua Sha Tools for Mobilizing Soft Tissue
- Exoskeleton-Assisted Physiotherapy for Progressive Motor Recovery
- Exoskeleton-Based Rehabilitation for Controlled Motor Training
- Compact Neuro Modulation Therapy Machine tDCS tPCS tACS tVNS
- Exoskeleton Rehabilitation Systems for Professional Clinical Use
- Exoskeleton Rehabilitation Systems for Strength, Control
- Exoskeleton Rehabilitation Technology for Safe Movement
- Phymaster Shockwave Physiotherapy Device for Chronic Pain Treatment
- Exoskeleton Systems for Upper Limb and Lower Limb Training
- Exoskeleton Systems Supporting Strength Mobility and Balance
- Exoskeleton Therapy Devices for Advanced Recovery Pathways
- Exoskeleton Technology for Assisted Motor Recovery Programs
- Non-Invasive Pelvic Floor Muscle Rehab Chair for Clinical Treatment
- Exoskeleton Therapy Systems Enhancing Motor Performance
- Exoskeleton Therapy Technology for Structured Rehabilitation
- High-Frequency Focus Shockwave System for Rapid Soft Tissue Repair
- Non-Surgical Pelvic Floor Muscle Therapy Chair for Patient Recovery
- Medical Exoskeleton Systems Supporting Therapy Excellence
- Medical-Grade Exoskeleton Systems for Modern Rehab Clinics
- Robotic Exoskeleton Devices for Assisted Movement Recovery
- Medical-Grade Exoskeleton Technology for Physiotherapy Centers
- Hospital Grade Multi Therapy Brain Stimulation System
- Medical-Grade Wearable Exoskeletons for Muscle Strength Training
- Medical-Grade Wearable Exoskeletons for Physiotherapy Centers
- Smart Brain And Vagus Regulation Therapy Equipment
- Advanced Pelvic Muscle Rehabilitation Chair for Physiotherapy Centers
- Clinical tDCS tPCS tACS tVNS Neuro Stimulation Equipment
- Robotic Exoskeleton Devices for Consistent Recovery Training
- Robotic Exoskeleton Devices for Precision Clinical Training
- Robotic Exoskeleton Systems Supporting Functional Training
- Robotic Exoskeleton Therapy for Advanced Motor Training
- Robotic Wearable Exoskeletons for Advanced Physiotherapy Use
- Robotic Wearable Exoskeletons for Consistent Physiotherapy
- Robotic Wearable Exoskeletons for Progressive Physiotherapy
- Smart Exoskeleton Rehabilitation Systems for Modern Clinics
- Smart Robotic Exoskeletons for Precision Physiotherapy
- Compact 4 in 1 Physiotherapy Device for Daily Pain Therapy
- Smart Robotic Exoskeletons for Precision Rehab Sessions
- Smart Wearable Exoskeletons for Precision Therapy Sessions
- Smart Wearable Exoskeletons for Targeted Therapy Sessions
- Wearable Exoskeleton Systems for Assisted Functional Movement
- Pelvic Floor Muscle Strengthening Chair for Post-Natal Recovery
- Phymaster Shockwave Shockwave Therapy Equipment for Rehab Centers
- Smart Brain Stimulation Equipment tDCS tACS tVNS tPCS
- Digital Brainwave Synchronization And Neuro Modulation System
- Digital Ankle Continuous Passive Motion Device For Post-Surgery Recovery
- Advanced PEMT Loop Therapy Device for Physiotherapy Clinics
- PEMT Pulse Electromagnetic Therapy Loop Device Touch Screen Model
- Clinical Pelvic Floor Muscle Rehab Chair for Incontinence Treatment
- Phymaster Shockwave Therapy Equipment for Orthopedic Rehabilitation
- Portable Neuro Stimulation Machine tDCS tACS tVNS tPCS
- Multi Mode Brain Stimulation Machine tDCS tPCS tACS tVNS
- Advanced 4 in 1 TENS EMS IFT Combo Unit for Physiotherapy Clinics
- Chiropractor Device 750N Electrical
- UniGym Advanced Rehab Trainer for Post-Surgery & Injury Recovery
- UniGym Arm and Leg Exercise Therapy Machine for Stroke Recovery
- Phymaster Shockwave Shockwave Therapy System for Physiotherapy Centers
- Phymaster Shockwave Physiotherapy Equipment for Tendonitis Treatment
- Medical Pelvic Muscle Therapy Chair for Incontinence Management
- Clinical Pelvic Floor Muscle Training Chair for Post-Surgical Recovery
- Shock Wave Therapy Machine With ED Treatment Pain Relief, For Hospital
- Compact Neuro Therapy Machine tDCS tPCS tACS tVNS Device
- Advanced Cold Therapy Machine for Swelling & Inflammation Control
- Portable Red Light Therapy for Shoulder Back Muscle Knee Joints Pain White
- Clinical Use Exoskeletons for Advanced Physiotherapy Programs
- Digital Laser Therapy Device with Cooling Fan-Adjustable 5-30 Minute Timer
- UniGym Digital Exercise Therapy Device for Stroke Recovery
- Professional Pedo Cycle Pedal Exerciser for Home Therapy Digital Electrical
- UniGym Rehabilitation Trainer for Home and Clinic Use
- Phymaster Shockwave Physiotherapy Equipment for Professional Pain Care
- UniGym: Your Portable Home Gym for Daily Exercise
- UniGym Smart Rehab Exercise System with Gaming Interaction
- Precision-Control Focus Shockwave Device for Advanced Physiotherapy Care
- smart tecar monopole rf radiofrequency ret cet physiotherapy machine
- Phymaster Shockwave Physiotherapy Equipment for Fast Recovery
- Digital Cryotherapy Compression System for Sports Physiotherapy
- UniGym Digital Exercise Therapy Device for Stroke and Rehabilitation
- Multi Functional Neuro Rehab Device tDCS tACS tVNS tPCS
- Motorised Cryo Cold Therapy Unit With 4 Attachments
- Automatic Ankle Joint CPM for Plantar Flexion and Dorsiflexion Training
- Compact Laser Therapy Machine with USB Type-C, Digital Display & Timer
- Digital Neuro Modulation Machine tDCS tPCS tACS tVNS
- Professional Brain Rehab Equipment tDCS tPCS tACS tVNS
- Professional Class 4 Laser Physiotherapy Equipment for Rehabilitation
- Smart Brain and Nerve Therapy System tDCS tACS tVNS tPCS
- Portable Brain and Nerve Therapy Device tDCS tACS tVNS tPCS
- Advanced Cortical And Vagus Nerve Regulation Equipment
- Clinical Brain and Nerve Therapy Machine tDCS tPCS tACS tVNS
- Air Compression Therapy DVT 6 Chamber 6 Modes for Legs
- Electric Cold Therapy System with Shoulder, Knee, Ankle and Back Wraps
- Advanced Cortical Stimulation And Vagus Therapy Unit
- Robotic Exoskeleton Devices for Controlled Motor Training
- Compact Neuro Rehab Equipment tDCS tPCS tACS tVNS Machine
- Phymaster Shockwave Professional Physiotherapy Shockwave Device
- Clinical Neuro Therapy Equipment tDCS tPCS tACS tVNS Device
- Konmed KM-560 Physiotherapy Biofeedback Device with Clinical Programs
- Konmed KM-560 EMG Biofeedback Device with Muscle Reeducation
- Konmed KM-560 Rehab Biofeedback Machine with Therapy Monitoring
- Konmed KM-560 Clinical Biofeedback Device with Muscle Activation
- Konmed KM-560 EMG Biofeedback Stimulator with Smart Control
- Konmed KM-560 4 Channel Biofeedback System with EMG Therapy
- Konmed KM-560 Physiotherapy Biofeedback Device with Muscle Training
- Konmed KM-560 EMG Biofeedback Equipment with Clinical Modes
- Konmed KM-560 4 Channel EMG Biofeedback Device with Therapy Functions
- Konmed KM-560 Biofeedback Machine with EMG Muscle Rehab
- Konmed KM-560 Professional Biofeedback Machine with Therapy Modes
- Konmed KM-560 EMG Biofeedback Device with Clinical Monitoring
- Konmed KM-560 Biofeedback Equipment with Muscle Activation Modes
- Konmed KM-560 4 Channel EMG Biofeedback System with Rehab Features
- Konmed KM-560 Clinical EMG Biofeedback Device with Therapy Programs
- Konmed KM-560 EMG Biofeedback Machine for Clinical Applications
- Konmed KM-560 Physiotherapys Biofeedback Device with EMG Analysis
- Konmed KM-560 EMG Biofeedback System for Rehab Exercises
- Konmed KM-560 Biofeedback Machine with Electrical Stimulation Mode
- Konmed KM-560 Pelvic Floor EMG Biofeedback Therapy Machine
- 850W PEMT Loop Therapy-Magnetic Therapy & Oscillation Device for Joint & Muscle Healing
- Konmed KM-560 Stroke Rehabilitation Biofeedback Device
- Konmed KM-560 EMG Biofeedback System for Muscle Coordinations
- Konmed KM-560 Rehab Biofeedback Machine with Therapy Settings
- Konmed KM-560 Clinical Biofeedback Device with Muscle Training
- Phymaster Shockwave Therapy System for Clinical & Hospital Use
- Konmed KM-560 EMG Biofeedback Stimulator with Rehab Control
- Konmed KM-560 4 Channel Biofeedback System with Clinical Therapy
- Konmed KM-560 Physiotherapys Biofeedback Device with Therapy Modes
- Konmed KM-560 Biofeedback Machine with EMG Therapy Programs
- Konmed KM-560 4 Channel EMG Biofeedback Device with Rehab Features
- Konmed KM-560 4 Channel EMG Stimulator with Biofeedback Therapy
- Konmed KM-560 Multi Channel EMG Therapy Device for Rehabilitation
- Konmed KM-560 Electrotherapy Biofeedback Device for Physiotherapy
- Konmed KM-560 Clinical EMG Biofeedback System for Hospitals
- Konmed KM-560 EMG Muscle Training and Rehab Machine
- Konmed KM-560 Advanced Biofeedback Physiotherapy Equipment
- Konmed KM-560 Physiotherapy EMG Biofeedback Machine with Touchscreen
- Konmed KM-560 4 Channel Rehab Biofeedback Equipment for Clinics
- Konmed KM-560 EMG Biofeedback Device with NMES FES ETS Therapy
- Phymaster Shockwave Therapy Equipment for Non-Surgical Pain Relief
- Konmed KM-560 EMG Based Muscle Recovery and Rehab Device
- Konmed KM-560 Rehab Biofeedback Equipment with Multi Therapy Modes
- Konmed KM-560 Biofeedback Machine for Pelvic Muscle Recovery
- Konmed KM-560 Physiotherapy EMG Biofeedback with Therapy Control
- Konmed KM-560 4 Channel EMG Biofeedback Device for Rehab Clinics
- Konmed KM-560 EMG Biofeedback System with Muscle Coordination
- Konmed KM-560 Advanced Biofeedback Device with Clinical Functions
- Konmed KM-560 EMG Biofeedback Machine with Therapy Settings
- Konmed KM-560 Biofeedback Device with Electrical Muscle Training
- Konmed KM-560 Advanced Biofeedback Device with Therapy Options
- Konmed KM-560 Biofeedback Machine with EMG Rehab Training
- Konmed KM-560 Smart Biofeedback Physiotherapy Machine
- Konmed KM-560 EMG Biofeedback System with Interactive Training
- Konmed KM-560 Neuromuscular Rehab Biofeedback Equipment
- Konmed KM-560 Digital EMG Biofeedback Device for Clinics
- Konmed KM-560 Portable EMG Physiotherapy Biofeedback Machine
- Konmed KM-560 Professional Physiotherapy Biofeedback Equipment
- Konmed KM-560 EMG Electrotherapy Device for Rehabilitation Centers
- Konmed KM-560 Digital Biofeedback Machine for Physiotherapy Treatment
- Konmed KM-560 Rehab Equipment with EMG Biofeedback and Stimulation
- Konmed KM-560 Clinical Physiotherapy Device with EMG Feedback System
- Konmed KM-560 4 Channel Muscle Stimulator with Biofeedback Therapy
- Shockwave Head Therapy Machine Massager, 7 Transmitters Effective Physical Shock Wave Anti-Cellulite Electromagnetic Deep Muscle Relax, Multicolour
- Silent Motor Ankle CPM Unit For Smooth Passive Movement
- Non-Surgical Pelvic Floor Muscle Conditioning Chair for Pelvic Wellness
- Clinical Pelvic Floor Rehabilitation Chair for Incontinence Management
- High-Performance Exoskeleton Devices for Rehabilitation Clinics
- Multi Functional tDCS tPCS tACS tVNS Neuro Device
- 7-Head Radial ESWT-Pneumatic, 10 Modes, Adjustable Energy
- Portable Ankle CPM Machine For Dorsiflexion-Plantar Flexion Therapy
- Multi Functional Neuro Stimulator tDCS tACS tVNS tPCS
- Neuro Rehab Therapy Device tDCS tPCS tACS tVNS
- 750N Chiropractic Adjusting Tool Instrument Electric
- Exoskeleton Technology Supporting Safe and Effective Rehabilitation
- Clinical Pelvic Floor Muscle Therapy Chair for Post-Operative Care
- Motorized Cryo Cooling System for Physiotherapy and Post-Surgery Recovery
- Rechargeable 670mW Laser Therapy Device 650nm Red + 808nm Infrared for Pain Relief
- Advanced Neuro Therapy Equipment tDCS tACS tVNS tPCS
- Affordable motorized electrical pedal exerciser for home therapy
- Economical Model Class 4 Laser Therapy Equipment for Pain Management
- Phymaster Shockwave Therapy Machine for Non-Surgical Pain Management
- Hospital Approved Pelvic Muscle Training Chair for Physiotherapy Therapy
- Motorized Digital Rehabilitation Pedal Bike Pedo Cycle Digital Electrical Motorized
- Compact Brain Therapy Machine tDCS tACS tVNS tPCS
- Magneto Therapy Unit
- Professional Brain Therapy Equipment tDCS tPCS tACS tVNS
- Portable Brain Stimulator Equipment tDCS tACS tVNS tPCS
- Professional Neuro Rehab Machine tDCS tACS tVNS tPCS
- Multi-Mode Pneumatic Shockwave Therapy Device
- Manual Chiropractic Adjusting Tool with Four Heads
- Clinical Pelvic Floor Muscle Stimulation Chair for Physiotherapy Clinics
- Electro Neuro Therapy Device tDCS tACS tVNS tPCS
- Class 4 Laser Therapy Equipment for Joint Pain and Rehabilitation
- Class IV Laser Therapy Machine for Pain Relief Clinics
- High Power Class IV Laser Therapy System for Orthopedic Clinics
- Class 4 Laser Therapy Device for Rehabilitation and Pain Management
- Economical Model Class IV Laser Therapy Machine for Sports Injury Clinics
- Class IV Laser Therapy Device for Physiotherapy and Sports Rehab
- Economical Class 4 Laser Therapy Equipment for Pain Management Clinics
- Professional Class IV Laser Therapy Machine for Rehabilitation Therapy
- High Power Class 4 Laser Therapy Equipment for Muscle Injury Treatment
- High Intensity Class 4 Laser Therapy Machine for Pain Relief Therapy
- Professional Class IV Laser Therapy System for Pain Treatment
- Economical Model Class IV Laser Therapy Device for Physiotherapy Hospitals
- High Performance Neuro Therapy Device tDCS tPCS tACS tVNS
- Advanced Class IV Laser Therapy System for Pain Relief Clinics
- Advanced Neuro Therapy Device tDCS tPCS tACS tVNS
- Class IV Laser Therapy Equipment for Pain Relief and Physiotherapy Clinics
- Class IV Laser Therapy System for Orthopedic and Sports Injury Treatment
- Class IV Laser Therapy Equipment
- Economical Model Class 4 Laser Therapy Equipment
- Class 4 Laser Therapy Device for Pain Relief and Rehabilitation
- Class 4 Laser Physiotherapy Device for Rehabilitation Centers
- Therapy Machine for Orthopedic Pain Management Clinics
- Class IV Laser Therapy System for Pain Relief Clinics
- Class IV Laser Therapy Machine for Physiotherapy Clinics
- Class 4 Laser Physiotherapy Equipment for Pain Management
- Professional Class IV Laser Therapy Machine for Physiotherapy Hospitals
- High Power Class 4 Laser Therapy System for Pain Relief Treatment
- Professional Pneumatic Shockwave Radial Therapy Machine
- Clinical Neuro Therapy Device tDCS tPCS tACS tVNS
- Professional Class IV Laser Therapy System for Rehabilitation Therapy
- Class 4 Laser Therapy Device for Orthopedic and Sports Rehabilitation
- Economical Model Class IV Laser Therapy Equipment for Pain Management
- Clinical Class 4 Laser Physiotherapy Machine for Pain Relief Clinics
- Economical Class 4 Laser Therapy Machine for Orthopedic Clinics
- Class 4 Laser Therapy Machine for Sports Injury Pain Relief
- Economical Model Class IV Laser Therapy Device for Physiotherapy Clinics
- High Intensity Class 4 Laser Physiotherapy Equipment for Pain Clinics
- Class IV Laser Therapy Equipment for Rehabilitation and Recovery Clinics
- High Power Class IV Laser Therapy Equipment for Rehabilitation Centers
- Professional Class 4 Laser Physiotherapy System for Pain Relief Treatment
- Class IV Laser Therapy Device for Orthopedic and Muscle Pain Relief
- Professional Class 4 Laser Therapy Equipment for Physiotherapy Hospitals
- Economical Class IV Laser Therapy Equipment for Pain Treatment Clinics
- Class 4 Laser Therapy Machine for Joint Pain and Recovery
- High Intensity Class 4 Laser Therapy Device for Rehabilitation Clinics
- Advanced Class IV Laser Therapy System for Muscle Pain Relief
- Economical Model Class 4 Laser Therapy Machine for Physiotherapy Centers
- Class IV Laser Therapy Equipment for Sports Injury Treatment Clinics
- Professional Class 4 Laser Physiotherapy Device for Pain Management
- High Power Class IV Laser Therapy Machine for Orthopedic Rehabilitation
- Advanced Class IV Laser Therapy Machine for Sports Injury Clinics
- Class 4 Laser Therapy Equipment for Joint Pain Rehabilitation
- Economical Class IV Laser Therapy Machine for Physiotherapy Hospitals
- High Power Class IV Laser Therapy Device for Muscle Injury Recovery
- Class 4 Laser Therapy Machine for Orthopedic Rehabilitation Centers
- Economical Model Class IV Laser Therapy System for Pain Treatment Clinics
- Clinical Class 4 Laser Physiotherapy Equipment for Rehabilitation Therapy
- Advanced Class IV Laser Therapy Equipment for Pain Management Clinics
- Class 4 Laser Therapy Machine for Physiotherapy Centers
- Impulse Waves Therapy Machine
- Biotronix Electronic Digital Goniometer
- Air Pressure Therapy System
- Biotronix InterCompression Sequential Lymphatic Drainage DVT Pump
- Biotronix ESWT GINHA HL1602 Portable Shockwave Therapy Physiotherapy
- Advanced Shockwave Therapy
- Biotronix Full Body Heating Blanket Far IR Infrared 2 Zone
- Pneumatic Shockwave Therapy Physiotherapy Device
- Biotronix Medical Pedal Motorized Electric Train Arm Leg Bicycle Trainer Elderly People Pedo Cycle
- Biotronix Motorized Electrical Digital Electronic Remote Controlled Physiotherapy Rehab Tilt Table
- Biotronix Motorized Pedocycle Physiotherapy
- Biotronix Rehabilitation Electric Pedo Cycle Seniors Physiotherapy Device
- Electronic Digital Display Joint Angle Measurement Goniometer Physio Chiropractor Research
- Full Body 7 Heads ED Treatment Physiotherapy
- MS Hot and Cold Contrast Physiotherapy
- Motorized Pedo Cycle
- Physiotherapy Ball Bosu Ball
- Portable Compact Mini Model Physiotherapy and Rehabilitation Shockwave Therapy
- Rechargeable Handheld Laser Therapy For Relieve Neck Back Arthritis Pain
- Remote Controlled Hi Low Tilt Table Physiotherapy and Rehabilitation Height Adjustable Tilting Table
- Shockwave Therapy Machine Shock Wave Body Muscle Massager
- Un Weight Mobility Trainer Partial Body Weight Support System Suspended
- Shock Wave Shockwave Joints Pain Physiotherapy
- Back Pain Relief Low Back Stretcher with Vibration And Heat
- Ultrasound Cavitation EMS Infrared Machine Slimming Physiotherapy
- Digital 6 Chambers Lymphatic Drainage Pump DVT Machine Pressure Therapy
- Ergometer Arm Exerciser Rehabilitation Physiotherapy Exerciser
- Digital Remote Controlled DC Motor Based Electrical Tilting Table
- Dual Probe Laser Red Visible Pointed Probe And Cluster Probe
- Hi Low Height Adjustable Traction Table
- Manual Mechanical Heavy Duty Physiotherapy Inversion Table
- Shockwave Therapy Clinical Touch Screen 7Heads Foot Switch
- Balance Training Unweighed Device Dual Motor Stroke Rehabilitation
- Unweighting System for Mobility Advance Partial Body Weight Support System
- Resistance Band Twisting Waist Disc Sport Balance Trainer
- Imported Motorised Pedo Cycle Physiotherapy and Rehabilitation Equipment
- Pedo Cycle Electrical Model With Legs Support for Stroke Paralysis Patinets
- Motorized Leg Exercise Machine Electrical Pedal Exerciser for Legs
- Motorized Physiotherapy Pedal Machine for Effective Rehabilitation with Leg Support
- Cutting-Edge Motorized Leg Exercise Machine: Stroke Recovery Digital Rehab Exerciser
- Digital Stroke Recovery System Automated Rehabilitation Bike Digital Pedo Cycle
- Electric mini pedal exerciser for rehabilitation Electric mini pedal exerciser for rehabilitation
- Electric pedal exerciser for physical therapy at home electric pedal exerciser
- Motorized exercise cycle for elderly therapy
- Pedo Cycle Electric pedal exerciser for rehabilitation exercises
- Pedo Cycle Adjustable motorized pedal exerciser for rehab
- Electric pedal cycle for upper and lower body exercises
- Pedo Cycle Innovative Motorized Electrical Pedal Exerciser for Home Use
- Physiotherapy pedal machine Home physiotherapy equipment Pedal exerciser for seniors
- Compact DVT and lymphedema treatment Portable lymphedema relief device
- Lymphedema Dvt Machine Lymphatic Drainage Pump
- Portable Body Pain Reduce Low level Cold Laser Therapy Handy Physiotherapy
- Arthritis Pain Relief Handheld Laser for Effective Therapy Physiotherapy LASER
- Handheld physiotherapy LASER Compact LASER device
- Laser Therapy Dual Probe Pointed and Cluster 750 mw Laser Therapy Equipment PHYSIO THERAPY MACHINE
- Low Level Laser Therapy Cold and Hot Laser Dual Probe 750 Mw 910nm Biotronix Physiotherapy Laser
- Dual probe laser therapy system Advanced pain relief laser device
- LASER Therapy Equipment Physiotherapy Pain Relief Digital LCD Based Pre Programmed
- dual probe laser for pain relief laser pain relief device 910nm
- LCD Display Pre Progrmmed Dual Probe Laser Pain Relief Device 910nm with 2 Year Warranty
- Hand Grip Dynamometer Digital Grip Strength Measurement
- Hand Grip Strength Analyzer
- Handgrip strength meter digital Digital hand grip dynamometer
- Hand grip strength digital Digital dynamometer hand grip
- dynamometer Hand grip dynamometer digital tester
- Hand grip strength digital Digital dynamometer hand grip
- Hand grip strength digital Digital dynamometer hand grip
- Digital handgrip force measurement device
- Hand grip strength dynamometer digital analyzer
- Self Adhesive Electrode Pads for EMS, TENS, IFT Electrotherapy Device ((Pin Size 1.5mm)(pack of 4
- muscle stimulator pads physiotherapy pads conductive electrode pads
- muscle stimulator pads physiotherapy pads conductive electrode pads
- Bubble Inclinometer Professional ROM Assessment Tool for Accurate Joint Mobility Analysis
- Physiotherapy bubble inclinometer Therapy
- bubble Professional physiotherapy inclinometer
- Inclinometer for therapists Precision bubble inclinometer
- Joint Motion Measuring Tool, Easy Rotation Accurate Measurement Scoliosis Torsional Angle Meter
- Plastic 0-360 Degree Bubble Inclinometer Physiotherapy, Size: Portable, Packaging Type: Box
- Physical Bubble Inclinometer Baseline Physical Therapy Inclinometer with 360 Rotating Dial
- Extracorporeal Pneumatic Shockwave Therapy Machine For Pain Relief
- Biotronix Pneumatic Shockwave Therapy Machine ED Treatment Pain Relief Extracorporeal ESWT
- Biotronix Impulse wave Therapy Shockwave Machine Muscle Pain Relief Massage ED Treatment
- Biotronix 8 Bar Step by 0.5bar Shock Wave Shockwave Joints Pain Physiotherapy and ED Treatment
- Biotronix Clinical ESWT Device Cutting-Edge Technology for Accelerated Muscle Recovery
- shockwave therapy for keloid scars blood flow enhancement
- Radial shockwave therapy for fat metabolism Shockwave therapy for hormonal balance
- Radial shockwave therapy for Achilles tendonitis relief
- Shockwave Machine Professional Clinical Touch Screen for Physiotherapy & ED
- Electric Knee Massage Machine, Cordless LED Touch Screen, 3 Modes, 3 Gears Temperature Adjustment,
- Knee Massager Arthritis Pain Arthritis Physical Therapy Knee Massager Equipment For Joint Pain
- Knee Massager Arthritis Pain Arthritis Physical Therapy Knee Massager Equipment For Joint Pain
- Electric knee heating pad massager Knee massager for cartilage damage
- Physiotherapy electrotherapy Combi 2 in one UST Ultrasound 1 Mhz and TENS 4 channel Digital
- CPM Machine Ankle
- Wrist CPM Machine
- Wrist CPM Machine for Mobility Recovery
- Wrist CPM Machine | Continuous Passive Motion Device for Rehabilitation & Mobility Recovery
- Adjustable Wrist CPM Therapy Machine | Post-Surgery Recovery & Physiotherapy Equipment
- Medical Wrist CPM Device | Hand & Wrist Rehabilitation Machine for Mobility Improvement
- Medical Wrist CPM Device | Hand & Wrist Rehabilitation Machine for Mobility Improvement
- Wrist CPM Machine for Post-Surgery Recovery | Continuous Passive Motion Therapy Device
- Professional Wrist Rehabilitation CPM Machine | Adjustable Speed & Motion Range for Therapy
- Wrist CPM Physiotherapy Machine | Orthopedic Recovery & Joint Mobility Device
- Wrist CPM Machine for Rehabilitation | Continuous Passive Motion Device for Wrist Therapy, Arthritis, and Post-Surgery Recovery
- Compact Wrist CPM Machine for Physical Therapy | Medical Wrist Rehabilitation Device for Home & Clinic Use
- Advanced Wrist CPM Therapy Machine | Orthopedic Rehabilitation Device for Mobility Enhancement & Pain Relief
- Best Wrist CPM Machine for Pain Relief | Post-Surgery, Stroke Recovery, and Arthritis Therapy Equipment
- Wrist CPM Machine for Home & Clinic Use | Continuous Passive Motion Therapy for Wrist Flexibility & Joint Recovery
- Adjustable Wrist CPM Physiotherapy Machine | Arthritis, Stroke & Post-Surgery Wrist Rehabilitation Device
- Wrist CPM Machine for Faster Recovery | Physiotherapy & Rehab Device for Wrist Injury & Mobility Enhancement
- Wrist CPM Machine for Joint Recovery | Smart Rehabilitation Device for Arthritis, Tendon Repair & Post-Surgery Care
- Robotic Hand Rehabilitation Glove
- Robotic Rehab Glove Hand Recovery & Physiotherapy
- Robotic Hand Therapy Glove Stroke, Arthritis & Injury Recovery
- Smart Rehab Glove Affordable Hand & Finger Therapy
- Robotic Hand Recovery Glove Cost-Effective Therapy
- Economical Stroke Therapy Glove Hand & Finger Rehabilitation Device
- Robotic Glove for Hand Therapy
- Smart Hand Therapy Glove Affordable Robotic
- Robotic gloves economical
- E12 Robotic Hand Therapy Glove Stroke Recovery & Rehabilitation Device
- E12 Smart Robotic Glove Affordable Hand & Finger Therapy for Recovery
- E12 Stroke Recovery Glove Neurological Hand Therapy & Physiotherapy
- E12 Robotic Hand Rehabilitation Glove Portable Physiotherapy Device
- Smart Robotic Glove E12 Hand & Finger Mobility Recovery Solution
- Robotic Hand Rehabilitation Glove E12 Finger Strength & Mobility Trainer
- E12 Smart Recovery Glove Motorized Hand Therapy for Stroke Patients
- E12 Affordable Robotic Hand Therapy Glove Home & Clinical Rehab
- Ankle CPM Machine Continuous Passive Motion Device for Rehabilitation & Physiotherapy
- Digital Ankle CPM Therapy Machine for Post-Surgery Recovery & Mobility Improvement
- Adjustable Ankle CPM Machine for Foot & Joint Rehabilitation Therapy
- Medical Grade Ankle CPM Machine for Post-Operative Therapy & Pain Relief
- Ankle Joint CPM Machine for Sprain, Injury & Surgery Recovery
- Portable Ankle CPM Rehabilitation Equipment with Digital LCD Display
- Automatic Ankle CPM Therapy Machine for Home & Clinical Use
- Professional Ankle CPM Machine for Hospitals & Physiotherapy Centers
- Ankle CPM Rehabilitation Device for Continuous Passive Motion Therapy
- Ankle CPM Machine for Continuous Passive Motion Therapy & Joint Recovery
- Professional Pneumatic Shockwave Therapy Machine Black Edition for Pain Relief and Physiotherapy
- High-Performance Black Pneumatic Shockwave Therapy Device for Muscle Recovery & Sports Injuries
- Portable Pneumatic Shockwave Therapy Machine Black Model for Pain Management & Rehabilitation
- Advanced Pneumatic Shockwave Therapy Equipment Black Edition for Clinic & Home Use
- Black Digital Pneumatic Shockwave Therapy Device for Physiotherapy & ED Treatment
- Medical-Grade Black Pneumatic Shockwave Machine for Joint Pain & Muscle Relaxation
- 10 Bar High Power Pneumatic Shockwave Therapy Machine Black for Sports Recovery
- New Arrival Black Pneumatic Shockwave Therapy Device for Beauty & Rehabilitation
- Premium Black Shockwave Therapy Machine for Physiotherapists & Chiropractors
- Adjustable Intensity Pneumatic Shockwave Therapy Device Black for Professional Use
- Black Pneumatic Shockwave Therapy Machine for Pain Relief, Muscle Recovery, and ED Treatment
- Pneumatic Shockwave Therapy Black Machine for Professional Physiotherapy
- Medical-Grade Pneumatic Shockwave Therapy Device Black for Joint Pain, Tendonitis, and Rehabilitation
- Portable Digital Pneumatic Shockwave Therapy Machine Black for Chiropractors and Pain Clinics
- Adjustable Pressure Black Pneumatic Shockwave Machine for Deep Tissue Massage and Pain Management
- Adjustable Pressure Black Pneumatic Shockwave Machine for Deep Tissue Massage and Pain Management
- Advanced Pneumatic Shockwave Therapy Machine Black for Tendon Healing and Muscle Stimulation
- High-Energy Black Pneumatic Shockwave Therapy Machine for Medical, Home, and Aesthetic Use
- Latest Black Pneumatic Shockwave Therapy Equipment for Clinics, Hospitals, and Wellness Centers
- Cryo Cold Compression Therapy Machine for Pain Relief & Injury Recovery Cold Compression Therapy Device for Post-Surgery & Sports Injury Rehabilita
- Cold & Compression Therapy Machine for Swelling Reduction & Fast Healing
- Adjustable Cryo Cold Therapy Wrap for Knee, Ankle, Shoulder, and Elbow
- Physio Cryotherapy Compression Machine for Athletes & Physiotherapy Clinics
- Cryotherapy Compression Machine for Athletes & Physiotherapy Clinics
- Professional Cryo Compression Recovery Unit for Physical Therapy & Sports Medicine
- Ice Therapy Machine with Compression for Joint Pain & Inflammation Relief
- Rechargeable Cryo Cold Compression Wrap for Home & Clinic Use
- Medical-Grade Cryo Cold Compression Therapy Machine for Pain Relief
- Cold Compression Therapy Device for Post-Injury & Post-Surgery Recovery
- Portable Cryotherapy Ice Compression Wrap for Swelling & Pain Management
- Cryo Cold Therapy System for Muscle Recovery & Joint Rehabilitation
- Professional Cryo Cold Therapy Machine for Physiotherapy & Rehabilitation Centers
- Rechargeable Cold Compression Wrap for Knee, Shoulder, and Ankle Pain Relief
- Ice Therapy Compression System for Sports Injuries & Muscle Recovery
- Advanced Cryo Compression Technology for Inflammation & Joint Pain Relief
- Cold Compression Therapy Equipment for Home, Gym, and Clinic Use
- Digital Cryo Cold Compression Machine with Adjustable Temperature & Pressure
- Professional TENS EMS IF RUSS Physiotherapy Device | Multi-Function Electrotherapy Machine
- Portable TENS EMS IF RUSS 4 Channel Electrotherapy Combo Unit for Clinic and Home Use
- Multi Therapy Combo Device TENS EMS Interferential RUSS Stimulation for Rehabilitation
- Digital Electrotherapy Machine TENS EMS IF RUSS Combo for Pain Management and Recovery
- Compression Air DVT Intermittent Pneumatic 6 Chamber 2Legs,1Arm,1Waist Cuff
- DVT 4 Ch Digital Pump with 2 Legs
- DVT Compression Therapy Unit Air Pressure Massager For Pain Relief Of Leg, Calf And Foot Massager Machine
- Pelvifine Kegel probe Kegel Toner Loop Adaptor for Prolapse for Men and Women
- rectal probe pr-06a for pelvic floor rehabilitation - compatible with EMS electrostimulators universal connection with 2 mm plug MEDITALIASERVICE
- Vaginal Probe for Pelvic Exerciser EMS Device
- High Precision Bubble Inclinometer for Physiotherapy and Range of Motion Measurement
- Medical Manual Bubble Inclinometer for Joint and Spine ROM Assessment Tool
- Physiotherapy ROM Measurement Tool Bubble Inclinometer for Joint Mobility Testing
- Handheld Chiropractic Adjustment Tool Manual
- Manual Four-Head Chiropractic Adjusting Tool for Deep Tissue Back and Spine Therapy
- Ergonomic Manual Chiropractic Adjustment Device with Four Interchangeable Heads
- Professional Manual Chiropractic Tool 4-Head Deep Tissue Back Massager
- Non-Electric Manual Chiropractic Adjusting Instrument with Four Heads for Muscle Relief
- Class 4 Laser Smart S10A for Post-Surgery Healing and Accelerated Recovery of Soft Tissue Damage
- Tabletop Professional Medical-Grade Diode Laser Therapy Equipment for Clinics, Hospital
- Professional Smart S10A Class IV Laser Therapy Device for Physical Therapy & Sports Injury Recovery
- 750N Electric Spine Chiropractor Electric Impulse Gun Actuator Massager
- Adjustable Cryo Compression Therapy System with Ice Wrap for Inflammation Reduction
- Medical Grade Cold and Compression Therapy Unit for Rehabilitation Clinics
- Adjustable Cryo Compression Therapy System with Ice Wrap for Inflammation Reduction
- Inflatable Cryo Compression Wrap with Cold Therapy Pad for Swelling and Pain
- Tecar Therapy Machine
- 4T Physiotherapy Machine, EMT2 Magnet Therapy Machine
- Dual Wavelength 980nm + 1470nm Diode Laser Machine for Piles Fistula Fissure Vascular Treatment
- Professional PEST Loop Physiotherapy Equipment for Hospital and Clinic Use
- 2 in 1 pmst physio magneto laser physiotherapy equipment EMS21
- 7 Head ESWT Machine-Pneumatic Radial Shockwave Therapy with Adjustable Energy
- Non-Invasive Pneumatic Shockwave Clinical Radial ESWT Device 7 Heads 10 Modes
- Advanced Pneumatic Shockwave Radial ESWT 7 Head System
- Pneumatic Shockwave Therapy Device 7 Heads 10 Modes
- Pneumatic Shockwave Radial ESWT Device Adjustable Frequency
- 7 Head Pneumatic Shockwave ESWT Machine for Clinics
- Multi-Mode Pneumatic Shockwave Clinical Radial ESWT
- Pneumatic Shockwave Radial Therapy Device 7 Heads
- Professional 7 Head Pneumatic Shockwave ESWT System
- 7 Head Pneumatic Shockwave Radial ESWT Machin
- Pneumatic Shockwave Radial ESWT System 10 Modes
- Adjustable Frequency Pneumatic Shockwave ESWT Device
- High-Performance Pneumatic Shockwave Therapy System
- Electromagnetic PEMT Loop Therapy Machine 850W
- PEMT Loop Therapy with Deep Oscillation & Pulse
- 18cm Penetration PEMT Loop Therapy Device
- PEMT Loop Therapy for Sports & Rehab
- PEMT Loop Therapy Professional Electromagnetic Machine
- PEMT Loop Therapy Device for Pain Relief
- High Power 850W PEMT Loop Therapy Machine
- PEMT Loop Therapy-Advanced EM Therapy Machine
- Advanced Pulsed Electromagnetic Therapy (PEMT) Device for Pain Relief & Rehabilitation
- 850W Electromagnetic Loop Therapy Device with 18cm Penetration Depth & 6000 Gauss Intensity
- PEMT Magnet Therapy Machine for Sports Injury, Chronic Pain & Physiotherapy Clinics
- Pulse Electromagnetic Loop Therapy Device with Multiple Frequency Modes (28 Hz + MF)
- Heavy-Duty 28Kg PEMT Loop Therapy Equipment with 15A Fuse Safety & 4500Hz Oscillation
- Portable PEMT Loop Therapy Machine for Hospitals, Clinics & Rehabilitation Centers
- Deep Tissue Electromagnetic PEMT Therapy Device for Muscle Healing & Joint Recovery
- Pulse Electromagnetic Loop Therapy Machine for Deep Tissue Rehabilitation
- PEMT Loop Therapy Device with Multi-Frequency Modes & High Magnetic Intensity
- Professional 850W Electromagnetic Therapy Machine for Physiotherapy Clinics
- Magnet Therapy PEMT Loop Device with Adjustable Power & Ring Handles
- Clinic-Grade PEMT Therapy Machine with 18cm Penetration Depth & Safety Fuse
- Heavy-Duty PEMT Loop Therapy Machine with 4500Hz Oscillation Technology
- 850W PEMT Loop Therapy Equipment-Advanced Rehabilitation & Pain Relief
- High-Power 850W PEMT Loop Therapy Machine-Electromagnetic & Deep Oscillation Therapy
- Electromagnetic Deep Oscillation Therapy Equipment for Physiotherapy Clinics
- Dual Handle PEMT Loop Device Professional Physiotherapy & Sports Rehab Equipment
- 850W PEMT Loop Therapy Machine-Electromagnetic Pulse & Deep Tissue Oscillation
- Professional Magnetic Therapy Device for Physiotherapy & Muscle Rehabilitation
- High-Frequency PEMT Loop Equipment with Multi-Pulse Settings for Pain Relief
- Advanced PEMT Loop Therapy System-Deep Penetration 18cm & Adjustable Magnetic Intensity
- Dual Handle PEMT Loop Machine for Targeted Muscle & Joint Therapy
- Multi-Frequency PEMT Loop Machine for Pain Management & Sports Rehab
- Advanced Electromagnetic Therapy System for Deep Muscle Stimulation
- PEMT Loop Therapy Equipment-Clinical-Grade Magnetic & Pulse Therapy Device
- High-Intensity PEMT Loop Therapy Machine-Effective Joint & Muscle Healing
- Professional PEMT Loop Therapy Machine for Pain Relief & Muscle Recovery
- 850W High-Power PEMT Loop Device with Deep Electromagnetic Therapy
- Deep Penetration 18cm PEMT Loop Therapy Machine for Physiotherapy Clinics
- PEMT Loop Machine with Adjustable Magnetic Intensity 1000-6000 Gauss
- Dual Handle PEMT Therapy Equipment for Targeted Muscle & Joint Treatment
- High-Frequency 4500Hz PEMT Loop Therapy Device for Professional Use
- Clinical-Grade PEMT Loop Therapy Machine-Deep Tissue & Pain Relief
- Professional PEMT Therapy Equipment with Large Single & Small Double Handles
- 850W Muscle Recovery & Physiotherapy PEMT Loop Machine
- Electromagnetic Deep Oscillation Therapy Device for Pain & Inflammation
- Multi-Frequency PEMT Loop Device-Effective Joint & Muscle Stimulation
- Deep Tissue PEMT Loop Therapy Equipment with Adjustable Pulse Settings
- Professional Magnetic & Electromagnetic PEMT Therapy Machine
- Clinical PEMT Loop Device for Musculoskeletal Pain & Sports Recovery
- Robust 28kg PEMT Loop Therapy Machine with Fused 15 Amp Safety
- Electromagnetic Muscle Therapy Device for Physiotherapy & Sports Rehab
- High-Power PEMT Loop Therapy Equipment-Pain Relief & Rehabilitation Solution
- High-Power 850W PEMT Loop Therapy Device-Electromagnetic Pulse & Deep Oscillation Therapy
- Professional PEMT Loop Machine for Muscle Recovery, Pain Relief & Sports Rehabilitation
- Advanced PEMT Loop Therapy Device-Deep Penetration & Adjustable Magnetic Pulse for Physiotherapy
- PEMT Loop Therapy Machine-850W Electromagnetic Pulse Therapy for Pain Relief & Physiotherapy
- High Power PEMT Loop Device 1000-6000 Gauss with Large & Small Ring Handles
- Pulse Electromagnetic Therapy Machine-Deep Oscillation Magnet Therapy Equipment
- Clinical Grade PEMT Therapy Device-4500Hz Oscillation, 850W Power, 28Kg Model
- Electromagnetic PEMT Loop Therapy Machine for Muscle Recovery & Rehabilitation
- PEMT Loop Machine for Physiotherapy Clinics-Pain Management & Sports Injury Recovery
- Pulse Electromagnetic Field Therapy Equipment with Double Ring Handle-850W
- PEMT Loop Therapy Device with 18cm Deep Penetration & High Intensity 6000 Gauss
- 850W PEMT Looiefp Therapy Machine-Electromagnetic Pulse & Magnet Therapy for Pain Relief
- PEMT Magnet Therapy Device-28Kg Heavy Duty, 15 Amp Fuse, Clinical Rehabilitation Use
- Professional PEMT Magnet Therapy Device-Deep Oscillation, 18cm Penetration, 850W Power
- Digital Laser Therapy Equipment with USB Type-C, Adjustable Timer, and Stand Holder
- Dual-Wavelength Laser Therapy Device for Arthritis, Muscle Recovery & Rehabilitation
- Home & Clinic Use Laser Therapy Machine with Safety Goggles and Rechargeable Battery
- Dual-Wavelength Laser Therapy Device with Digital Display & Rechargeable Battery
- Portable 670mW Laser Physiotherapy Machine with Cooling Fan & USB-C Charging
- Red+Infrared Light Therapy Device 650nm & 808nm with Adjustable Timer
- Home & Clinic Use Laser Therapy Device with Long-Lasting Battery & Cooling Fan
- Rechargeable Laser Therapy Machine with Cooling Fan, Digital Display & Adjustable Holding Stand
- Digital Laser Therapy Equipment with 4 Infrared & 14 Red Diodes-670mW Power Output
- Rechargeable Portable Laser Therapy Device with Cooling Fan, 30-Min Timer & Adjustable Stand
- Physiotherapy Laser Device with Adjustable Stand, Cooling System & Dual Wavelength Output
- Portable Dual-Wavelength Laser Therapy Device for Arthritis, Joint Pain & Muscle Recovery
- Rechargeable Handy Laser Therapy Device with Dual Wavelength & Cooling Fan
- Handheld Infrared & Red Light Laser Therapy Device with Digital Timer
- Dual-Diode Laser Therapy Equipment 808nm+650nm for Joint & Nerve Pain
- Laser Therapy Device with Adjustable Holding Stand & Cooling System
- Rechargeable Red & Infrared Laser Therapy Device for Home & Clinic Use
- Professional Laser Therapy Machine with Digital Display & USB Type-C Port
- Laser Pain Relief Device with Cooling Fan & 30-Min Adjustable Timer
- Compact Dual Wavelength Laser Therapy Device for Physiotherapy & Rehab
- Handheld Rechargeable Laser Therapy Equipment with Adjustable Stand
- Dual Wavelength Laser Physiotherapy Device for Deep Tissue Treatment
- Digital Laser Therapy Device with Fan Cooling and Long Battery Backup
- Portable Red Light Therapy Machine for Pain, Arthritis & Muscle Stiffness
- Rechargeable Infrared Laser Therapy Device with Display & Cooling Fan
- Physiotherapy Laser Therapy Device 808nm+650nm with Safety Goggle
- Multi-Diode Laser Therapy Machine with 18 Laser Tubes & USB-C Charging
- Digital Handy Laser Therapy Device for Back Pain & Muscle Recovery
- Red and Infrared Dual Laser Device for Pain Management & Healing
- Compact Physiotherapy Laser Machine with Timer and Cooling Function
- Rechargeable Laser Therapy Device with Stand, Fan Cooling & Digital Display
- Portable Laser Therapy Device for Pain Relief & Inflammation
- Dual Light Laser Physiotherapy Device 808nm + 650nm Rechargeable
- Professional-Grade Portable Laser Therapy Device-Ideal for Home & Clinic Use
- Portable Infrared Red Light Laser Therapy Equipment with Adjustable Holding Stand
- High-Power 10W Diode Laser Therapy Device with Touch Screen
- Precision Laser Therapy 980nm Class 4 Laser with Multiple Lens Options
- Professional Diode Laser for Rehabilitation & Pain Management-Class 4 Physiotherapy Laser
- Compact Class 4 Laser Therapy Machine-10W, Air-Cooled, Touch Control
- 980nm Diode Laser for Targeted Physiotherapy Treatments-Class 4 Laser for Pain Relief & Rehabilitation
- Portable 10W Diode Laser Therapy with CW/Pulse Modes-Class 4 Physiotherapy Laser
- Medical-Grade Laser Therapy Device with 10mm & 20mm Lens Options-Class 4 Physiotherapy Laser
- Efficient 980nm Laser for Physiotherapy Clinics & Rehab Centers-Class 4 Diode Laser for Pain Relief & Rehabilitation
- Advanced 980nm Class 4 Diode Laser Therapy Device-10W for Physiotherapy & Pain Relief
- Advanced Class 4 Diode Laser Therapy Device 980nm 10W for Professional Pain Relief and Physiotherapy Treatments
- Professional Diode Laser Therapy Machine Designed for Deep Tissue Healing and Fast Pain Relief in Rehabilitation Centers
- Medical Grade Diode Laser Therapy Device with Continuous and Pulse Modes for Effective Muscle Recovery and Joint Pain Relief
- Class IV High-Power Laser Therapy System 980nm 10W with 10mm and 20mm Lens Tubes for Versatile Physiotherapy Applications
- Digital Touch Screen Controlled Class 4 Diode Laser Therapy Machine for Pain Reduction, Sports Injury Recovery, and Inflammation Relief
- Advanced Physiotherapy 980nm Class 4 Diode Laser Equipment for Fast Healing, Deep Tissue Regeneration, and Nerve Stimulation
- Compact and Powerful 10W 980nm Diode Laser Therapy Device with Air Cooling for Professional Clinics and Rehabilitation Centers
- Smart Class IV Laser Therapy Unit with Adjustable Power, Multiple Lens Options, and Safety Emergency Switch for Physiotherapy Clinics
- Premium Class 4 Diode Laser Therapy Device 980nm 10W for Pain Management, Muscle Recovery, and Physiotherapy Treatments
- Advanced Medical Laser Therapy Equipment Class IV 980nm Diode Type with Touch Screen and Air Cooling System
- Class IV Diode Laser Physiotherapy Equipment 980nm
- 980nm High-Intensity Class 4 Laser Therapy Device for Pain Management
- Medical Grade Class IV Laser Therapy Equipment 10W 980nm for Clinical Physiotherapy and Deep Tissue Treatment
- Class 4 Diode Laser Therapy Machine 980nm 10W For Physiotherapy And Rehabilitation
- High Power 980nm Class 4 Diode Laser Therapy Equipment For Pain Relief Treatment
- 10W Class 4 Laser Therapy Device 980nm For Physiotherapy And Sports Injury Recovery
- Medical Grade Class IV Diode Laser Therapy System 980nm 10W For Pain Management
- Portable 10W Class 4 Laser Therapy Equipment 980nm For Clinic Use
- Physiotherapy Class 4 Laser Device 980nm 10W With 10mm Treatment Handle
- Professional 980nm Class 4 Laser Therapy System For Rehabilitation And Pain Relief
- Class 4 980nm 10W Diode Laser Therapy Equipment For Pain Management And Healing
- Portable 980nm Class 4 Laser Therapy Device For Physiotherapy Clinics
- Class 4 Diode Laser Physiotherapy Equipment 980nm For Sports Injury Recovery
- Medical Physiotherapy Class 4 Laser Therapy Unit 10W 980nm With Touch Screen
- Advanced Class 4 10W Laser Therapy Machine For Physiotherapy Applications
- 980nm Diode Laser Therapy Equipment Class 4 Type For Professional Use
- 10W Diode Laser Therapy Device for Pain Relief & Rehab
- Portable 980nm Laser Therapy Machine with Touch Screen
- CW/Pulse Mode Diode Laser 10W for Muscle & Joint Pain
- Professional Diode Laser Machine for Physiotherapy Clinics
- 980nm Laser Pain Relief Machine with Emergency Stop Switch
- Compact 7.5kg 10W Diode Laser Therapy Equipment
- Multi-Spot Diode Laser Machine for Physiotherapy-Pain Relief & Rehabilitation
- Spherical Lens Laser Therapy Device (10mm & 20mm)-Advanced Diode Laser Machine for Physiotherapy & Pain Relief
- High-Power 980nm Laser Therapy Machine for Clinic Use
- Advanced 10W Diode Laser for Muscle & Joint Rehabilitation
- Portable Diode Laser Therapy Machine for Sports Clinics
- CW/Pulse 980nm Laser Therapy Equipment for Fast Recovery
- Professional Laser Therapy Machine with Lens Tube Options
- Professional Laser Therapy Machine with Lens Tube Options 980nm Diode Laser Physiotherapy Device
- 980nm Diode Laser Device for Pain Relief & Muscle Recovery Portable Laser Therapy Machine
- Compact Physiotherapy Laser Machine with Emergency Switch Portable Diode Laser Therapy Equipment
- High-Efficiency Class 4 Laser Device 10W for Clinics Professional Physiotherapy Laser Therapy Equipment
- Multi-Spot Laser Therapy Equipment for Joint Pain Relief Advanced Physiotherapy Laser Machine
- 10W Portable Diode Laser Machine for Rehab Centers Advanced Physiotherapy Laser Therapy Device
- Advanced Laser Therapy Device 980nm for Muscle Therapy Professional Diode Laser Physiotherapy Equipment
- Professional Physiotherapy Diode Laser Machine 10W High Power Laser Therapy Device for Pain Relief & Rehabilitation
- CW/Pulse Mode Diode Laser Therapy Device for Physiotherapy
- Class 4 Diode Laser Therapy Machine 980nm 10W for Pain Relief and Physiotherapy
- High Power Diode Laser Therapy Equipment 980nm 10W for Physiotherapy Clinics
- Class 4 Medical Laser Therapy Machine 980nm Diode Laser for Pain and Muscle Recovery
- Physiotherapy Laser Therapy Device Class 4 Diode 10W 980nm with Touch Screen
- 10W 980nm Diode Laser Therapy System for Physiotherapy, Pain Relief, and Healing
- Professional Class 4 Diode Laser Therapy Equipment 980nm for Rehabilitation Centers
- Class 4 Laser Pain Relief Device 980nm 10W with Air Cooling and Touch Display
- 980nm 10W Class IV Laser Therapy Machine for Pain, Inflammation, and Recovery
- Portable 980nm Diode Laser Therapy Device 10W Power for Clinical Physiotherapy Use
- High-Intensity Class 4 Laser Therapy Device 980nm 10W for Deep Tissue Pain Relief
- Portable 980nm Diode Laser Therapy Machine Class 4 for Physiotherapy and Recovery
- Medical Grade Class 4 Diode Laser 980nm 10W for Pain Management Treatment
- Class IV 980nm Diode Laser Physiotherapy Equipment with Touch Screen Control
- Advanced Class 4 Laser Therapy System for Pain Relief, Healing & Inflammation Reduction
- 980nm Diode Laser Therapy Equipment-10W Power, Air Cooling, Digital Interface
- Class 4 Diode Laser Therapy System-980nm, 10W, Professional Physiotherapy Device
- Pain Relief and Tissue Healing Diode Laser Therapy Device 980nm Class 4
- 10W 980nm Diode Laser for Muscle Pain, Joint Therapy, and Post-Surgery Rehabilitation
- Clinical Laser Therapy Equipment Class 4 980nm for Pain and Inflammation Control
- High Power Class 4 Diode Laser Physiotherapy Machine 980nm with Emergency Switch
- Digital Laser Therapy Device 980nm Diode Class 4 for Professional Physiotherapists
- 980nm Laser Therapy Device with 10mm and 20mm Lens Tubes Class 4 Diode Model
- Therapeutic Class 4 Diode Laser 980nm 10W for Physiotherapy and Sports Medicine Use
- Physiotherapy Diode Laser Machine 980nm Class IV-Pain Relief and Rehabilitation Tool
- 980nm Class 4 Laser Therapy Device for Deep Tissue Repair and Pain Reduction
- Professional Pain Management Laser Therapy Equipment 10W Class 4 Diode 980nm
- Clinical Grade Class 4 Laser Therapy System 980nm 10W-Air-Cooled Design
- Digital 6 Chamber DVT Pump For Physiotherapy And Rehabilitation
- 6 Chamber Sequential Compression Therapy Device For DVT And Lymphedema
- DVT 6 Chamber Basic Model Air Compression Pump With Digital Display
- Advanced Biotronix DVT 6 Chamber Compression Therapy Device For Leg, Arm & Waist
- 6 Chamber DVT Pump Digital Touch Panel With 4 Modes And Timer
- Sequential Air Compression Therapy Unit 6 Chamber Digital Model
- Digital 6 Chamber DVT Compression Pump For Lymphedema Therapy
- Digital 6 Chamber DVT Machine For Air Compression Therapy And Recovery
- High Performance Biotronix 6 Chamber DVT Pump With Digital Pressure Display
- DVT 6 Chamber Compression Therapy Device For Improved Circulation
- Digital DVT Pump 6 Chamber Model With Touch Panel And Adjustable Pressure
- 6 Chamber Sequential Pneumatic Compression Device For Leg And Arm Therapy
- Advanced DVT 6 Chamber Pump With Digital Time & Pressure Display
- Pneumatic Compression Therapy Device 6 Chamber Digital Touch Model
- Professional 6 Chamber DVT Therapy Device For Physiotherapy Clinics
- 6 Chamber Digital Compression Pump With Multi-Mode Air Therapy
- Digital DVT 6 Chamber Therapy Unit With Leg, Arm & Waist Sleeves
- Digital DVT 6 Chamber Air Compression Therapy Device with Touch Panel
- Digital 6 Chamber Sequential Air Compression DVT Pump for Lymphedema and Recovery
- DVT 6 Chamber Digital Compression Therapy Device for Improved Blood Circulation
- Air Compression DVT Digital Pump 6 Chamber for Medical and Physiotherapy Applications
- Digital 6 Chamber DVT Air Compression Therapy Unit for Rehabilitation and Recovery
- 6 Chamber Digital Display DVT Compression Device with Timer and 4 Operating Modes
- Digital DVT Air Compression Machine 6 Chamber Model for Home and Hospital Use
- DVT 6 Chamber Digital Sequential Compression Pump for Lymphedema Therapy
- Digital DVT Pump 6 Chamber Touch Panel Model Operation
- 6 Chamber DVT Therapy Unit Digital Model for Rehabilitation and Circulatory Health
- DVT Air Compression Therapy Device Digital Display Type with Adjustable Pressure
- Digital DVT Sequential Therapy Machine 6 Chamber for Medical and Physio Centers
- 6 Chamber Digital Air Compression DVT Pump with Leg, Arm and Waist Attachments
- Digital 6 Chamber DVT Therapy Unit with Timer Options and Touch Control Display
- Digital DVT Pump 6 Chamber Basic Model for Physiotherapy and Circulatory Recovery
- 6 Chamber Digital DVT Machine with Touch Display, Remote and Timer Setting
- DVT Sequential Air Compression 6 Chamber Digital Pump for Rehabilitation Therapy
- Digital Display Air Compression DVT Pump 6 Chamber for Post-Surgery Care
- 6 Chamber DVT Digital Sequential Therapy Machine for Leg and Arm Recovery
- Digital DVT Pump 6 Chamber Device for Circulation, Lymphedema and Swelling Control
- 6 Chamber Air Compression DVT Therapy Digital Unit for Home and Clinical Use
- DVT 6 Chamber Digital Pump with Adjustable Timer, Pressure and Touch Panel Display
- Digital Air Compression DVT Therapy Pump 6 Chamber Model for Physio Clinics
- 6 Chamber Digital DVT Compression Pump for Recovery, Circulation and Therapy
- DVT Sequential Air Compression Digital 6 Chamber Pump for Rehabilitation and Care
- Digital DVT 6 Chamber Compression Therapy Device with Timer and Touch Control Panel
- Advanced Exoskeleton Robot for Gait Training and Paralysis Rehabilitation
- Motorized Robotic Exoskeleton for Stroke Rehabilitation and Mobility Therapy
- AI-Controlled Robotic Rehabilitation Device for Stroke and Spinal Cord Injury Recovery
- Physiotherapy Exoskeleton Robot for Neuro Rehabilitation and Mobility Training
- Full Body Robotic Exoskeleton for Rehabilitation, Stroke, and Paralysis Patients
- Rehabilitation Robotic Exoskeleton for Physical and Neurological Recovery
- Robotic Gait Training Exoskeleton for Physiotherapy and Rehab Centers
- Digital Motorized Exoskeleton Robot for Stroke Recovery and Mobility Assistance
- Robotic Exoskeleton Rehabilitation System for Stroke and Paralysis Patients
- Smart Robotic Exoskeleton for Physiotherapy and Neuro Rehabilitation Clinics
- AI-Based Exoskeleton Rehabilitation Device for Stroke Recovery Therapy
- Robotic Walking Exoskeleton for Mobility and Balance Training in Rehabilitation
- Motorized Exoskeleton Robot for Physiotherapy and Spinal Cord Injury Recovery
- Robotic Assistive Exoskeleton for Neuro and Physical Rehabilitation
- Exoskeleton Walking Robot for Stroke, Paralysis, and Injury Rehabilitation
- Intelligent Rehabilitation Exoskeleton Robot with Adjustable Support Levels
- Digital Robotic Exoskeleton for Limb Movement and Strength Training
- AI-Powered Exoskeleton Robot for Physiotherapy and Recovery Sessions
- Robotic Exoskeleton Mobility Trainer for Stroke and Neuro Patients
- Advanced Robotic Therapy Exoskeleton for Post-Stroke Rehabilitation
- Robotic Rehabilitation Exoskeleton Machine for Neurological Disorders
- Physiotherapy Exoskeleton Machine for Stroke and Paralysis Patients
- Next-Generation Exoskeleton Rehabilitation Robot for Clinical Therapy Use
- Full Body Robotic Exoskeleton for Neuro and Motor Recovery Applications
- Robotic Exoskeleton for Physical Rehabilitation and Stroke Therapy
- Exoskeleton Robot for Motor Function Recovery and Gait Training
- Advanced Exoskeleton Therapy Robot for Stroke and Spinal Injury Recovery
- Neuro Rehabilitation Exoskeleton Robot for Limb Mobility Training
- Motorized Exoskeleton Rehabilitation Robot for Hospital and Clinical Use
- AI-Driven Robotic Exoskeleton Device for Physiotherapy and Neuro Recovery
- Digital Exoskeleton Robot for Advanced Physiotherapy and Rehabilitation
- Robotic Rehabilitation Exoskeleton for Arm and Leg Therapy Exercises
- Smart Motorized Exoskeleton System for Stroke and Paralysis Patients
- Automatic Robotic Exoskeleton for Full Body Neuro Rehabilitation
- Exoskeleton Robotic Trainer for Physical and Neurological Recovery
- AI-Powered Rehabilitation Exoskeleton for Stroke and Spinal Cord Injury Therapy
- Robotic Exoskeleton Rehabilitation Trainer for Upper and Lower Limbs
- Rehabilitation Robotics Exoskeleton for Stroke and Motor Recovery
- Professional Focused Shockwave Therapy Machine for Deep Tissue Recovery
- Digital Focused Shockwave Therapy Device for Rehabilitation and Ortho Use
- High Precision Focused Shockwave Therapy Equipment for Pain Management
- Focused ESWT Therapy Machine for Tendon, Shoulder, and Plantar Fasciitis Treatment
- Portable Focused Shockwave Therapy Machine for Clinic and Hospital Use
- Medical Focused Shockwave Therapy Device for Muscle and Joint Pain
- Focused Extracorporeal Shockwave Therapy Machine for Deep Tissue Healing
- Focused Shockwave Therapy System for Physiotherapy and Sports Injury Rehab
- Professional Focused Shockwave Therapy Equipment for Rehabilitation Centers
- Focused Shockwave Therapy Machine with Adjustable Energy and Frequency
- Imported Focused Shockwave Therapy Device for Orthopedic and Sports Medicine
- Advanced Focused Shockwave Therapy Equipment for Chronic Pain Treatment
- Focused Shockwave Therapy Machine for Back, Neck, and Shoulder Pain Relief
- Focused Shockwave Therapy Machine for Pain Relief and Faster Recovery
- Focused Shockwave Therapy Equipment for Hospitals and Rehabilitation Clinics
- Focused ESWT Shockwave Therapy Device for Muscle Stimulation and Healing
- Focused Shockwave Therapy MachineNon-Surgical Pain Relief Solution
- Focused Shockwave Therapy Device for Physiotherapy and Rehabilitation Clinics
- Professional Focused Shockwave Therapy Machine for Pain and Muscle Recovery
- Advanced Focused Shockwave Therapy Equipment for Deep Tissue Healing
- Focused ESWT Therapy Machine for Sports Injury and Tendon Pain Treatment
- High-Energy Focused Shockwave Therapy Device for Orthopedic Applications
- Focused Shockwave Therapy Machine for Shoulder, Knee, and Back Pain
- Medical Focused Shockwave Therapy Equipment for Muscle Regeneration
- Focused Shockwave Therapy Machine for Physiotherapy and Clinic Use
- Digital Focused Shockwave Therapy Device with Adjustable Energy Levels
- Portable Focused Shockwave Therapy Equipment for Home and Clinical Use
- Focused Shockwave Therapy Device for Plantar Fasciitis and Tendonitis Relief
- Imported Focused Shockwave Therapy Machine for Professional Physiotherapists
- Imported Focused Shockwave Therapy Machine for Rehabilitation Centers
- Focused Shockwave Therapy Equipment for Deep Muscle Tissue Stimulation
- Focused Shockwave Therapy Machine for Hospitals and Physical Therapy Centers
- Focused Shockwave Therapy Device for Fast and Effective Pain Management
- Focused Shockwave Therapy Equipment for Back, Shoulder, and Leg Pain
- Premium Focused Shockwave Therapy Machine with Digital Display Control
- Focused Shockwave Therapy Machine for Physiotherapy and Pain Management
- High-Performance Focused Shockwave Therapy Machine for Orthopedic Clinics
- Portable Focused Shockwave Therapy Device for Sports Injury Treatment
- Focused Extracorporeal Shockwave Therapy Equipment for Physical Therapy
- Advanced Focused Shockwave Therapy Machine for Chronic Pain Relief
- Focused Shockwave Therapy Device for Tendonitis and Plantar Fasciitis Treatment
- Medical Focused Shockwave Therapy Equipment for Deep Tissue Stimulation
- Focused Shockwave Therapy System for Physiotherapy and Orthopedic Care
- Focused ESWT Shockwave Therapy Equipment for Fast Tissue Recovery
- Compact Focused Shockwave Therapy Device with Digital Display Panel
- High-Energy Focused Shockwave Therapy Device for Muscle Therapy
- Focused Shockwave Therapy Machine for Deep Muscle Pain and Injury Healing
- Physiotherapy Focused Shockwave Therapy Equipment for Medical Professionals
- Focused ESWT Therapy Device for Orthopedic and Sports Medicine Clinics
- Premium Focused Shockwave Therapy Machine for Effective Pain Relief
- Focused Shockwave Therapy Equipment for Deep Tissue Pain and Injury Recovery
- High-Energy Focused Shockwave Therapy Machine for Physiotherapy Clinics
- Professional Focused Shockwave Therapy Device for Orthopedic Rehabilitation
- Digital Focused Shockwave Therapy Equipment for Muscle and Tendon Treatment
- Advanced Focused Shockwave Therapy Machine for Back and Shoulder Pain Relief
- Focused Shockwave Therapy Device for Sports Injury and Muscle Therapy
- Portable Focused Shockwave Therapy Equipment for Clinic and Home Use
- Focused ESWT Shockwave Therapy Machine for Physiotherapy Applications
- Medical Focused Shockwave Therapy Device for Chronic Pain Management
- Compact Focused Shockwave Therapy Machine with Digital Display System
- Focused Extracorporeal Shockwave Therapy Equipment for Faster Recovery
- Imported Focused Shockwave Therapy Device for Orthopedic and Rehab Centers
- Focused Shockwave Therapy Equipment for Joint, Muscle, and Nerve Pain
- Professional ESWT Focused Shockwave Therapy Machine for Hospitals
- High-Precision Focused Shockwave Therapy Device for Physiotherapy Experts
- Focused Shockwave Therapy Machine for Deep Tissue Stimulation and Repair
- Focused Shockwave Therapy Equipment for Leg, Arm, and Back Pain Relief
- Portable Focused ESWT Machine for Muscle Regeneration and Pain Reduction
- Medical Focused Shockwave Therapy System for Rehabilitation and Wellness
- Focused Shockwave Therapy Machine for Muscle Pain and Injury Recovery
- High-Energy Focused Shockwave Therapy Device for Deep Tissue Stimulation
- Professional Focused Shockwave Therapy Machine for Orthopedic Treatment
- Portable Focused ESWT Therapy Device for Clinics and Hospitals
- Advanced Focused Shockwave Therapy Machine for Chronic Pain Management
- Focused Shockwave Therapy Device for Deep Tissue Healing and Pain Relief
- Compact Focused ESWT Therapy Device for Rehabilitation and Pain Therapy
- Focused Shockwave Therapy System for Non-Invasive Pain Treatment
- Focused Shockwave Therapy Equipment for Shoulder and Knee Pain Relief
- Imported Focused ESWT Therapy Device for Physiotherapy Applications
- Focused Shockwave Therapy Machine for Back and Leg Muscle Recovery
- Professional Focused Shockwave Therapy Device for Tendon and Nerve Pain
- Professional Focused Shockwave Therapy Device for Physiotherapy and Rehabilitation
- Advanced Focused Shockwave Therapy Equipment for Muscle and Joint Pain Relief
- Electromagnetic Focused Shockwave Therapy Unit for Deep Tissue Healing
- Focused Shockwave Therapy System for Sports Injury and Chronic Pain Recovery
- KM-580 Digital EMG Biofeedback Therapy Equipment for Physiotherapy Clinics
- Advanced Biofeedback Therapy Equipment KM-580 for EMG and ETS Training
- KM-580 Dual Channel EMG ETS FES NMES Physiotherapy Biofeedback Device
- KM-580 Touch Screen Biofeedback and Stimulation Unit with Rechargeable Battery
- KM-580 EMG ETS NMES FES Digital Physiotherapy Biofeedback Machine
- KM-580 Biofeedback and Electrical Stimulation Equipment for Pain Relief & Rehab
- KM-580 EMG Biofeedback System with PC Report Printing Feature
- Advanced Physiotherapy Biofeedback Device KM-580 with EMG & TENS Modes
- KM-580 EMG Biofeedback & Electrical Stimulation Device for Physiotherapy
- Advanced KM-580 Biofeedback Therapy Equipment for Rehabilitation Centers
- KM-580 Dual Channel EMG ETS FES NMES Biofeedback Therapy Machine
- KM-580 Professional Biofeedback Stimulator for Stroke and Muscle Recovery
- Digital Biofeedback Stimulator KM-580 with EMG and Multi-Mode Stimulation
- KM-580 EMG NMES FES TENS Pelvic and Swallowing Therapy Device
- KM-580 Touch Screen Biofeedback & Electrical Stimulation System
- Portable KM-580 Rechargeable Biofeedback Device with PC Connectivity
- KM-580 Multi-Channel EMG Biofeedback Therapy Unit for Clinics
- KM-580 Neuromuscular and Rehabilitation Biofeedback Stimulator
- KM-580 Comprehensive Biofeedback System for Physiotherapy Applications
- Professional Biofeedback & Stimulation Therapy Machine KM-580
- KM-580 Multi-Function EMG Biofeedback & TENS Pain Relief Device
- KM-580 Advanced EMG Biofeedback System for Neuro and Muscle Rehabilitation
- KM-580 Digital Biofeedback Therapy Machine with Dual EMG Channels
- KM-580 Physiotherapy Biofeedback & Electrical Stimulator with Touch Screen
- KM-580 Multi-Mode EMG Biofeedback Stimulator for Clinical Use
- KM-580 EMG ETS NMES FES Stimulator for Pelvic & Neuromuscular Therapy
- KM-580 Smart Biofeedback Stimulator with Rechargeable Battery & Report Printing
- KM-580 EMG Biofeedback & Electrotherapy System for Rehabilitation
- Advanced KM-580 Biofeedback Therapy Equipment for Rehabilitation Centers
- KM-580 Smart Biofeedback and Electrical Stimulation Unit for Muscle Recovery
- KM-580 Dual Channel EMG Biofeedback Machine with Touch Screen Display
- KM-580 Electrical Muscle Stimulation & Biofeedback Therapy Unit
- KM-580 Digital EMG Biofeedback System for Stroke and Paralysis Recovery
- KM-580 EMG ETS FES NMES TENS Biofeedback Therapy Machine for Clinics
- KM-580 Smart Biofeedback Therapy Equipment with PC Report Feature
- KM-580 Multi-Mode Biofeedback Device for Muscle Training and Pain Relief
- KM-580 EMG Biofeedback Device for Pain Management & Neuromuscular Therapy
- KM-580 EMG Biofeedback System for Neurological and Physiotherapy Rehab
- KM-580 Physiotherapy Biofeedback Machine with Dual EMG Channels
- KM-580 Professional Biofeedback and Electrical Stimulation Device
- KM-580 Advanced EMG Triggered Stimulation (ETS) Therapy Equipment
- KM-580 Multi-Channel Biofeedback & TENS Therapy Device for Physiotherapy
- KM-580 Digital Physiotherapy Biofeedback Machine with Rechargeable Battery
- KM-580 EMG Biofeedback and Stimulation Unit for Clinical Use
- KM-580 Smart Biofeedback & Neuromuscular Electrical Stimulator Device
- KM-580 Comprehensive EMG Biofeedback Therapy System for Rehabilitation Clinics
- KM-580 EMG Biofeedback Device with Games, Templates & Training Programs
- KM-580 Portable Biofeedback and Electrical Therapy Machine with Touch Display
- KM-580 EMG ETS Biofeedback System for Advanced Physiotherapy Treatment
- KM-580 Biofeedback Device for Neuromuscular and Pain Management Therapy
- KM-580 EMG Biofeedback Therapy Equipment with PC Software for Report Printing
- KM-580 EMG Biofeedback and Stimulation Device for Physiotherapy & Rehab
- KM-580 Dual Channel EMG Biofeedback Therapy Machine with LCD Touch Display
- KM-580 EMG NMES FES TENS Physiotherapy Biofeedback Therapy Device
- KM-580 Professional EMG Biofeedback System for Clinical Use
- KM-580 Multi-Mode EMG Biofeedback & Neuromuscular Electrical Stimulator
- KM-580 EMG ETS NMES FES Pelvic and Swallowing Therapy Machine
- KM-580 Biofeedback Stimulator with EMG, ETS, NMES, FES, TENS & Pelvic Therapy Functions Advanced Physiotherapy & Rehabilitation Device
- KM-580 EMG Biofeedback Therapy Machine for Physiotherapy & Rehabilitation Clinics
- KM-580 Multifunctional Biofeedback EMG Stimulator for Stroke and Pelvic Floor Therapy
- KM-580 Advanced EMG ETS NMES FES Pelvic & Swallowing Therapy Device
- KM-580 Dual Channel EMG Biofeedback and Four Channel Stimulation Therapy Unit
- KM-580 EMG Triggered Stimulation (ETS) Machine with Pain Relief and Muscle Training Modes
- KM-580 Physiotherapy Biofeedback Device with Touch Screen and PC Report Software
- KM-580 Clinical Biofeedback Stimulator for Neuromuscular Rehabilitation & Pain Management
- KM-580 Swallowing EMG & Pelvic Floor Biofeedback Therapy Equipment
- KM-580 Digital EMG ETS Biofeedback Physiotherapy Machine
- KM-580 Biofeedback Stimulator with EMG, ETS, NMES, FES, Pelvic & Swallowing Therapy Programs
- KM-580 Advanced EMG Biofeedback Physiotherapy Device with Dual EMG & Four Stim Channels
- KM-580 Touch Screen EMG Biofeedback Therapy Device with Custom Modes
- KM-580 Neuromuscular Electrical Stimulation Device with EMG Triggered Feedback
- KM-580 Clinical Biofeedback EMG Stimulator for Physical Therapy & Muscle Recovery
- KM-580 Dual Channel EMG and Four Channel STIM Biofeedback Therapy System
- KM-580 Physiotherapy Biofeedback Device for EMG Training, Pain & Muscle Rehab
- KM-580 Professional EMG Biofeedback Stimulator with PC Connectivity and Data Reports
- KM-580 Rechargeable EMG Biofeedback System for Stroke & Neurological Rehabilitation
- KM-580 Physiotherapy Biofeedback & Stimulation Device with Pain Relief Programs
- KM-580 EMG Triggered Stimulation Biofeedback System for Pelvic & Swallowing Therapy
- KM-580 All-in-One Physiotherapy Biofeedback Machine for EMG, ETS, NMES & FES
- KM-580 EMG Biofeedback Therapy Machine with Mirror Therapy and Games
- KM-580 EMG Biofeedback & Stimulation Unit for Clinical and Home Physiotherapy
- KM-580 Biofeedback EMG ETS NMES FES Therapy Device for Physiotherapy & Rehabilitation
- KM-580 EMG Biofeedback Stimulator for Pelvic Floor, Swallowing & Neuromuscular Therapy
- KM-580 Digital EMG Biofeedback System with PC Connectivity and Report Printing
- KM-580 Biofeedback Physiotherapy Equipment with Touch Display & Rechargeable Battery
- KM-580 EMG Triggered Stimulation Device for Pelvic, Swallowing & Sports Training
- KM-580 Dual Channel EMG with ETS, NMES, and FES for Professional Physiotherapists
- KM-580 EMG Triggered Biofeedback and Electrical Stimulation Device for Rehabilitation Therapy
- KM-580 Advanced EMG Biofeedback Physiotherapy Equipment with Touch Screen and PC Software
- KM-580 EMG Biofeedback and Neuromuscular Stimulation Device for Pelvic and Swallowing Training
- KM-580 EMG Triggered Biofeedback System for Stroke, Sports, Pain Relief and Muscle Re-Education
- Electrical Motorized Wrist CPM Machine for Wrist Joint Physiotherapy and Recovery
- Continuous Passive Motion Wrist CPM Machine for Post-Surgery Recovery
- Digital Wrist CPM Therapy Equipment with Remote and Adjustable Intensity Levels
- Digital Wrist CPM Therapy Equipment with Remote Control
- Automatic Wrist CPM Device for Wrist Flexion & Extension Therapy (090 Range)
- Wrist Joint CPM Machine-Premium Electrical Model with Remote Controlled Operation
- Advanced Wrist CPM Rehabilitation Device-Speed & Angle Adjustable (19 Levels)
- Remote Controlled Wrist CPM Equipment-Ideal for Stroke and Post-Trauma Therapy
- Wrist CPM Therapy Machine-Electrical Motorized Device for Wrist Rehabilitation
- Premium Wrist CPM Machine with Remote Control and Adjustable Range of Motion
- Wrist CPM Continuous Passive Motion Device for Hand and Wrist Therapy
- Wrist CPM Therapy Machine-Electrical Motorized Device for Wrist Joint Rehabilitation
- Premium Motorized Wrist CPM Unit with Adjustable Speed and Range of Motion
- Wrist Joint Continuous Passive Motion Machine for Physiotherapy and Recovery
- Electric Wrist CPM Device with Remote Control for Rehabilitation Centers
- Automatic Wrist CPM Machine for Joint Flexibility and Mobility Improvement
- Digital Wrist CPM Therapy Equipment with Adjustable Speed and Angle Settings
- Physiotherapy Wrist CPM Machine-Premium Electrical Model for Rehab Use
- Advanced Wrist CPM Therapy Unit-Smooth and Controlled Joint Movement
- Electrical Motorized Wrist CPM Machine with Dual Motion (Flexion/Dorsiflexion)
- Premium Wrist CPM Machine for Orthopedic and Neurological Rehabilitation
- Motorized CPM Device for Wrist Joint Exercise and Mobility Restoration
- Electric Wrist CPM Therapy Equipment for Hospitals and Physiotherapy Clinics
- Wrist Joint CPM Premium Model-Remote Controlled Rehabilitation Equipment
- Automatic Wrist Exercise CPM Machine-Adjustable Angle and Speed Control
- Therapy Device for Wrist Joint Recovery-Motorized Continuous Motion Machine
- Professional Wrist CPM Equipment-Ideal for Physiotherapy and Post-Surgery Care
- High-Performance Wrist CPM Machine for Clinical and Home Rehabilitation Use
- Wrist CPM Premium Model for Physiotherapy and Post-Operative Recovery
- Motorized Wrist CPM Device with Adjustable Speed and Motion Range
- Automatic Wrist CPM Machine-Ideal for Rehabilitation and Recovery Therapy
- Wrist Rehabilitation CPM Machine-Motorized Physiotherapy Equipment
- Electric Wrist CPM Therapy Device for Restoring Wrist Flexibility and Mobility
- Continuous Passive Motion Wrist Machine-Premium Model with Digital Controls
- Remote Controlled Wrist CPM Therapy Device-Adjustable Range and Speed
- Wrist Physiotherapy CPM Machine with 90 Palmarflexion and Dorsiflexion
- Electrical Wrist CPM Device-Compact, Portable, and Easy to Operate
- Rehabilitation CPM Machine for Wrist Joint Recovery and Flexibility Training
- Digital Wrist CPM Physiotherapy Device for Post-Surgery Rehabilitation
- Wrist CPM Unit for Joint Exercise and Mobility Restoration-Electric Model
- Motorized Wrist CPM Therapy Machine-Adjustable Motion and Intensity Levels
- Electric Wrist Rehabilitation CPM Device-Remote Controlled Premium Model
- Compact Wrist CPM Device-Continuous Passive Motion Equipment for Therapy
- Wrist CPM Machine for Passive Motion Therapy and Post-Injury Recovery
- Electric Wrist Exercise CPM Device-Adjustable Range for Joint Movement
- Automatic Wrist CPM Machine-Safe and Effective Rehabilitation Solution
- Premium Electrical Wrist CPM Machine for Physiotherapy and Rehabilitation Use
- Automatic Wrist CPM Therapy Device-Continuous Passive Motion Equipment
- Wrist Joint Continuous Passive Motion Machine-Remote Controlled Design
- Electric Wrist Rehabilitation CPM Device for Orthopedic Therapy Centers
- Advanced Motorized Wrist CPM Machine with Adjustable Speed and Angle
- Digital Wrist CPM Physiotherapy Equipment for Joint Recovery and Flexibility
- Wrist Rehabilitation CPM Machine-Premium Electrical Model for Therapy Clinics
- Electrical Wrist CPM Device for Post-Operative and Injury Rehabilitation
- Continuous Passive Motion Machine for Wrist Joint Exercise and Mobility Training
- Automatic Wrist CPM Therapy Unit-Ideal for Clinical and Home Rehabilitation
- Wrist Joint Therapy CPM Machine-Smooth and Controlled Range of Motion
- Motorized Wrist CPM Machine-Designed for Rehabilitation and Joint Recovery
- Digital Wrist CPM Therapy Machine-Simple Remote Controlled Operation
- Electric Wrist CPM Equipment-Compact, User-Friendly Physiotherapy Device
- Digital Wrist CPM Machine-90 Flexion and Dorsiflexion Range
- Wrist CPM Physiotherapy Device-Automatic Motorized Recovery Equipment
- Professional Wrist CPM Therapy Machine for Hospitals and Clinics
- Electric Wrist Continuous Passive Motion Device for Therapeutic Exercises
- Wrist Joint CPM Device-Ideal for Orthopedic and Neurological Rehabilitation
- Electrical Motorized Wrist CPM Machine-Adjustable Intensity and Speed Levels
- Wrist CPM Premium Model-Digital Physiotherapy Device for Joint Flexibility
- Wrist CPM Premium Model-Advanced Rehabilitation and Recovery Equipment
- Automatic Wrist Rehabilitation Machine-Safe and Efficient Recovery Solution
- Compact Electrical Wrist CPM Machine-Suitable for Clinical and Home Therapy
- Electric Wrist CPM Equipment-Remote Controlled Therapy Machine
- Wrist CPM Machine-Adjustable Speed and Intensity for Personalized Treatment
- Wrist CPM Physiotherapy Machine-Electrical Model with Remote Operation
- Digital Wrist CPM Device for Physiotherapy and Post-Surgery Mobility
- Wrist CPM Machine-Automatic Electric Device for Joint Rehabilitation Therapy
- Continuous Passive Motion Wrist Machine-Premium Model for Physiotherapy Use
- Motorized Wrist CPM Equipment with Adjustable Motion Control Settings
- Advanced Electrical Wrist CPM Device for Post-Injury and Post-Surgery Recovery
- Wrist CPM Therapy Equipment-Remote Controlled Physiotherapy Device
- Automatic Wrist Rehabilitation CPM Machine-Digital Speed and Angle Control
- Wrist Joint Physiotherapy CPM Machine for Continuous Passive Exercise
- Premium Motorized Wrist CPM Machine-Compact and Efficient Design
- Rehabilitation CPM Machine for Wrist Joint Flexibility and Strength Restoration
- Continuous Passive Motion Therapy Machine for Wrist Joint Exercise
- Imported Digital Ankle Continuous Passive Motion Machine for Rehab
- Ankle CPM Therapy Unit With Angle Indication and Comfort Straps
- Electric Ankle Mobilization CPM With 19 Speed and Angle Levels
- Rehabilitation Ankle CPM Machine With Long Press Flexion & Stretch Modes
- Ankle Physiotherapy CPM Device With Motorized Operation and Straps
- Advanced Ankle CPM Unit With Digital Controls for Post-Surgery Recovery
- Clinic Grade Ankle Continuous Passive Motion Device With Remote
- Motorized Ankle CPM Machine With Adjustable Flexion and Extension Range
- Imported Motorized Ankle CPM Machine With Remote Control
- Ankle Continuous Passive Motion Device For Rehab & Recovery
- Motorized Ankle CPM Unit With Adjustable Speed And Angle Levels
- Plantar Flexion & Dorsiflexion Ankle CPM For Physiotherapy Clinics
- Digital Ankle CPM With Angle Indication And Adjustable Straps
- Rehabilitation Ankle CPM Machine With 19 Level Speed Control
- Remote Operated Ankle CPM For Post-Surgery Mobility Training
- Motorized Ankle Therapy CPM With Long-Press Flexion & Stretch Modes
- Professional Ankle CPM Device For Hospital And Clinic Use
- Adjustable Ankle CPM Machine For Physiotherapy And Rehabilitation
- Remote Operated Ankle CPM Machine For Post-Surgery Recovery
- Advanced Ankle CPM Unit With Long-Press Flexion And Stretch Modes
- Ankle Therapy CPM Machine With Angle Indication And Comfort Straps
- Professional Motorized Ankle CPM For Hospital And Clinic Use
- High-Quality Imported Ankle CPM For Mobility & ROM Restoration
- Durable Ankle CPM Device For Physiotherapy And Rehabilitation Centers
- Imported Ankle CPM Therapy Machine With Digital Angle Display
- Ankle Joint CPM Machine With Remote, Speed & Angle Adjustments
- Advanced Rehab Ankle CPM Unit For Post-Injury Mobility Training
- Automatic Ankle CPM Machine With Long-Press Flexion & Stretch Options
- Clinical Grade Motorized Ankle CPM For Physiotherapy Departments
- Adjustable Ankle CPM Equipment For Ligament & Post-Surgery Rehab
- Premium Motorized Ankle CPM With 19 Speed And Angle Levels
- Remote-Controlled Ankle CPM System For Safe Passive Motion Therapy
- High-Performance Ankle CPM Machine For Rehabilitation Clinics
- Imported Digital Ankle CPM With Soft Adjustable Straps
- Ankle Mobility CPM Device Designed For Comfort And Smooth Operation
- Ankle CPM With Precision Angle Control For Effective Therapy Sessions
- Hospital Use Motorized Ankle CPM With Long Press Stretch Mode
- Sturdy Ankle CPM Machine For Daily Physiotherapy And ROM Recovery
- Imported Motorized Ankle CPM Unit For Smooth Passive Motion Therapy
- Ankle CPM Machine With Remote Control And Adjustable Therapy Levels
- Rehabilitation Ankle CPM System For Safe PlantarDorsiflexion Movement
- Motorized Ankle Therapy CPM Designed For Post-Surgical Recovery
- Professional Ankle CPM Device With Long-Press Flexion And Stretch Modes
- Heavy-Duty Motorized Ankle CPM For Physiotherapy Treatment Centers
- Ankle CPM For Restoring Joint Mobility After Injury Or Surgery
- Digital Ankle CPM Machine With Multiple Speed And Intensity Settings
- Angle Adjustable Ankle CPM Machine Suitable For All Age Groups
- Imported Ankle CPM Rehabilitation Machine For Passive Joint Movement
- Motorized Ankle CPM Device For Smooth Plantar And Dorsiflexion Training
- Ankle Therapy CPM Machine With Remote And Adjustable Range Settings
- High-Accuracy Ankle CPM System For Post-Fracture And Post-Surgery Rehab
- Advanced Motorized Ankle CPM For Daily Physiotherapy Use
- Ankle Joint CPM Device With Digital Angle Indication And Speed Levels
- Durable Ankle CPM Machine Designed For Hospital And Clinic Treatment
- Rehab-Grade Ankle CPM With Long-Press Flexion And Stretch Features
- Easy-To-Operate Ankle CPM Machine With Comfortable Support Straps
- Automatic Ankle CPM Therapy System For Controlled Movement Training
- Motorized Ankle Mobility CPM Designed For Lower Limb Recovery
- Professional Ankle CPM Unit For Better Mobility And Flexibility Training
- Angle And Speed Adjustable Ankle CPM For Lower Limb Physiotherapy
- Imported Motorized Ankle CPM For Controlled Flexion And Extension Therapy
- Ankle CPM Rehabilitation Device With Adjustable Speed And Angle Control
- Remote Operated Ankle CPM For Early-Stage Recovery And Mobility Training
- Heavy-Duty Ankle CPM Machine Designed For Continuous Therapy Sessions
- Motorized Ankle CPM Machine For Physiotherapy & Rehabilitation
- Adjustable Range Ankle CPM Unit For Mobility Training
- Clinical Grade Ankle Rehabilitation CPM Equipment
- Physiotherapy Ankle CPM Machine For Joint Mobilization
- Imported Ankle CPM Device With Adjustable Speed & Angle
- Ankle Rehab CPM Machine With Comfort Straps & Foot Plate
- Electric Ankle Continuous Motion Therapy Machine For Clinics
- Ankle CPM Exercise Machine For Post-Immobilization Rehab
- Compact Design Motorized Ankle CPM Rehabilitation Device
- Cryo Cold Therapy Machine With Shoulder, Knee, Ankle, Back Pads
- Professional Cryo Cooling System for Ortho & Sports Injury Care
- Motorised Cold Therapy Machine for Ortho Rehabilitation
- Cryo Cooling Therapy System with Universal Joint Pads
- Cryo Therapy Device for Post-Operative Swelling Reduction
- Rehabilitation Cold Therapy Unit with Multiple Attachments
- Motorised Cryo Cooling Machine for Joint & Muscle Recovery
- Cold Therapy Circulation System for Physiotherapy Centres
- Cryo Cooling Machine Designed for Sports Injury Treatment
- Automatic Motorised Cold Therapy Device for Faster Healing
- Advanced Motorised Ice Therapy Unit for Clinical Use
- Cryo Therapy Circulation Device for Inflammation Relief
- Medical Grade Cryo Cooling Unit with 4 Body Attachments
- Motorised Cryo Cooling Machine for Targeted Cold Therapy
- Cold Therapy Unit with Interchangeable Joint Pads
- Electric Cryo Therapy Device for Pain & Swelling Control
- Therapeutic Cold Circulation Machine for Rehabilitation
- Cryo Cold Therapy Equipment for Post-Injury Recovery
- High-Performance Motorised Cooling Unit for Physiotherapy
- Cold Therapy Machine with Shoulder, Knee, Ankle, Back Attachments
- Motorised Cryo Therapy Device for Clinical and Home Treatment
- Electric Cryo Cooling Therapy Unit for Pain Relief and Rehabilitation with Multi-Joint Pads
- Motorized Cryo Cold Therapy Unit with 4 Attachments for Pain Relief & Rehab
- Electric Cold Compression Therapy Machine for Shoulder, Knee, Ankle & Back
- Rehabilitation Cryo Cooling Machine with Multi-Joint Pads
- Advanced Cryo Compression Therapy Device for Clinics & Sports Rehab
- Motorized Cryo Therapy Unit for Swelling Reduction and Injury Recovery
- Cold Therapy Machine with 4 Interchangeable Attachments
- Physiotherapy Cryo Compression Device for Injury Recovery
- Cryo Cooling System for Shoulder, Knee, Ankle, Back & Hip Treatment
- Motorized Cryo Cooling Therapy Machine for Complete Joint Rehabilitation
- Cold Compression Therapy Unit with Shoulder, Knee, Ankle & Back Pads
- Electric Cryo Therapy Device for Orthopedic and Physiotherapy Use
- Motorized Cold Therapy System for Pain Management & Post-Operative Care
- Cryo Cooling Treatment Machine with 4 Body Area Attachments
- Rehab Cold Therapy Cooler for Joint Injuries and Muscle Inflammation
- Electric Cryo Compression Unit Suitable for Clinics and Home Users
- Multi-Joint Cryo Therapy Device for Shoulder, Hip, Knee & Ankle Relief
- Advanced Cold Therapy System for Rehabilitation Centres and Hospitals
- Motorized Cryotherapy Machine for Swelling, Pain Relief and Recovery
- Cold Therapy Machine with Interchangeable Pads for Full Body Support
- Cryo Cold Therapy Motorized System for Joint Pain Management
- Motorized Cryo Therapy Unit for Targeted Joint Cooling and Recovery
- Electric Cold Therapy Machine with Pads for Shoulder, Knee, Ankle & Back
- Cold Therapy Motorized Cooler for Orthopedic and Sports Injury Care
- Therapeutic Cryo Cooling Machine for Clinics, Hospitals and Rehab Centers
- Multi-Joint Cryo Compression Device for Shoulder, Hip, Knee & Ankle
- Cold Therapy Machine with Motorized Circulation for Faster Recovery
- Electric Cold Therapy Device for Physiotherapy and Ortho Treatment
- Complete Cryo Cooling System with Interchangeable Body Wraps
- Cold Therapy Circulation Machine for Sports Rehab and Medical Use
- Motorized Cryo Cooling Machine for Joint Pain Relief and Rehabilitation
- Cryo Compression Therapy Unit for Medical and Sports Injury Recovery
- Cryo Cooling Treatment Device with 4 Specialized Attachments
- Electric Cryo Therapy Cooler for Targeted Cold Compression
- Cold Therapy Machine with Adjustable Pads for Multiple Body Areas
- Motorized Cryo Compression Unit for Effective Swelling Reduction
- Cryo Cooling Circulation Machine for Shoulder, Knee, Ankle & Hip
- Electric Cryo Cold Therapy Machine for Pain, Swelling & Inflammation
- Cold Compression Machine for Shoulder, Back, Knee and Ankle Treatment
- Cryo Cooling Unit for Physiotherapy, Ortho and Sports Medicine
- Cryo Therapy Motorized Device for Professional Rehab Clinics
- Electric Cryo Cooling Therapy Machine for Fast Joint Recovery
- Motorized Cold Therapy Unit with Dedicated Pads for All Major Joints
- Cryo Cooling Rehab Machine for Pain Relief and Post-Trauma Care
- Electric Cryo Circulation Therapy Device for Physiotherapy Centres
- Cold Therapy Motorized Device for Shoulder, Knee, Ankle & Back Support
- Cold Therapy Motorized Cooler Unit for Pain Relief & Rehabilitation
- Electric Cryo Therapy Device for Swelling, Pain & Inflammation Relief
- Motorized Cold Therapy Unit for Orthopedic and Sports Recovery
- High-Performance Cryo Cooling Unit with 4 Body Attachments
- Motorized Cooling Therapy Machine for Post-Operative Recovery
- Motorized Cryo Therapy Unit for Swelling & Inflammation Control
- Motorized Cryo Cooling Therapy Device for Full Body Joint Care
- Advanced Electric Cold Therapy Machine for Rehabilitation Use
- Cold Circulation Therapy System for Shoulder, Knee, Ankle & Back
- Professional Cryo Therapy Device for Clinical Pain Management
- Motorized Cold Pad Therapy Device for Pain & Inflammation Control
- Cryopush Digital Cold Compression Therapy Device with Motorized Cooling and 4 Attachments
- Cryopush Cold & Intermittent Compression Machine for Knee, Ankle and Shoulder Pain Relief
- Motorized Cryopush Cold Compression Unit with 9 Modes and Adjustable Pressure Levels
- Cryopush Digital Cryotherapy and Compression Device with LCD Display and Timed Sessions
- Cold Compression Therapy System by Cryopush with 3.6L Reservoir for Faster Recovery
- Professional Cryopush Cold Compression Device with Multi-Joint Pads
- Cryopush Digital Cold Therapy Machine with Intermittent Compression for Swelling Reduction
- Cryopush Digital Cold Compression Therapy Device with LCD Display
- Motorized Cold and Compression System with 4 Joint Attachments
- Cryopush 3.6L Cold Compression Machine for Physiotherapy Clinics
- Digital Cold Therapy Unit with Knee, Ankle and Shoulder Pads
- Motorized Cryotherapy Compression Device for Injury Recovery
- Multi-Joint Cold Compression Device with Digital Control Panel
- Cryopush Cold Therapy and Compression Machine for Swelling Reduction
- Professional Motorized Cold Compression Unit for Sports Injury Care
- Cold Compression System with Adjustable Modes and Time Settings
- Motorized Cooling and Compression Device for Joint Rehabilitation
- Cryopush 4-Pad Cold Compression Unit for Home and Clinic Use
- Digital LCD Cold Therapy Device with High-Power Compression
- Cryopush Cold Therapy Machine for Knee, Shoulder and Ankle Support
- Advanced Cold Compression Unit with 9 Modes and Timer Options
- Motorized Cold Compression System for Effective Injury Treatment
- Cryopush Digital Cooling and Compression Unit with 1.8" Display
- Cryopush Motorized Cold Compression Therapy Machine for Faster Recovery
- Digital Cooling and Compression Device with Multiple Joint Attachments
- Cryopush 3.6L Cold Compression Therapy Unit for Injury Management
- Advanced Digital Cold Compression Machine for Orthopedic Rehabilitation
- Cryopush Cold Therapy System with Adjustable Compression Levels
- Motorized Cold Compression Device with Timer and LCD Display
- Digital Cold Compression Unit Ideal for Post-Operative Recovery
- Cryopush Intermittent Compression Cold Therapy Device for Clinics
- Cold Compression Therapy Machine with Motorized Operation
- Cryopush Cold Therapy Device for Joint Pain and Swelling Control
- Motorized Digital Cold Therapy System for Home and Clinical Use
- Cryopush Cold and Compression Therapy Device for Enhanced Healing
- Portable Digital Cold Compression Unit with Easy Controls
- Cryopush Multi-Pad Cold Compression System for Full Joint Coverage
- Advanced Cryopush Cold Compression Therapy Unit with High Efficiency
- Cryopush Cold Compression Machine with User-Friendly LCD Display
- Professional Digital Cold Compression Therapy System for Pain Relief
- Motorized Cold Compression Device Suitable for Orthopedic Care
- Digital Cold and Compression Unit with Intermittent Pressure
- Cryopush Cold Compression Device Designed for Physiotherapy Centres
- Cryopush Digital Cold Compression Unit for Muscle and Joint Recovery
- Motorized Cold Compression Therapy Machine with Easy LCD Controls
- Cryopush Advanced Cooling and Compression Device for Rehab Clinics
- Digital Cold Therapy Machine with Multi-Joint Pads and Timer
- Cryopush Cold Compression System Designed for Post-Injury Care
- Motorized Digital Cold Compression Device for Pain and Swelling Relief
- Cryopush Cold Therapy Unit with High-Capacity 3.6L Reservoir
- Cold Compression Therapy Machine with Adjustable Pressure Modes
- Cryopush Digital Cold Compression Device for Orthopedic Treatment
- Multi-Joint Cryopush Cold Therapy System with Smart LCD Display
- Motorized Cold Compression Unit Ideal for Sports Injury Recovery
- Cryopush Advanced Cold Therapy Machine for Knee and Shoulder Support
- Digital Cooling and Compression Device with 9 Treatment Modes
- Cryopush Motorized Cold Therapy Unit for Clinic and Home Use
- Professional Cold Compression Device with Multi-Area Coverage
- Cryopush Cold Compression Therapy System with Long Hose Design
- Digital Cryotherapy Compression Machine for Fast Inflammation Relief
- Cold Compression Therapy Device with Adjustable Time Cycles
- Cryopush Cold Therapy Machine for Comprehensive Joint Rehabilitation
- Digital Cold Compression System with Sturdy Motorized Operation
- Cryopush Multi-Joint Cooling Device for Physiotherapy Centres
- Motorized Cryopush Cold Compression Machine with Enhanced Performance
- Digital Cold Therapy and Compression Unit with User-Friendly Interface
- Cryopush Cold Compression Device for Effective Post-Surgery Recovery
- Advanced Digital Cold Therapy System with Customizable Settings
- Cryopush Cold Compression Machine Designed for Daily Clinical Use
- Motorized Digital Cold Compression Therapy Unit with Precision Cooling
- Cryopush Cold Therapy System with Quick and Efficient Operation
- Professional Digital Cold Compression Device for Injury and Rehab Support
- Motorized Cold Therapy System with Multi-Joint Support Attachments
- Digital Cooling and Compression Therapy Unit for Rehab Professionals
- Cryopush Intermittent Cold Compression Machine with LCD Interface
- Advanced Motorized Cold Compression Unit for Injury Prevention
- Cryopush Digital Cold Therapy Device for Swelling and Pain Control
- Multi-Pad Cold Compression System for Physiotherapy Treatment
- Digital Cryotherapy Compression Unit with Adjustable Intensity
- Cryopush Cold Therapy Device with Precise Temperature Management
- Professional Motorized Cold Compression System for Athletes
- Cryopush Digital Cold Therapy and Compression Device for Hospitals
- Cold Compression Machine with Long Hose and High-Capacity Tank
- Cryopush Multi-Joint Cold Therapy System with Strong Motor
- Focused Shockwave Therapy System for Advanced Musculoskeletal Treatment
- High Precision Focused Shockwave Physiotherapy Device for Ortho & Sports Rehab
- Medical Focus Shockwave Machine for Trigger Point and Deep Tissue Therapy
- Medical Focus Shockwave Machine for Trigger Point
- Focus Shockwave Physiotherapy Equipment for Calcification & Tendinopathy Care
- Acoustic Focus Shockwave Therapy Device for Professional Clinical Applications
- Focused Pressure Wave Shockwave Instrument with Multi-Mode Treatment Protocols
- Ultra-Efficient Focus Shockwave Rehabilitation Device for Hospitals & Clinics
- Medical Focus Shockwave Deep Penetration Device for Musculoskeletal Disorders
- Advanced Focused Shockwave Therapy Device for Deep Penetration Tissue Regeneration
- Deep Targeting Focused Shockwave System for Physiotherapy and Rehab Centres
- Precision-Control Focus Shockwave Machine for Musculoskeletal Disorders
- Advanced Focused Shockwave Unit for Soft Tissue Regeneration
- Clinical-Grade Focus Shockwave Device for Osteoarthritis and Myofascial Release
- Focus Shockwave Physiotherapy Machine for Sports Medicine and Orthopaedic Care
- Non-Invasive Focus Shockwave Treatment Machine for Rehabilitation Clinics
- Focused Shockwave Physiotherapy System for Enhanced Healing Response
- Professional Grade Focus Shockwave Device for Tendon Repair and Mobility Recovery
- Latest Technology Focus Shockwave Therapy Solution for Deep Tissue Penetration
- Innovative Focus Shockwave Device for Physiotherapy and Sports Injury Recovery
- High-Performance Focused Shockwave Therapy Equipment for Advanced Pain Relief
- Next-Gen Focus Shockwave Physiotherapy Machine for Chronic Pain Disorders
- High-Intensity Focus Shockwave Therapy Device for Rehabilitation Centres
- Precision Medical Focus Shockwave Unit for Tendon and Ligament Recovery
- Compact Clinical Focus Shockwave System for Physiotherapy Departments
- Focus Shockwave Pain Management Device for Orthopedic Rehabilitation
- Deep Target Focused Shockwave Generator for Sports Injury Healing
- Digital Focus Shockwave Device with Custom Therapy Protocol Programs
- Focus Shockwave Treatment Instrument for Advanced Physio Applications
- Strong Energy Focused Shockwave Machine for Myofascial Release and Trigger Pain
- Medical Grade Focus Shockwave System for Tissue Regrowth and Circulation Boost
- Focus Shockwave Therapy Equipment for Frozen Shoulder and Joint Stiffness
- Dynamic Focus Shockwave Rehabilitation Unit for Post-Traumatic Recovery
- Smart Controlled Focus Shockwave Device for Physiotherapy and Athletes
- Shockwave Focus Therapy Machine for Tendonitis, Ligament Tears and Joint Dysfunction
- Clinical Strength Focus Shockwave System for Osteoarthritis and Degenerative Joints
- Multi-Setting Focus Shockwave Physiotherapy Unit for Professional Rehab
- Focus Shockwave Therapeutic Device for Chronic Plantar and Heel Disorders
- Enhanced Penetration Focus Shockwave Machine for Deep Muscle Layers
- Physiotherapy Focus Shockwave Apparatus for Rehab Hospitals and Clinics
- Evidence-Based Focus Shockwave Machine for Chronic Musculoskeletal Disorders
- Focus Shockwave Treatment System for Faster Rehabilitation and Pain Reduction
- Performance Driven Focus Shockwave Unit for Athletes and Sports Medicine
- Heavy-Duty Focus Shockwave Therapy Equipment for Intensive Clinical Use
- Focus Shockwave Physiotherapy Device for Collagen Synthesis and Tissue Repair
- Focus Shockwave Pain Relief System for Chronic Tendon and Joint Conditions
- Professional Focus Shockwave Generator for Deep Soft Tissue Recovery
- Modern Focus Shockwave Therapy Machine for Orthopaedic Rehabilitation
- Deep Layer Focused Shockwave Device for Trigger Points and Myofascial Tightness
- Nova Focus Shockwave Therapy Unit for Next-Level Clinical Outcomes
- PulsePro Focus Shockwave Machine for Targeted Deep Healing
- RegenX Focus Shockwave Device for Accelerated Tissue Repair
- HyperFocus Shockwave Therapy Machine for Critical Pain Conditions
- Ultra Precision Focus Shockwave Therapy Machine for Deep Clinical Recovery
- Advanced Focus Shockwave System for High-Speed Orthopedic Rehabilitation
- High-Impact Focus Shockwave Device for Severe Chronic Pain Relief
- Clinical Focus Shockwave Therapy Equipment for Sports Injury Management
- Next-Level Focus Shockwave Machine for Deep Ligament & Tendon Repair
- Electromagnetic Focus Shockwave Therapy Unit for Targeted Tissue Regeneration
- Professional Focus Shockwave System for Non-Surgical Pain Treatment
- High-Energy Focus Shockwave Therapy Machine for Faster Mobility Restoration
- PowerCore Focus Shockwave Machine for Maximum Deep Tissue Impact
- PrimeCare Focus Shockwave Therapy Device for Orthopedic Excellence
- Infinity Focus Shockwave System for Continuous Healing Performance
- ProActive Focus Shockwave Therapy Unit for Rapid Functional Recovery
- OrthoElite Focus Shockwave Machine for Professional Pain Management
- RegenForce Focus Shockwave Therapy System for Accelerated Healing
- UltraMed Focus Shockwave Device for High-Precision Clinical Results
- Astra Focus Shockwave Therapy Machine for Next-Gen Rehabilitation
- Zenith Focus Shockwave Therapy System for Deep Musculoskeletal Recovery
- Professional Focus Shockwave Therapy Device for Sports Performance Recovery
- Advanced Clinical Focus Shockwave Machine for Orthopedic Pain Solutions
- High-Power Focus Shockwave System for Chronic Tendon & Ligament Injuries
- Next-Gen Focus Shockwave Therapy Machine for Non-Invasive Recovery Care
- Clinical Performance Focus Shockwave Therapy System for Long-Lasting Results
- Crown Focus Shockwave Therapy System for Elite Clinical Performance
- ForceEdge Focus Shockwave Machine for Maximum Therapeutic Penetration
- MedX Focus Shockwave Therapy Device for Advanced Orthopedic Care
- UltraForce Focus Shockwave System for Rapid Recovery Outcomes
- PrimeAxis Focus Shockwave Therapy Unit for Deep Targeted Healing
- RegenPulse Focus Shockwave Machine for High-Speed Tissue Regeneration
- OrthoDrive Focus Shockwave Therapy System for Total Pain Control
- InfinityCore Focus Shockwave Therapy System for Complete Rehabilitation Excellence
- PrecisionMed Focus Shockwave Therapy Machine for Advanced Joint Restoration
- UltraZen Focus Shockwave Therapy System for Pain-Free Mobility Recovery
- Advanced Focus Shockwave Therapy Machine for Deep Pain Resolution
- High-Definition Focus Shockwave System for Targeted Orthopedic Healing
- Professional Focus Shockwave Device for Sports Injury & Muscle Recovery
- Ultra-Control Focus Shockwave Therapy Machine for Precision Pain Therapy
- Next-Stage Focus Shockwave System for Chronic Joint & Tendon Conditions
- Clinical Precision Focus Shockwave Therapy Equipment for Faster Recovery
- High-Performance Focus Shockwave Device for Advanced Physiotherapy Clinics
- TargetCore Focus Shockwave Therapy System for Deep Musculoskeletal Repair
- RapidHeal Focus Shockwave Machine for Long-Term Pain Management
- OrthoPrime Focus Shockwave Therapy System for Advanced Clinical Results
- MaxEdge Focus Shockwave Machine for High-Power Deep Therapy
- PulseAxis Focus Shockwave Therapy Unit for High-Speed Orthopedic Recovery
- EliteCore Focus Shockwave Machine for Deep Clinical Performance
- ProMotion Focus Shockwave Therapy System for Pain-to-Performance Recovery
- AstraPulse Focus Shockwave Device for Next-Level Therapeutic Outcomes
- OrthoFusion Focus Shockwave Therapy Machine for Complete Pain Restoration
- VitalForce Focus Shockwave Therapy System for Deep Healing Excellence
- Professional Focus Shockwave Therapy Device for Sports & Clinical Use
- High-Impact Focus Shockwave Machine for Chronic Pain Management
- Next-Gen Focus Shockwave Therapy System for Deep Joint & Spine Care
- High-Speed Focus Shockwave Therapy Machine for Rapid Functional Recovery
- Targeted Focus Shockwave System for Ligament, Tendon & Muscle Injuries
- Hospital-Grade Focus Shockwave Therapy Unit for Long-Term Pain Relief
- PrimeForce Focus Shockwave Therapy System for Elite Healing Performance
- RegenCore Focus Shockwave Machine for Accelerated Tissue Repair
- UltraMedix Focus Shockwave Therapy Device for Advanced Ortho Solutions
- PulsePrime Focus Shockwave System for Deep Clinical Impact
- OrthoWave Pro Focus Shockwave Therapy Unit for Precision Pain Control
- MaxRegen Focus Shockwave Machine for High-Power Musculoskeletal Therapy
- ElitePrecision Focus Shockwave Therapy System for Professional Clinics
- ApexRehab Focus Shockwave Device for Next-Level Recovery Outcomes
- ProHeal Focus Shockwave Therapy Machine for Long-Lasting Pain Freedom
- ZenPro Focus Shockwave Therapy System for Deep Healing & Mobility Care
- Ultra-Target Focus Shockwave Therapy Machine for Deep Clinical Pain Relief
- Advanced Focus Shockwave System for High-Speed Tissue Regeneration
- Professional Focus Shockwave Therapy Device for Sports Injury Rehabilitation
- High-Energy Focus Shockwave Machine for Severe Musculoskeletal Disorders
- Next-Phase Focus Shockwave Therapy System for Deep Joint Mobility Recovery
- Precision-Control Focus Shockwave Device for Targeted Orthopedic Therapy
- Clinical-Grade Focus Shockwave Therapy Equipment for Long-Term Pain Care
- Rapid-Response Focus Shockwave Therapy Machine for Soft Tissue Repair
- Power-Driven Focus Shockwave System for Advanced Physiotherapy Treatment
- Hospital-Class Focus Shockwave Therapy Unit for Complete Pain Resolution
- DVT 6 Chamber Digital Compression Therapy System for Legs, Waist & Arm Rehabilitation
- 6 Chamber DVT Pump with Touch Panel & Remote | Professional Lymphedema Therapy Device
- Advanced Digital DVT Compression Machine with 4 Modes & Multiple Sleeves
- DVT 6 Chamber Sequential Air Compression Device for Swelling & Circulation Therapy
- Clinical Grade 6 Chamber DVT Pump with Leg Extensions, Waist & Arm Sleeves
- Digital DVT Compression Therapy Unit with Adjustable Pressure 30250 mmHg
- Professional 6 Chamber DVT Lymphedema Pump with Timer & Dual Displays
- Remote-Controlled DVT 6 Chamber Air Compression System for Physiotherapy Clinics
- Touch Panel DVT Therapy Device with 4 Treatment Modes & Digital Readout
- Complete 6 Chamber DVT Compression Solution for Edema, Varicose Veins & Recovery
- Digital 6 Chamber DVT Therapy Pump with Multi-Sleeve Support for Full Limb Care
- Professional Grade DVT 6 Chamber Compression System with Smart Touch Display
- Advanced Sequential Air Compression DVT Machine with Remote & Digital Controls
- 6 Chamber Digital DVT Pump for Lymphedema, Swelling Reduction & Circulation Boost
- Clinical DVT Compression Device with 4 Operating Modes and Adjustable Pressure
- Full Body Air Compression Therapy System with Leg, Waist & Arm Sleeves
- High-Performance DVT 6 Chamber Pump with Timer Settings & Digital Pressure Display
- Digital 6 Chamber Sequential Compression Unit for Physiotherapy & Recovery
- Touch Panel DVT Therapy Device with Remote and Multi-Mode Operation
- Modern DVT Air Compression Pump for Varicose Veins & Edema Management
- DVT 6 Chamber Digital System with Leg Extensions and Complete Sleeve Set
- Smart Digital DVT Compression Pump for Post-Surgery Rehabilitation
- Fully Automatic 6 Chamber Air Compression Device for Deep Circulation Therapy
- Hospital-Grade DVT Pump with Digital Timer, Pressure Range & Remote Control
- Compact 6 Chamber Digital DVT System for Daily Clinical Use
- Ultra-Comfort 6 Chamber DVT Compression Pump with Digital Monitoring
- Digital Sequential Compression Therapy Machine with Complete Sleeve Set
- Premium 6 Chamber DVT Pump for Edema, Lymphedema & Vein Health Support
- Advanced Limb Compression Therapy Device with 4 Digital Modes
- Therapeutic 6 Chamber Air Compression Pump for Improved Blood Flow
- Smart DVT Compression Therapy Unit with Adjustable Pressure & Timer
- Digital Multi-Chamber DVT Device for Effective Swelling Reduction
- Ergonomic 6 Chamber Compression Therapy Pump for Clinics & Home Care
- Precision-Controlled DVT Pump with Digital Pressure & Time Settings
- Full-Limb Compression Therapy Machine with Remote & Touch Panel
- 6 Chamber Digital Air Pressure Therapy System for Post-Operative Recovery
- Reliable DVT Compression Device with Multiple Modes and Leg Extensions
- Digital Sequential Compression Pump for Fast Recovery & Circulation Care
- Compact DVT 6 Chamber Therapy Unit for Everyday Physiotherapy Use
- Digital DVT 6 Chamber Compression Pump for Professional Physiotherapy Use
- Smart Air Compression DVT Device with Multi-Zone Treatment and Digital Control
- 6 Chamber Sequential DVT Therapy Pump for Complete Lower & Upper Limb Care
- Advanced Digital DVT Pump with Pressure Control and Multiple Sleeve Options
- Therapeutic 6 Chamber Compression System for Edema and Circulation Recovery
- Digital DVT Machine with Touch Interface and Full Sleeve Connectivity
- Professional Sequential Compression Pump with 4 Digital Therapy Modes
- 6 Chamber Digital Air Compression Therapy Unit for Fast Swelling Relief
- Modern DVT Pump with Adjustable Pressure Range and Smart Timer Settings
- Comprehensive DVT Compression Device for Leg, Waist & Arm Rehabilitation
- Multi-Mode DVT 6 Chamber Therapy Pump for Clinical and Home Treatments
- Digitally Controlled Sequential Compression Device for Recovery Support
- Digital Air Compression DVT Unit with Remote Navigation & Touch Panel
- KM521 Pelvic Muscle Stimulator For Pelvic Floor Rehabilitation and Women's Health
- Advanced Dual Channel Pelvic Floor Rehabilitation Stimulator KM521
- KM521 Pelvic Muscle Therapy Device with Voice Reminder & Animation Display
- Pelvic Floor Muscle Stimulation Machine with 0-99 Intensity-KM521
- Dual Channel Pelvic Muscle Re-education Stimulator KM521
- Digital Pelvic Floor Strengthening Device with 9 Preset ModesKM521
- KM521 Pelvic Muscle Stimulator for Pelvic Floor Strengthening and Rehabilitation
- Advanced Pelvic Floor Muscle Stimulation Machine KM521 with Dual Channel Output
- Pelvic Muscle Rehabilitation Device KM521 with TFT Colour Screen and Voice Guide
- KM521 Pelvic Floor Therapy Machine for Women Health and Muscle Recovery
- Rechargeable Pelvic Muscle Stimulator KM521 with 9 Therapy Modes
- High Power Class 4 Laser Therapy Machine for Advanced Physiotherapy Clinics
- Clinical Grade Class 4 Laser Therapy Machine for Pain Relief and Rehabilitation
- Professional Class 4 Laser Therapy Device for Advanced Physiotherapy Treatment
- High Power Medical Class 4 Laser Machine for Deep Tissue Physiotherapy
- Advanced Physiotherapy Class 4 Laser Therapy Machine with High Output Power
- Class 4 High Power Laser Therapy System for Sports and Orthopedic Rehabilitation
- Premium Class 4 Laser Therapy Equipment for Physiotherapy and Pain Clinics
- High Performance Class 4 Laser Therapy Machine for Clinical Rehabilitation Use
- Advanced Class 4 Laser Therapy Device for Musculoskeletal Pain Management
- High Power Class 4 Laser Physiotherapy Machine for Faster Patient Recovery
- Clinical Use Class 4 Laser Therapy System for Advanced Physiotherapy Centers
- Professional High Power Class 4 Laser Therapy Machine for Rehab Clinics
- Next Generation Class 4 Laser Therapy Equipment for Physiotherapy Professionals
- High Intensity Class 4 Laser Therapy Machine for Deep Tissue Healing
- Advanced Class 4 Laser Therapy Solution for Modern Physiotherapy Clinics
- High Power Class 4 Laser Therapy Device for Orthopedic and Sports Rehab
- Medical Grade Class 4 Laser Therapy Machine for Advanced Clinical Practice
- Powerful Class 4 Laser Therapy System for Physiotherapy and Rehabilitation Clinics
- Advanced Class 4 Laser Therapy Equipment for Modern Rehabilitation Centers
- Professional High Intensity Class 4 Laser Machine for Physiotherapy Use
- Clinical Grade High Power Class 4 Laser Therapy System
- Advanced Physiotherapy Laser Therapy Machine Class 4 High Output
- High Performance Class 4 Laser Therapy Device for Rehab Clinics
- Medical Class 4 Laser Therapy Machine for Advanced Pain Management
- Professional Class 4 Laser Therapy System for Physiotherapy Experts
- High Power Class 4 Laser Physiotherapy Equipment for Deep Tissue Treatment
- Advanced Clinical Class 4 Laser Therapy Machine for Rehabilitation
- Class 4 Laser Therapy Machine for Sports Injury Physiotherapy Clinics
- High Intensity Class 4 Laser Therapy Device for Orthopedic Rehabilitation
- Professional Grade Class 4 Laser Therapy Equipment for Physiotherapy
- High Power Medical Laser Therapy System Class 4 for Rehab Use
- Clinical Class 4 Laser Therapy Machine for Musculoskeletal Pain Relief
- Advanced Physiotherapy Class 4 Laser Therapy Device High Output
- High Power Class 4 Laser Therapy System for Pain and Rehab Clinics
- Professional Class 4 Laser Physiotherapy Machine for Clinical Practice
- High Intensity Class 4 Laser Therapy Machine for Physiotherapy Treatment
- Clinical Use High Power Class 4 Laser Therapy Device
- Advanced Class 4 Laser Therapy Machine for Pain Relief Clinics
- Professional High Power Laser Therapy Equipment Class 4
- Class 4 Laser Therapy System for Advanced Physiotherapy Applications
- High Power Clinical Laser Therapy Machine Class 4
- Advanced Physiotherapy Laser Machine Class 4 High Intensity
- Medical Grade Class 4 Laser Therapy Device for Rehab Clinics
- Professional Class 4 Laser Therapy Machine for Physiotherapy Clinics
- High Power Class 4 Laser Therapy Machine for Advanced Rehabilitation Clinics
- Advanced Class 4 Laser Therapy Device for Professional Physiotherapy Use
- Clinical Grade High Intensity Class 4 Laser Therapy Machine
- High Performance Class 4 Laser Therapy Equipment for Pain Management
- Professional Class 4 Laser Therapy Machine for Modern Physiotherapy Clinics
- Advanced High Power Class 4 Laser Therapy System for Rehabilitation
- High Intensity Class 4 Laser Therapy Machine for Deep Tissue Treatment
- Advanced Clinical Class 4 Laser Therapy Equipment for Physiotherapy Centers
- Professional High Power Class 4 Laser Therapy Device for Rehab Clinics
- Clinical Use Class 4 Laser Therapy Machine for Advanced Pain Care
- High Performance Class 4 Laser Physiotherapy Machine for Daily Clinical Use
- Advanced Class 4 Laser Therapy System for Musculoskeletal Rehabilitation
- High Power Class 4 Laser Therapy Equipment for Sports Physiotherapy Clinics
- Professional Medical Class 4 Laser Therapy Machine for Rehabilitation
- Advanced Physiotherapy Class 4 Laser Therapy Device for Pain Management
- High Intensity Clinical Class 4 Laser Therapy Machine
- Advanced Clinical Laser Therapy Machine Class 4 for Physiotherapy
- Medical Grade Class 4 Laser Therapy Device for Pain Relief Clinics
- Professional Class 4 Laser Physiotherapy Equipment for Clinical Treatment
- Advanced High Power Laser Therapy Device Class 4 for Clinics
- Clinical Class 4 Laser Therapy Equipment for Modern Physiotherapy
- Professional Class 4 Laser Therapy Machine for Sports Injury Rehab
- Advanced Medical Class 4 Laser Therapy Device for Physiotherapy Clinics
- High Power Clinical Class 4 Laser Therapy Equipment
- Professional Grade High Power Class 4 Laser Therapy System
- High Power Class 4 Laser Therapy Machine for Professional Physiotherapy Clinics
- Advanced Class 4 Laser Therapy System for Clinical Rehabilitation Use
- High Intensity Medical Class 4 Laser Therapy Machine for Pain Relief
- Professional Class 4 Laser Therapy Equipment for Advanced Physiotherapy
- Clinical Grade High Power Class 4 Laser Therapy Device
- Advanced Physiotherapy Class 4 Laser Therapy Machine for Pain Management
- Medical Grade Class 4 Laser Therapy Equipment for Physiotherapy Centers
- Professional High Intensity Class 4 Laser Therapy Machine
- Advanced Clinical Class 4 Laser Therapy Device for Rehab Applications
- High Power Class 4 Laser Therapy Machine for Orthopedic Physiotherapy
- Advanced Medical Laser Therapy Machine Class 4 for Rehabilitation
- Professional Grade Class 4 Laser Therapy Machine for Advanced Clinics
- Advanced Physiotherapy Laser Therapy Device Class 4 High Output
- High Performance Class 4 Laser Therapy Equipment for Sports Rehabilitation
- Medical Grade Class 4 Laser Therapy System for Pain Management Clinics
- High Performance Class 4 Laser Therapy Equipment for Daily Clinical Use
- Professional Class 4 Laser Therapy Machine for Orthopedic Rehabilitation
- High Power Class 4 Laser Therapy System for Sports Rehab Clinics
- Medical Class 4 Laser Therapy Machine for Advanced Physiotherapy Care
- Professional Grade Class 4 Laser Therapy Equipment for Rehab Professionals
- High Intensity Clinical Class 4 Laser Therapy Machine for Pain Relief
- Professional Class 4 Laser Therapy Device for Advanced Treatment Rooms
- Clinical Use Class 4 Laser Therapy System for Physiotherapy Clinics
- Professional High Power Class 4 Laser Therapy Solution for Rehab Centers
- High Power Class 4 Laser Therapy Machine for Advanced Physiotherapy Practice
- Advanced Clinical Class 4 Laser Therapy System for Pain Relief
- Professional High Intensity Class 4 Laser Therapy Equipment
- Medical Grade Class 4 Laser Therapy Machine for Rehabilitation Clinics
- Professional Class 4 Laser Therapy Device for Orthopedic Physiotherapy
- High Power Clinical Class 4 Laser Therapy Equipment for Pain Care
- Advanced Physiotherapy Class 4 Laser Therapy System for Clinics
- Medical Class 4 Laser Therapy Machine for Deep Tissue Healing
- High Power Class 4 Laser Therapy Machine for Daily Physiotherapy Use
- Clinical Grade Class 4 Laser Therapy Equipment for Rehab Clinics
- Advanced Physiotherapy Laser Therapy System Class 4 High Output
- Advanced Class 4 Laser Therapy System for Professional Pain Management
- Advanced Medical Class 4 Laser Therapy System for Deep Tissue Care
- High Performance Class 4 Laser Therapy Machine for Modern Clinics
- Professional Grade Class 4 Laser Therapy Equipment for Rehabilitation
- Advanced Physiotherapy Class 4 Laser Therapy Machine High Output
- Medical Class 4 Laser Therapy System for Orthopedic Rehabilitation
- Clinical Use Class 4 Laser Therapy Equipment for Pain Relief
- Advanced Clinical Class 4 Laser Therapy Device for Rehab Practice
- High Intensity Class 4 Laser Therapy Machine
- High Performance Class 4 Laser Therapy System for Physiotherapy Clinics
- Medical Grade Class 4 Laser Therapy Equipment for Advanced Treatment
- High Power Clinical Class 4 Laser Therapy Device
- Advanced Physiotherapy Class 4 Laser Therapy Equipment for Clinics
- Professional High Power Class 4 Laser Therapy Equipment for Rehab Centers
- Medical Grade Class 4 Laser Therapy Machine for Deep Tissue Rehabilitation
- High Intensity Class 4 Laser Therapy System for Modern Physiotherapy
- Advanced Physiotherapy Class 4 Laser Therapy Machine for Clinical Use
- Professional Grade Class 4 Laser Therapy Machine for Physiotherapy Clinics
- Advanced Class 4 Laser Therapy Machine for Orthopedic Physiotherapy
- Medical Class 4 Laser Therapy Device for Advanced Pain Care
- Professional High Intensity Class 4 Laser Therapy System
- Advanced Clinical Class 4 Laser Therapy Machine for Rehab Professionals
- Medical Grade High Power Class 4 Laser Therapy System
- Advanced Physiotherapy Class 4 Laser Therapy Device for Modern Clinics
- Professional Class 4 Laser Therapy Machine for Musculoskeletal Rehab
- High Power Class 4 Laser Therapy Machine for Advanced Physiotherapy Treatment
- Advanced Class 4 Laser Therapy System for Clinical Pain Relief
- Medical Grade High Intensity Class 4 Laser Therapy Machine
- Advanced Physiotherapy Class 4 Laser Therapy System for Daily Practice
- High Performance Class 4 Laser Therapy Device for Rehabilitation Centers
- Professional High Power Class 4 Laser Therapy Machine for Clinics
- Professional Class 4 Laser Therapy Equipment for Modern Rehab Clinics
- Medical Class 4 Laser Therapy System for Musculoskeletal Rehabilitation
- High Intensity Class 4 Laser Therapy Machine for Physiotherapy Clinics
- Professional Grade Class 4 Laser Therapy Device for Advanced Care
- Advanced Class 4 Laser Therapy Machine for Sports Physiotherapy
- High Power Clinical Class 4 Laser Therapy System for Rehab Use
- Medical Grade Class 4 Laser Therapy Equipment for Pain Clinics
- Professional Class 4 Laser Therapy Machine for Orthopedic Rehab
- Advanced Physiotherapy Laser Therapy System Class 4 High Power
- High Performance Class 4 Laser Therapy Equipment for Clinical Practice
- Professional High Intensity Class 4 Laser Therapy
- Advanced Medical Class 4 Laser Therapy Machine for Rehabilitation
- KM521 Pelvic Muscle Stimulator with TFT Colour Display & Voice Guidance
- KM521 Advanced Pelvic Floor Muscle Training Device for Rehab Use
- KM521 Digital Pelvic Floor Muscle Strengthening Machine for Therapy
- KM521 Smart Pelvic Muscle Training Device for Women & Men Therapy
- KM521 Pelvic Floor Exercise & Muscle Stimulation Machine
- KM521 Pelvic Muscle Rehab Trainer for Post Delivery & Surgery
- KM521 Pelvic Muscle Training Machine for Core & Bladder Control
- KM521 Digital Pelvic Floor Muscle Therapy Device for Rehabilitation
- KM521 Professional Pelvic Muscle Stimulator for Clinical & Home Use
- KM521 Advanced Pelvic Floor Muscle Rehab System with Guided Programs
- KM521 Pelvic Muscle Stimulation Device with TFT Screen & Voice Support
- KM521 Clinical Pelvic Floor Muscle Training Equipment for Rehab Centers
- KM521 Smart Pelvic Floor Muscle Training Machine with Preset Modes
- KM521 Digital Pelvic Muscle Strengthening Device with Voice Guidance
- KM521 Rechargeable Pelvic Floor Muscle Therapy Machine with Animation
- KM521 Smart Pelvic Muscle Rehabilitation Machine with Guided Programs
- KM521 Pelvic Muscle Therapy Device with Adjustable Intensity Levels
- KM521 Physiotherapy Pelvic Floor Muscle Training Equipment for Clinics
- KM521 Pelvic Muscle Stimulation Machine for Postnatal Recovery
- KM521 Medical Grade Pelvic Floor Muscle Rehab Device for Therapy
- KM521 Digital Pelvic Muscle Training System with Colour Screen
- KM521 Advanced Pelvic Floor Muscle Therapy Equipment with Preset Modes
- KM521 Pelvic Muscle Strength Training Device for Core Stability
- KM521 Professional Pelvic Floor Muscle Stimulator for Rehab Centers
- KM521 Clinical Pelvic Muscle Strengthening Equipment for Physiotherapy
- KM521 Digital Pelvic Muscle Stimulation System for Rehabilitation
- KM521 Pelvic Muscle Training Machine with TFT Screen & Voice Support
- KM521 Pelvic Muscle Stimulator with TFT Colour Screen & Voice Reminder
- KM521 Dual Channel Pelvic Muscle Therapy Machine with Adjustable Intensity
- KM521 Rechargeable Pelvic Muscle Training Equipment with Animation Display
- KM521 Medical Grade Pelvic Muscle Strengthening Device
- KM521 Pelvic Floor Therapy Machine with Colour Display & Voice Alerts
- KM521 Physiotherapy Pelvic Floor Muscle Training System
- KM521 Pelvic Muscle Training Machine for Bladder Control & Core Support
- KM521 Professional Pelvic Muscle Stimulator for Clinical & Home Therapy
- KM521 Pelvic Muscle Stimulation Device with TFT Screen & Dual Output
- KM521 Clinical Pelvic Floor Muscle Training Equipment for Therapy
- KM521 Pelvic Muscle Strength Improvement Machine for Daily Use
- KM521 Smart Pelvic Floor Muscle Training Device with Guided Modes
- KM521 Pelvic Muscle Stimulator with Dual Channel & Colour Display
- KM521 Digital Pelvic Muscle Strengthening Machine with Voice Guidance
- KM521 Rechargeable Pelvic Floor Muscle Training Device for Therapy
- KM521 Clinical Pelvic Muscle Rehabilitation Machine for Physiotherapy
- KM521 Smart Pelvic Floor Muscle Trainer with Preset Therapy Modes
- KM521 Medical Grade Pelvic Floor Muscle Training Equipment
- KM521 Pelvic Muscle Stimulation Machine for Strength & Control
- KM521 Physiotherapy Pelvic Floor Muscle Therapy System
- KM521 Pelvic Muscle Strength Training Machine for Core Stability
- KM521 Professional Pelvic Floor Muscle Stimulator for Daily Therapy
- KM521 Pelvic Floor Muscle Therapy Machine for Women & Men
- KM521 Rechargeable Pelvic Floor Muscle Training Machine
- KM521 Digital Pelvic Muscle Strengthening Device with Voice Reminder
- KM521 Rechargeable Pelvic Floor Muscle Therapy Equipment
- KM521 Clinical Pelvic Muscle Rehabilitation Device for Physiotherapy
- KM521 Smart Pelvic Floor Muscle Trainer with Adjustable Intensity
- KM521 Pelvic Muscle Therapy Machine for Strength & Control Training
- KM521 Medical Grade Pelvic Floor Muscle Stimulation Device
- KM521 Physiotherapy Pelvic Muscle Training System for Rehab
- KM521 Digital Pelvic Floor Muscle Therapy Device with Colour Screen
- KM521 Pelvic Floor Muscle Strength Training Equipment
- KM521 Pelvic Muscle Strength Improvement Device for Regular Use
- KM521 Smart Pelvic Floor Muscle Training Device with Preset Modes
- KM521 Professional Pelvic Muscle Stimulator for Daily Rehabilitation
- KM521 Rechargeable Pelvic Muscle Stimulation Machine for Therapy
- KM521 Pelvic Muscle Strength Improvement Device for Regular Training
- KM521 Pelvic Muscle Stimulator with Colour Screen for Pelvic Therapy
- KM521 Advanced Pelvic Floor Muscle Training Device with Guided Programs
- KM521 Digital Pelvic Muscle Strengthening System for Rehabilitation
- Professional Back Leg Chest Dynamometer for Physiotherapy & Rehabilitation
- Clinical Back, Leg and Chest Strength Dynamometer for Muscle Testing
- Back, Leg and Chest Muscle Strength Dynamometer for Physiotherapy Clinics
- Advanced Back Leg Chest Dynamometer for Orthopedic & Sports Assessment
- Medical Grade Back, Leg and Chest Dynamometer for Strength Measurement
- Back Leg Chest Dynamometer for Isometric Muscle Strength Testing
- Physiotherapy Back, Leg and Chest Dynamometer for Rehabilitation Progress Tracking
- Back, Leg and Chest Strength Testing Dynamometer for Sports & Medical Use
- Durable Back Leg Chest Dynamometer with High Accuracy Scale
- Back, Leg and Chest Dynamometer for Clinical & Fitness Strength Analysis
- Professional Muscle Strength Dynamometer for Back, Legs & Chest
- Multi-Purpose Back, Leg and Chest Dynamometer for Medical Assessment
- Back, Leg and Chest Dynamometer for Complete Muscle Strength Evaluation
- Back, Leg and Chest Dynamometer for Clinical & Field Strength Testing
- Back Leg Chest Dynamometer for Fitness, Rehab & Performance Testing
- Medical Grade Pelvic Floor Muscle Training Chair for Incontinence Care
- High Performance Pelvic Muscle Stimulation Chair for Rehabilitation Clinics
- Digital Clinical Pelvic Floor Rehabilitation Chair with Non-Invasive Therapy
- Hospital Approved Pelvic Muscle Stimulation Chair for Physiotherapy
- Professional Pelvic Health Rehabilitation Chair with Non-Invasive Technology
- Pelvic Floor Therapy Chair for Non-Invasive Incontinence Treatment
- Professional Pelvic Muscle Strengthening Chair for Medical Centers
- Digital Pelvic Floor Muscle Activation Chair for Pelvic Health Care
- Smart Clinical Pelvic Floor Therapy Chair with Automated Programs
- Pelvic Floor Muscle Conditioning Chair for Womens & Mens Health
- Medical Pelvic Muscle Training Chair for Core & Pelvic Stability
- Advanced Pelvic Floor Muscle Therapy Chair for Rehabilitation Hospitals
- Pelvic Muscle Recovery & Strength Training Chair for Clinical Practice
- Professional Pelvic Floor Rehabilitation Chair for Long-Term Care
- Digital Pelvic Muscle Therapy Chair for Post-Operative Recovery
- Pelvic Floor Muscle Stimulation Chair for Complete Pelvic Wellness
- Medical Grade Pelvic Muscle Stimulation Chair for Pelvic Health Clinics
- Smart Pelvic Floor Muscle Stimulation Chair for Rehabilitation Centers
- Pelvic Floor Muscle Recovery Chair for Post-Surgery Rehabilitation
- Advanced Pelvic Muscle Activation Chair for Womens & Mens Health
- Medical Pelvic Floor Therapy Chair for Complete Pelvic Rehabilitation
- Digital Clinical Pelvic Muscle Training Chair with Automated Programs
- Pelvic Floor Muscle Conditioning Chair for Long-Term Rehabilitation
- Professional Pelvic Muscle Therapy Chair for Advanced Physiotherapy
- Clinical Pelvic Floor Muscle Stimulation Chair for Advanced Rehabilitation
- Advanced Pelvic Muscle Training Chair for Physiotherapy & Recovery
- Professional Pelvic Floor Muscle Activation Chair for Clinical Practice
- Hospital Grade Pelvic Muscle Therapy Chair for Physiotherapy Centers
- Smart Pelvic Floor Rehabilitation Chair with Automated Treatment Programs
- Clinical Pelvic Floor Muscle Training Chair for Post-Treatment Recovery
- Advanced Pelvic Muscle Therapy Chair for Medical & Rehab Facilities
- Pelvic Floor Strength & Control Training Chair for Clinical Use
- Advanced Clinical Pelvic Floor Therapy Chair for Muscle Rehabilitation
- Pelvic Muscle Training & Stimulation Chair for Physiotherapy Clinics
- Smart Pelvic Floor Muscle Therapy Chair for Rehabilitation Centers
- Hospital Approved Pelvic Floor Muscle Training Chair for Therapy Clinics
- Pelvic Muscle Recovery Chair for Complete Pelvic Health Management
- Professional Pelvic Floor Rehab Chair for Advanced Patient Care
- Advanced Pelvic Floor Muscle Rehabilitation Chair for Clinical Therapy
- Non-Invasive Pelvic Floor Therapy Chair for Muscle Strength Recovery
- Digital Pelvic Floor Muscle Rehab Chair for Incontinence Management
- Hospital Grade Pelvic Muscle Therapy Chair for Patient Rehabilitation
- Smart Pelvic Floor Muscle Training Chair for Clinical Practice
- Advanced Pelvic Muscle Conditioning Chair for Physiotherapy Centers
- Professional Pelvic Muscle Rehab Chair for Long-Term Patient Care
- Medical Grade Pelvic Muscle Strength Training Chair for Physiotherapy
- Smart Clinical Pelvic Floor Muscle Training Chair for Medical Use
- Pelvic Muscle Strengthening Therapy Chair for Womens & Mens Health
- Non-Surgical Pelvic Floor Muscle Therapy Chair for Clinical Practice
- Pelvic Floor Muscle Conditioning Chair for Post-Surgery Recovery
- Non-Invasive Pelvic Floor Muscle Training Chair for Clinical Programs
- Digital Clinical Pelvic Muscle Training Chair with Intelligent Programs
- Medical Grade Pelvic Muscle Therapy Chair for Physiotherapy Centers
- Professional Pelvic Muscle Rehabilitation Chair for Advanced Therapy
- Digital Pelvic Floor Muscle Strength Training Chair for Pelvic Health
- Hospital Grade Pelvic Muscle Activation Chair for Clinical Use
- Advanced Pelvic Floor Muscle Therapy Chair For Rehabilitation Hospitals
- Pelvic Floor Muscle Strengthening Chair For Comprehensive Pelvic Care
- Clinical Benefits of Using Exoskeletons in Rehabilitation Centers
- Medical Exoskeleton Systems for Professional Physiotherapy Use
- Robotic Exoskeleton Systems Supporting Functional Independence
- Advanced Exoskeleton Rehabilitation Systems for Clinical Use
- Exoskeleton-Assisted Recovery for Improved Joint Mobility
- Medical Exoskeleton Solutions for Assisted Physiotherapy
- Exoskeleton Technology Enhancing Modern Rehabilitation Practice
- Exoskeleton-Assisted Training for Muscle Activation and Balance
- Medical Exoskeleton Systems for Neuro and Ortho Therapy
- Exoskeleton Technology for Structured Physiotherapy Programs
- Exoskeleton Therapy Systems for Neurological Motor Recovery
- Advanced Robotic Exoskeletons for Clinical Rehabilitation
- Exoskeleton Systems Enhancing Strength Endurance and Coordination
- Medical Exoskeleton Technology for Assisted Motor Training
- Robotic Wearable Exoskeletons for Safe Movement Assistance
- Exoskeleton Therapy Devices for Strength Balance and Control
- Exoskeleton Rehabilitation Systems for Long-Term Motor Training
- Exoskeleton Rehabilitation Systems for Safe Movement Control
- Wearable Medical Exoskeletons for Long-Term Therapy Programs
- Exoskeleton Therapy Technology for Targeted Motor Recovery
- Exoskeleton Technology Enhancing Modern Rehabilitation
- Medical Exoskeletons for Functional Mobility Support
- Wearable Exoskeleton Devices for Assisted Strength Training
- Advanced Exoskeleton Therapy Devices for Rehabilitation Clinics
- Wearable Exoskeleton Devices for Assisted Functional Training
- Smart Medical Exoskeletons for Precision Rehabilitation
- Phymaster Shockwave Professional Shockwave Therapy Machine
- Phymaster Shockwave Advanced Shockwave Therapy Equipment
- Phymaster Shockwave Physiotherapy System for Fast Pain Recovery
- Phymaster Shockwave Shockwave Therapy Machine for Pain Management
- Phymaster Shockwave Therapy Equipment for Non-Surgical Pain Treatment
- Phymaster Shockwave Physiotherapy Device for Orthopedic Rehabilitation
- Phymaster Shockwave Shockwave Therapy System for Orthopedic Clinics
- Phymaster Shockwave Physiotherapy Device for Joint & Muscle Pain
- Phymaster Shockwave Shockwave Therapy Machine for Rehabilitation Use
- Phymaster Shockwave Shockwave Therapy Device for Sports & Ortho Pain
- Phymaster Shockwave Professional Shockwave Pain Therapy Equipment
- Phymaster Pain Relief Device for Physiotherapy Treatment
- Phymaster Shockwave Therapy Machine for Muscle & Joint Rehabilitation
- Phymaster Shockwave Professional Physiotherapy Shockwave Equipment
- Phymaster Shockwave Physiotherapy Machine for Advanced Rehabilitation
- Phymaster Shockwave Pain Management Shockwave Therapy Device
- Phymaster Shockwave Pain Relief Physiotherapy Equipment
- Phymaster Shockwave Physiotherapy Device for Clinical Rehabilitation
- Phymaster Advanced Pain Relief Shockwave Therapy System
- Phymaster Shockwave Professional Shockwave Therapy System
- Phymaster Therapy Device for Sports Rehabilitation
- Phymaster Shockwave Shockwave Therapy Equipment for Pain Relief Treatment
- Phymaster Shockwave Shockwave Therapy Device for Orthopedic Pain Care
- Phymaster Shockwave Therapy System for Rehab Centers
- Phymaster Shockwave Therapy Machine for Joint Pain & Mobility Improvement
- Phymaster Shockwave Physiotherapy Equipment for Sports Injury Rehabilitation
- Phymaster Shockwave Therapy Equipment for Clinical Recovery
- Phymaster Shockwave Physiotherapy Device for Advanced Rehabilitation
- Phymaster Shockwave Therapy Machine for Sports Injury Rehabilitation
- Phymaster Shockwave Pain Relief Therapy Device
- Phymaster Shockwave Advanced Physiotherapy Equipment
- Phymaster Shockwave Physiotherapy Machine for Professional Clinical Use
- Phymaster Shockwave Physiotherapy Machine for Chronic Pain Relief
- Phymaster Shockwave Pain Management Physiotherapy System
- Phymaster Shockwave Shockwave Therapy Device for Physiotherapy Clinics
- Phymaster Shockwave Physiotherapy Equipment for Joint Pain Care
- Phymaster Shockwave Advanced Shockwave Therapy Machine
- Phymaster Shockwave Physiotherapy System for Muscle Recovery Therapy
- Phymaster Therapy Machine for Orthopedic Rehabilitation
- Phymaster Shockwave Therapy Equipment for Sports Clinics
- Phymaster Shockwave Advanced Pain Therapy Shockwave Equipment
- Phymaster Shockwave Pain Management Therapy Machine for Clinics
- Phymaster Shockwave Therapy Machine for Joint & Muscle Pain Care
- Phymaster Shockwave Professional Shockwave Therapy Device
- Phymaster Shockwave Physiotherapy System for Orthopedic Pain Relief
- Digital Neuromodulation Therapy Equipment With Vagus Nerve Stimulation
- Multi Function Brainwave And Vagus Stimulator Machine
- Digital Transcranial And Vagus Nerve Therapy Device
- Neuro Plasticity Enhancement Therapy Equipment
- Portable Multi Mode Transcranial Therapy Machine
- Brainwave Synchronization And Vagus Activation Device
- Multi Channel Neuro Stimulation Therapy Equipment
- Medical Grade TDCS TACS TPCS TVNS Machine
- Digital Brain And Autonomic Nervous System Modulator
- Research And Clinical Neuro Stimulation Device
- Bio VNS-1 Multi Therapy Brain Stimulation Equipment
- Clinical Brainwave And Vagus Therapy Machine
- Digital TDCS And TVNS Therapy Console
- Advanced Neuro Rehabilitation Stimulation Machine
- Precision Brain And Autonomic Therapy Device
- Professional Brain And Vagus Activation Machine
- Medical Grade Multi Mode Brain Stimulator
- Digital Neuro Activation And Recovery Equipment
- Multi Waveform Brain Stimulation Machine
- Research And Clinical Neuro Modulation Equipment
- Advanced Electrical Brain Modulation Machine
- Multi Functional Brain And Nerve Therapy Device
- Smart Controlled Neurological Stimulation Equipment
- Advanced Brain Rehabilitation And Therapy Console
- Medical Brainwave Synchronization System
- Professional TDCS And TVNS Therapy Equipment
- Research Grade Brainwave And Vagus Stimulation Unit
- Digital Vagus Nerve And Brain Stimulator
- Integrated Neuro Rehab And Brainwave Therapy System
- Bio VNS-1 Advanced Multi Mode Brain Stimulation System
- Professional Transcranial And Vagus Therapy Device
- Clinical Brain And Vagus Nerve Stimulation Equipment
- Hospital Grade Non Invasive Brain Stimulator
- Research Grade Multi Mode Neuro Stimulation Unit
- Clinical TDCS TACS And TVNS Therapy System
- Professional Neuromodulation And Rehab Device
- Advanced Brain Plasticity Enhancement Machine
- Digital Neurological Recovery And Therapy Equipment
- Multi Mode Neuro Stimulation Device
- Clinical Grade Transcranial And Vagus Nerve Therapy Equipment
- Advanced Digital Neuromodulation
- Multi Therapy Brain Stimulation Equipment
- Professional Neuro Activation And Rehab Machine
- Digital Brainwave Synchronization Therapy Device
- Advanced Electrical Cortical And Neural Modulation System
- Clinical Neurological Recovery And Brain Stimulation Machine
- Advanced Brain Plasticity Enhancement And Cognitive Rehab System
- Integrated Multi Protocol Neuro Stimulation And Therapy Machine
- Hospital Use Brain And Cognitive Enhancement Machine
- Hospital Grade Multi Mode Brain Stimulation Equipment
- Professional Brain And Nerve Modulation Therapy Equipment
- Complete Multi Mode Neurological Stimulation And Rehab Device
- Smart Brain And Vagus Stimulation Therapy Machine
- Medical Neuromodulation And Brainwave Regulation Equipment
- Integrated Brain Recovery And Neuro Activation Therapy System
- Brainwave And Vagus Activation Therapy Equipment
- Precision Neurological Modulation And Therapy Machine
- Multi Protocol Brainwave Regulation And Therapy Equipment
- Medical Grade Transcranial And Vagus Nerve Stimulator
- Advanced Brain Plasticity And Cognitive Enhancement Therapy System
- Integrated Multi Mode Brain Stimulation And Recovery Machine
- Professional Brainwave And Autonomic Nerve Modulation System
- Clinical TDCS TPCS TACS TVNS Neuro Rehabilitation Equipment
- Digital Transcranial Direct And Alternating Current Therapy Machine
- Bio VNS-1 Advanced Multi Channel Brain And Vagus Nerve Stimulation System
- Professional Multi Therapy Brain And Nerve Stimulation Equipment
- Advanced Electrical Brain And Vagus Rehabilitation Device
- Medical Brain And Cognitive Rehabilitation Equipment
- Smart Digital Brainwave And Vagus Therapy Machine
- Advanced Transcranial Brain Stimulation And Rehab System
- Complete Neuromodulation And Neural Therapy Equipment
- Advanced Transcranial Direct And Pulse Current Therapy Device
- Professional Brain And Nerve Recovery Stimulation Machine
- Advanced Multi Channel Neuromodulation And Rehab System
- Hospital Grade Brain And Cognitive Recovery Device
- Precision Brainwave And Neural Activation Therapy Machine
- Integrated Brain And Neural Recovery Modulation Machine
- Professional Multi Mode Brain And Nerve Therapy System
- Precision Controlled Neurological Stimulation And Modulation Machine
- Multi Channel Brain And Vagus Nerve Activation Device
- Advanced Cortical And Autonomic Nervous System Modulation Equipment
- Hospital Use Brainwave Modulation And Rehab Device
- Hospital Grade Multi Protocol Brain Stimulation And Rehab System
- Hospital Grade Neurological Activation And Recovery Machine
- Digital Brainwave And Neural Plasticity Therapy System
- Integrated Neuro Stimulation And Brain Recovery System
- Professional Brain And Autonomic Nervous System Activation Device
- Hospital Grade Non Invasive Brain And Neural Therapy System
- Integrated Multi Protocol Neurological Stimulation And Recovery Machine
- Medical Grade Transcranial Direct And Alternating Current Stimulator
- Precision Controlled Brainwave And Neural Modulation Equipment
- Hospital Use Brainwave And Cognitive Rehabilitation System
- Integrated Brain And Neural Activation Therapy Machine
- Medical Neuromodulation And Brain Recovery Equipment
- Advanced Electrical Brainwave Modulation And Rehab Device
- Complete Multi Mode Neurological Therapy And Stimulation Machine
- Digital Neuro Activation And Brainwave Control System
- Advanced Brain Plasticity Enhancement And Stroke Therapy Machine
- Precision Controlled Transcranial And Vagus Nerve Stimulation Device
- Smart Brain And Autonomic Regulation Therapy Machine
- Complete Brain Stimulation And Neuro Recovery Therapy System
- Professional Brainwave And Vagus Activation Equipment
- Digital Multi Therapy Neurological Stimulation Machine
- Precision Brainwave Modulation And Rehab Equipment
- Hospital Grade Non Invasive Brain Activation And Therapy System
- Advanced Multi Protocol Neurological Therapy And Stimulation Machine
- Medical Grade Brain Plasticity Enhancement And Rehab System
- Multi Mode Brain And Neural Cognitive Therapy Device
- Precision Controlled Brainwave And Cortical Stimulation System
- Integrated Neuro Activation And Brain Therapy System
- Hospital Use Brain And Cognitive Enhancement Device
- Digital TDCS And TVNS Brain Modulation System
- Clinical Brain Recovery And Neuro Activation Equipment
- Advanced Cortical Modulation And Rehab Machine
- Integrated TDCS TPCS TACS TVNS Brain Stimulation Equipment
- Precision Controlled Neurological Stimulation Equipment
- Professional Brain And Vagus Modulation Therapy System
- Hospital Use Brain And Nerve Stimulation System
- Complete Brainwave And Vagus Nerve Activation Equipment
- Brainwave And Vagus Nerve Activation Equipment
- Advanced Brainwave And Vagus Nerve Stimulation System
- Clinical Brainwave Therapy And Vagus Nerve Stimulation Equipment
- Professional Brainwave Control And Vagus Nerve Therapy Machine
- PEMT Pulse Electromagnetic Loop Therapy Device for Physiotherapy
- Premium PEMT Loop Therapy Machine with Touch Screen
- PEMT Loop Therapy Physiotherapy Equipment Premium Model
- Electromagnetic Pulse Therapy Loop Device for Pain Management
- PEMT Pulse Electromagnetic Therapy Machine Premium Model
- Pulse Electromagnetic Loop Therapy Device for Orthopedic Pain Relief
- PEMF Pulse Electromagnetic Therapy Device with Loop Applicator
- Professional PEMT Loop Therapy Machine for Physiotherapy Treatment
- Pulse Electromagnetic Therapy Machine for Pain Relief Therapy
- PEMT Pulse Electromagnetic Therapy Loop Device Premium Model
- Electromagnetic Pulse Therapy Loop Machine Clinical Model
- PEMF Pulse Electromagnetic Therapy Device for Orthopedic Clinics
- Pulse Electromagnetic Therapy Loop Equipment for Rehabilitation
- PEMT Physiotherapy Loop Therapy Device Premium Model
- PEMT Pulse Electromagnetic Therapy Machine for Rehabilitation
- Electromagnetic Pulse Therapy Loop Device Physiotherapy Equipment
- Pulse Electromagnetic Therapy Equipment for Orthopedic Pain Relief
- Pulse Electromagnetic Therapy Loop Device for Rehabilitation Centers
- Advanced PEMF Loop Therapy Device Premium Physiotherapy Model
- PEMF Loop Therapy Machine Clinical Physiotherapy Equipment
- Professional PEMT Loop Therapy Device for Pain Management
- Premium PEMT Pulse Electromagnetic Loop Therapy Device for Physiotherapy
- PEMT Pulse Electromagnetic Therapy Loop Device for Pain Relief Treatment
- PEMT Loop Therapy Device for Physiotherapy Clinics
- Premium PEMT Pulse Electromagnetic Therapy Machine
- Electromagnetic Therapy Equipment for Physiotherapy Centers
- PEMT Loop Therapy Machine for Pain Management Therapy
- Electromagnetic Pulse Therapy Device Loop Applicator Model
- PEMF Pulse Electromagnetic Therapy Machine for Physiotherapy
- Advanced PEMT Pulse Electromagnetic Therapy Machine
- PEMF Loop Therapy Device for Physiotherapy and Rehabilitation
- Pulse Electromagnetic Therapy Machine for Orthopedic Therapy
- Professional PEMF Pulse Electromagnetic Therapy Device
- Pulse Electromagnetic Therapy Device Loop Type Physiotherapy Equipment
- PEMT Loop Therapy Machine for Joint and Muscle Pain Relief
- Advanced PEMT Loop Therapy Device for Pain Management
- Pulse Electromagnetic Therapy Loop Device for Pain Management
- Class 4 Laser Therapy Device for Orthopedic Rehabilitation
- Class IV Laser Therapy Equipment for Muscle and Joint Pain
- High Intensity Class 4 Laser Therapy Device for Rehabilitation
- High Power Class IV Laser Therapy Equipment for Clinics
- Economical Class 4 Laser Therapy Machine for Orthopedic Therapy
- Medical Grade Class IV Laser Therapy Device for Pain Management
- Laser Physiotherapy Equipment for Joint Pain Therapy
- Professional Class 4 Laser Therapy Machine for Sports Rehabilitation
- High Power Class IV Laser Therapy System for Physiotherapy Centers
- Economical Class 4 Laser Therapy Device for Physiotherapy Centers
- Professional Class IV Laser Therapy System for Medical Rehabilitation
- Advanced Class 4 Laser Therapy Equipment for Rehabilitation Centers
- High Power Class 4 Laser Therapy Equipment for Physiotherapy Centers
- Advanced Class IV Laser Therapy Machine for Orthopedic Clinics
- Economical Class 4 Laser Therapy Machine for Physiotherapy Centers
- Professional Class IV Laser Therapy Equipment for Pain Relief Clinics
- Economical Model Class 4 Laser Therapy Equipment for Medical Use
- Economical Class 4 Laser Therapy Equipment for Physiotherapy Pain Relief
- Class IV Laser Therapy Device for Sports Injury Pain Treatment
- Class IV Laser Therapy System for Physiotherapy Clinics
- Advanced Class IV Laser Therapy Equipment for Rehabilitation Centers
- High Intensity Class 4 Laser Physiotherapy Device for Rehabilitation Centers
- Economical Class 4 Laser Therapy Device for Pain Relief and Rehabilitation
- Professional Class 4 Laser Physiotherapy Device for Pain Relief Treatment
- High Intensity Class 4 Laser Physiotherapy Device for Orthopedic Rehabilitation
- Portable 4 in 1 Combo Therapy Machine for Pain Relief & Rehab
- 4 in 1 Combo Therapy Equipment for Muscle & Nerve Treatment
- Advanced 4 in 1 Physiotherapy Device for Pain Management Use
- Brain Stimulation Device tDCS tPCS tACS tVNS
- Clinical Neuro Therapy Equipment tDCS tPCS tACS tVNS
- Professional Brain Therapy Device tDCS tACS tVNS tPCS
- High Performance Brain Therapy Machine tDCS tPCS tACS tVNS
- Electro Brain Therapy Machine tDCS tACS tVNS tPCS
- Electro Neuro Rehab Device tDCS tPCS tACS tVNS
- Professional Brain Rehab Device tDCS tPCS tACS tVNS
- Digital Neuro Therapy Equipment tDCS tACS tVNS tPCS
- Neuro Rehab Equipment tDCS tPCS tACS tVNS Device
- Brain Stimulation Equipment tDCS tACS tVNS tPCS
- High Efficiency Neuro Stimulation Device tDCS tPCS tACS tVNS
- Multi-Mode Neuro Therapy Machine tDCS, tACS, tVNS, tPCS
- Clinical Neuro Rehab Machine tDCS tPCS tACS tVNS Equipment
- High Performance Brain Stimulation Device tDCS tACS tVNS tPCS
- Smart Neuro Therapy Equipment tDCS tPCS tACS tVNS System
- Digital Neuro Rehab Equipment tDCS tPCS tACS tVNS Machine
- Clinical Brain Stimulation Equipment tDCS tACS tVNS tPCS
- Neuro Brain Therapy Device tDCS tPCS tACS tVNS Equipment
- tDCS tACS tPCS tVNS Brain and Nerve Stimulation Therapy Device
- Advanced Neuro Stimulation System tDCS tPCS tACS tVNS Device
- Advanced Brain and Neuro Therapy Device tDCS tACS tVNS tPCS
- Portable Neuro Modulation Therapy Machine tDCS tPCS tACS tVNS
- Professional Neuro Modulation Machine tDCS tACS tVNS tPCS Device
- Advanced Brain Neuro Therapy Machine tDCS tACS tVNS tPCS
- Konmed KM-560 Pelvic Rehab Biofeedback Device with EMG Output
- Konmed KM-560 Biofeedback System with Electrical Muscle Stimulation
- Konmed KM-560 EMG Biofeedback Device with NMES FES for Muscle Rehab
- Konmed KM-560 Physiotherapy EMG Biofeedback with NMES
- Konmed KM-560 4 Channel EMG Biofeedback Device for Therapy
- Konmed KM-560 EMG Biofeedback Machine for Sports Rehab
- Konmed KM-560 EMG Biofeedback Device with Multi Therapy
- Konmed KM-560 4 Channel Biofeedback System for Therapy
- Konmed KM-560 Pelvic Rehab Biofeedback Machine with EMG
- Konmed KM-560 4 Channel Physiotherapy Biofeedback Stimulation
- Konmed KM-560 EMG Biofeedback Device with FES Therapys
- Konmed KM-560 Clinical EMG Biofeedback Device with Gamings
- Konmed KM-560 EMG Biofeedback Machine for Clinical Uses
- Konmed KM-560 EMG Biofeedback System
- Konmed KM-560 EMG Biofeedback Machine with ETS Triggers
- Konmed KM-560 Professional Biofeedback Device
- Konmed KM-560 Rehab Biofeedback Equipment with EMGS
- Konmed KM-560 Clinical EMG Biofeedback Device with FES
- Konmed KM-560 Physiotherapy Biofeedback Devices
- Konmed KM-560 Biofeedback Device with Electrical Therapy
- Konmed KM-560 4 Channel EMG Biofeedback System for hospital Therapy
- Konmed KM-560 Physiotherapy Biofeedback Device with ETS Modes
- Konmed KM-560 EMG Biofeedback System for Muscle Strengthening
- Konmed KM-560 EMG Biofeedback System for Recovery Training
- Konmed KM-560 Biofeedback Equipment with Advanced Therapy
- Konmed KM-560 Clinical Biofeedback Device with Muscle Rehab
- Konmed KM-560 Professional Biofeedback Machine with Advanced Modes
- Konmed KM-560 EMG Biofeedback Stimulator with Therapy Settings
- Konmed KM-560 4 Channel Biofeedback System with Muscle Activation
- Konmed KM-560 4 Channel Physiotherapy Biofeedback mahine
- Konmed KM-560 4 Channel EMG Biofeedback Device for Therapy Use
- Konmed KM-560 EMG Biofeedback System for Strength Recovery
- Konmed KM-560 Physiotherapy Biofeedback
- Konmed KM-560 Professional Biofeedback Equipment with Rehab Programs
- Konmed KM-560 EMG Biofeedback Device with Muscle Strength Programs
- Konmed KM-560 Pelvic Floor Biofeedback Machine with EMG Feedback
- Konmed KM-560 Advanced EMG Therapy Device with Biofeedback Technology
- Konmed KM-560 EMG Biofeedback Equipment for Muscle Re-Education
- Konmed KM-560 Multi Therapy Biofeedback Machine for Muscle Recovery
- Konmed KM-560 Portable Physiotherapy Device with EMG Biofeedback
- Konmed KM-560 EMG Biofeedback Therapy Machine for Advanced Rehabilitation
- Konmed KM-560 EMG Based Therapy Device with Multi Channel Output
- Women Pelvic Muscle Exerciser for Bladder Control and Strength
- Pelvic Muscle Trainer for Postpartum and Incontinence Care
- Kegel Exercise Trainer for Women Pelvic Health and Fitness
- Advanced Pelvic Biofeedback Device KM533-4 with Wireless Monitoring & App Control
- Wireless EMG Biofeedback Pelvic Floor Trainer KM-533-4 with ETS & App Support
- Portable 4 in 1 Physiotherapy Combo Device with TENS EMS IFT Therapy Modes
- KM-533-4 Pelvic Kegel Toner with EMG Biofeedback for Urinary Incontinence
- App Based Pelvic Floor Rehabilitation Device KM-533-4 with EMG & ETS
- KM533-4 Wireless Pelvic Muscle Trainer with Biofeedback for Bladder Control
- EMG Biofeedback Kegel Exercise Device KM-533-4 for Women Health
- 4 in 1 Combo Physiotherapy Unit with TENS EMS IFT NMES Therapy
- Clinical 4 in 1 Electrotherapy Device for Pain Management & Muscle Recovery
- KM-533-4 Clinical Pelvic Floor Rehabilitation System with EMG & ETS
- Advanced Pelvic Biofeedback Device KM533-4 with Wireless App Control
- Physiotherapy Pelvic Muscle Trainer KM-533-4 with EMG Biofeedback
- KM-533-4 Pelvic Rehabilitation Machine for Incontinence Therapy
- Medical Grade Pelvic Floor Trainer KM533-4 with Smart Biofeedback
- Professional Electrotherapy Combo Machine 4 in 1 Pain Relief Device
- 4 in 1 Digital Physiotherapy Equipment for Nerve & Muscle Stimulation
- Portable Electrotherapy Combo Unit with Multiple Therapy Modes
- 4 in 1 Physiotherapy Machine for Pain Relief Muscle Rehab & Therapy
- KM533-4 Smart Kegel Trainer with EMG Biofeedback & Mobile App
- Wireless Pelvic Floor Trainer KM-533-4 for Home and Clinic Use
- KM-533-4 Pelvic Muscle Strengthening Device with Real-Time Feedback
- Smart Pelvic Therapy Device KM533-4 for Bladder Control Training
- KM-533-4 Kegel Exercise Machine with EMG Biofeedback & ETS
- KM-533-4 EMG Pelvic Trainer with App Based Monitoring
- Wireless Kegel Trainer KM533-4 Biofeedback Device
- KM-533-4 Pelvic Floor Therapy Machine with ETS Mode
- EMG Biofeedback Pelvic Device KM533-4 for Rehabilitation
- Smart Pelvic Trainer KM-533-4 for Incontinence Care
- KM-533-4 Pelvic Floor Strengthening Device with EMG Technology
- Wireless Pelvic Biofeedback Trainer KM533-4 for Muscle Recovery
- KM-533-4 App Based Kegel Trainer with EMG Monitoring
- Pelvic Muscle Rehabilitation Device KM-533-4 with Smart Feedback
- KM533-4 Advanced Kegel Therapy Device for Women Health
- KM-533-4 Wireless EMG Pelvic Floor Trainer with ETS for Kegel Therapy
- Smart Pelvic Biofeedback Device KM533-4 with App for Muscle Training
- KM-533-4 Pelvic Rehabilitation Trainer with EMG Feedback & ETS Mode
- Wireless Kegel Exercise Device KM533-4 for Pelvic Muscle Strengthening
- KM-533-4 EMG Biofeedback Trainer for Bladder Control and Recovery
- Advanced Pelvic Floor Therapy Device KM533-4 with Smart App Integration
- KM-533-4 Kegel Toner with EMG Feedback for Women Health Therapy
- Wireless Pelvic Muscle Rehab Device KM533-4 with ETS Technology
- KM-533-4 Pelvic Biofeedback Machine for Physiotherapy and Rehabilitation
- Smart Kegel Training Device KM533-4 with Real-Time EMG Monitoring
- KM-533-4 Pelvic Muscle Trainer with ETS and Biofeedback Technology
- Wireless EMG Kegel Device KM533-4 for Incontinence Management
- KM-533-4 App Controlled Pelvic Floor Trainer with Smart Feedback
- Pelvic Rehabilitation System KM533-4 with EMG Biofeedback & ETS Mode
- KM-533-4 Kegel Therapy Device with Wireless EMG Monitoring
- Smart Pelvic Muscle Trainer KM533-4 for Home Physiotherapy Use
- KM-533-4 EMG Pelvic Trainer for Muscle Re-Education Therapy
- Wireless Pelvic Floor Strengthener KM533-4 with Biofeedback Support
- KM-533-4 Advanced Pelvic Therapy Machine with ETS and EMG
- Smart Pelvic Rehabilitation Device KM533-4 with Mobile App Support
- KM-533-4 EMG Biofeedback Kegel Trainer for Women Recovery
- Wireless Pelvic Muscle Strengthening Device KM533-4 with ETS
- KM-533-4 Pelvic Floor Trainer for Postpartum Recovery Therapy
- Smart Kegel Therapy Device KM533-4 with Real-Time Muscle Feedback
- KM-533-4 Pelvic Muscle Rehab System with EMG and App Connectivity
- KM-533-4 Wireless Pelvic Floor Trainer with EMG Biofeedback and ETS Mode
- Advanced Kegel Trainer KM533-4 with Smart App and Biofeedback System
- KM-533-4 Pelvic Muscle Therapy Device with EMG Monitoring for Rehabilitation
- Wireless EMG Biofeedback Kegel Trainer KM533-4 for Bladder Control Therapy
- KM-533-4 Smart Pelvic Floor Strengthening Device with ETS Technology
- Professional Pelvic Rehab Machine KM533-4 with EMG Biofeedback Support
- KM-533-4 Pelvic Muscle Trainer for Women with App Based Monitoring System
- Wireless Pelvic Therapy Device KM533-4 with Real-Time EMG Feedback
- KM-533-4 Advanced Kegel Trainer with EMG and ETS Technology
- Smart Pelvic Floor Therapy System KM533-4 with ETS and EMG Technology
- KM-533-4 Wireless Kegel Exercise Device with Smart Biofeedback Support
- Pelvic Muscle Rehabilitation Trainer KM533-4 with EMG Feedback System
- KM-533-4 Advanced Pelvic Biofeedback Machine with App Connectivity
- Wireless EMG Pelvic Trainer KM533-4 for Physiotherapy and Recovery
- KM-533-4 Smart Kegel Toner with Biofeedback for Pelvic Muscle Strengthening
- Pelvic Floor Strengthening Machine KM533-4 with EMG and ETS Features
- KM-533-4 EMG Biofeedback Device for Pelvic Health and Rehabilitation
- Wireless Pelvic Muscle Trainer KM533-4 with Smart Monitoring Technology
- KM-533-4 Kegel Therapy System with EMG Biofeedback and App Support
- Smart Pelvic Rehab Trainer KM533-4 for Home and Clinical Use
- KM-533-4 Pelvic Floor Trainer for Urinary Incontinence and Recovery
- Wireless Kegel Trainer KM533-4 with EMG Biofeedback Technology
- KM-533-4 Pelvic Muscle Strengthening Device with Smart ETS Mode
- Advanced Pelvic Therapy Device KM533-4 with Wireless Biofeedback
- KM-533-4 Smart Pelvic Muscle Rehab System with EMG Monitoring
- KM-533-4 Wireless EMG Pelvic Trainer with Smart ETS Biofeedback System
- Advanced Pelvic Floor Kegel Device KM533-4 with App Based Control
- KM-533-4 Pelvic Muscle Rehabilitation Device with Wireless EMG Feedback
- Smart EMG Biofeedback Kegel Trainer KM533-4 for Bladder Control Therapy
- KM-533-4 Wireless Pelvic Floor Therapy Machine with ETS Function
- Professional Kegel Training Device KM533-4 with Real-Time Biofeedback
- KM-533-4 Pelvic Muscle Strengthening Trainer with App Monitoring
- Wireless Pelvic Rehab Device KM533-4 with EMG Biofeedback Technology
- KM-533-4 Smart Pelvic Floor Trainer for Women Health and Recovery
- Advanced EMG Kegel Therapy Device KM533-4 with ETS and App Support
- KM-533-4 Pelvic Biofeedback Trainer with Wireless Smart Control
- Smart Pelvic Muscle Therapy Device KM533-4 with EMG Monitoring
- KM-533-4 Kegel Exercise Trainer with Biofeedback and ETS Mode
- Wireless EMG Pelvic Floor Strengthener KM533-4 for Rehabilitation
- KM-533-4 Pelvic Muscle Recovery Device with Smart App Integration
- Advanced Pelvic Rehab System KM533-4 with EMG Biofeedback Technology
- KM-533-4 Smart Kegel Trainer for Pelvic Strength and Incontinence Care
- Wireless Pelvic Therapy Trainer KM533-4 with Real-Time EMG Tracking
- KM-533-4 Pelvic Floor Exercise Device with ETS and Biofeedback System
- Smart Pelvic Muscle Trainer KM533-4 with Wireless App Connectivity
- KM-533-4 EMG Biofeedback Therapy Device for Pelvic Rehabilitation
- Wireless Kegel Therapy Machine KM533-4 with Smart Monitoring System
- KM-533-4 Pelvic Floor Trainer with Advanced EMG and ETS Features
- Smart Pelvic Rehab Device KM533-4 with Biofeedback and App Support
- KM-533-4 Wireless Kegel Trainer for Pelvic Muscle Strengthening
- KM-533-4 Smart EMG Pelvic Floor Trainer with Wireless App Control
- Advanced Wireless Kegel Device KM533-4 with ETS Biofeedback System
- KM-533-4 Pelvic Muscle Therapy Trainer with Real-Time EMG Feedback
- Smart Pelvic Floor Rehabilitation Device KM533-4 with App Monitoring
- KM-533-4 Wireless Biofeedback Kegel Trainer for Bladder Control Support
- Professional Pelvic Therapy Machine KM533-4 with EMG and ETS Technology
- KM-533-4 Pelvic Floor Strengthening Trainer with Smart Biofeedback
- Wireless EMG Pelvic Rehab Device KM533-4 with App Based Tracking
- KM-533-4 Smart Kegel Exercise Device with Biofeedback and ETS Mode
- Advanced Pelvic Muscle Trainer KM533-4 with Wireless Monitoring System
- KM-533-4 Pelvic Biofeedback Therapy Device for Women Health Recovery
- Smart Pelvic Floor Trainer KM533-4 with EMG Monitoring and App Support
- KM-533-4 Wireless Kegel Therapy Device with Real-Time Biofeedback
- Pelvic Muscle Rehab System KM533-4 with EMG and Smart ETS Function
- KM-533-4 Advanced Pelvic Trainer with Biofeedback and App Connectivity
- Wireless Pelvic Muscle Strengthener KM533-4 with Smart Feedback System
- KM-533-4 EMG Kegel Trainer for Pelvic Health and Incontinence Therapy
- Smart Pelvic Therapy Trainer KM533-4 with Wireless Biofeedback Support
- KM-533-4 Pelvic Floor Exercise Machine with EMG and ETS Technology
- Advanced Wireless Pelvic Rehab Device KM533-4 with Smart Monitoring
- KM-533-4 Smart Biofeedback Kegel Device for Muscle Re-Education
- Wireless Pelvic Trainer KM533-4 with EMG Biofeedback and App System
- KM-533-4 Pelvic Rehabilitation Trainer with Advanced ETS and EMG
- Smart Pelvic Muscle Therapy Machine KM533-4 with App Integration
- KM-533-4 Wireless EMG Pelvic Floor Trainer for Clinical and Home Use
- KM-533-4 Wireless Pelvic Biofeedback Trainer with Smart ETS Control
- Advanced EMG Pelvic Floor Device KM533-4 with App Based Monitoring
- KM-533-4 Kegel Therapy Trainer with Wireless Biofeedback Technology
- Smart Pelvic Muscle Rehab Device KM533-4 with Real-Time EMG Feedback
- KM-533-4 Pelvic Floor Strengthening System with ETS and App Support
- Wireless Kegel Trainer KM533-4 with Smart EMG Biofeedback System
- KM-533-4 Pelvic Therapy Device with Advanced Biofeedback and ETS Mode
- Smart Pelvic Rehabilitation Trainer KM533-4 with Wireless Monitoring
- KM-533-4 EMG Biofeedback Pelvic Trainer for Muscle Recovery Therapy
- Wireless Pelvic Muscle Strength Trainer KM533-4 with Smart App Control
- KM-533-4 Pelvic Floor Exercise Trainer with EMG and ETS Technology
- Advanced Pelvic Rehab Device KM533-4 with Biofeedback and App System
- KM-533-4 Smart Kegel Therapy Device with Wireless EMG Monitoring
- Wireless Pelvic Muscle Therapy Trainer KM533-4 with Real-Time Feedback
- KM-533-4 Pelvic Floor Trainer with Smart Biofeedback and ETS Features
- Smart Pelvic Muscle Strengthener KM533-4 with App Based Tracking
- EMG Biofeedback Pelvic Device KM-533-4 for Muscle Strength and Care
- Kegel Exercise Machine KM-533-4 for Pelvic Floor Training and Control
- Pelvic Floor Therapy Device KM-533-4 with Easy Wireless App Control
- App-Based Pelvic Muscle Trainer KM-533-4 for Daily Home Therapy
- Wireless Pelvic Floor Strength Device KM-533-4 with Smart Features
- EMG Pelvic Therapy Machine KM-533-4 for Muscle Recovery and Training
- Smart Kegel Trainer KM-533-4 for Pelvic Strength and Stability
- Pelvic Muscle Training Device KM-533-4 with Biofeedback Support
- Kegel Therapy Device KM-533-4 for Better Pelvic Health and Control
- App-Controlled Pelvic Rehab Device KM-533-4 for Easy Daily Use
- Wireless Pelvic Muscle Trainer KM-533-4 with Smart Biofeedback System
- Pelvic Floor Exercise Trainer KM-533-4 for Home and Clinic Therapy
- Smart Pelvic Therapy Machine KM-533-4 for Muscle Strengthening
- Smart Pelvic Therapy Machine KM-533-4 for Muscle Strengthening
- EMG Biofeedback Kegel Trainer KM-533-4 for Recovery and Care
- Wireless Pelvic Floor Therapy Device KM-533-4 with App Monitoring
- Pelvic Muscle Strength Trainer KM-533-4 with Smart Training Support
- Kegel Exercise Device KM-533-4 for Pelvic Muscle Improvement
- Smart Pelvic Floor Rehab Machine KM-533-4 for Easy Therapy Use
- EMG Pelvic Trainer KM-533-4 for Muscle Activation and Strength
- Smart Pelvic Floor Trainer KM-533-4 with Wireless App-Based Therapy
- EMG Biofeedback Pelvic Device KM-533-4 for Muscle Strength and Control
- Kegel Exercise Machine KM-533-4 for Pelvic Floor Strength Training
- Pelvic Floor Therapy Device KM-533-4 with Easy App Control System
- App-Based Pelvic Muscle Trainer KM-533-4 for Home Rehabilitation
- Wireless Pelvic Floor Strength Device KM-533-4 with Smart Features
- EMG Pelvic Therapy Machine KM-533-4 for Muscle Recovery and Care
- Smart Kegel Trainer KM-533-4 for Pelvic Strength and Daily Exercise
- Pelvic Muscle Training Device KM-533-4 with Biofeedback Technology
- Kegel Therapy Device KM-533-4 for Better Pelvic Muscle Control
- App-Controlled Pelvic Rehab Trainer KM-533-4 for Easy Daily Use
- Wireless Pelvic Muscle Trainer KM-533-4 with EMG Biofeedback Support
- Pelvic Floor Exercise Trainer KM-533-4 for Home and Clinic Therapy
- Smart Pelvic Therapy Machine KM-533-4 for Muscle Strength Improvement
- EMG Biofeedback Kegel Trainer KM-533-4 for Recovery and Training
- Wireless Pelvic Floor Therapy Device KM-533-4 with Smart Monitoring
- Pelvic Muscle Strength Trainer KM-533-4 with Easy App Support
- Kegel Exercise Device KM-533-4 for Pelvic Muscle Stability
- Smart Pelvic Floor Rehab Machine KM-533-4 for Daily Therapy Use
- EMG Pelvic Trainer KM-533-4 for Muscle Activation and Recovery
- Smart Pelvic Floor Trainer KM-533-4 with App-Based Kegel Therapy
- EMG Biofeedback Pelvic Device KM-533-4 for Muscle Strength and Control
- Kegel Exercise Machine KM-533-4 for Pelvic Floor Strengthening
- Pelvic Floor Therapy Device KM-533-4 with Wireless App Support
- App-Based Pelvic Muscle Trainer KM-533-4 for Daily Home Therapy
- Wireless Pelvic Floor Strength Device KM-533-4 with Smart Features
- EMG Pelvic Therapy Machine KM-533-4 for Muscle Recovery and Care
- Smart Kegel Trainer KM-533-4 for Pelvic Strength and Stability
- Pelvic Muscle Training Device KM-533-4 with Biofeedback System
- Kegel Therapy Device KM-533-4 for Better Pelvic Muscle Control
- App-Controlled Pelvic Rehab Device KM-533-4 for Easy Daily Use
- Wireless Pelvic Muscle Trainer KM-533-4 with EMG Biofeedback Support
- Pelvic Floor Exercise Trainer KM-533-4 for Home and Clinic Use
- Smart Pelvic Therapy Machine KM-533-4 for Muscle Strength Improvement
- EMG Biofeedback Kegel Trainer KM-533-4 for Recovery and Training
- Wireless Pelvic Floor Therapy Device KM-533-4 with Smart Monitoring
- Pelvic Muscle Strength Trainer KM-533-4 with Easy App Control
- Kegel Exercise Device KM-533-4 for Pelvic Muscle Stability
- Smart Pelvic Floor Rehab Machine KM-533-4 for Daily Therapy Use
- EMG Pelvic Trainer KM-533-4 for Muscle Activation and Strength
- Smart Pelvic Floor Trainer KM-533-4 with Wireless App-Based Therapy
- EMG Biofeedback Pelvic Device KM-533-4 for Muscle Strengthening and Control
- Kegel Exercise Machine KM-533-4 for Pelvic Floor Strength and Stability
- Pelvic Floor Therapy Device KM-533-4 with Easy App-Based Operation
- App-Based Pelvic Muscle Trainer KM-533-4 for Daily Rehabilitation Use
- Wireless Pelvic Floor Strength Device KM-533-4 with Smart Biofeedback Support
- EMG Pelvic Therapy Machine KM-533-4 for Muscle Recovery and Training
- Smart Kegel Trainer KM-533-4 for Pelvic Strength and Daily Exercise
- Pelvic Muscle Training Device KM-533-4 with Biofeedback Technology
- Kegel Therapy Device KM-533-4 for Better Pelvic Muscle Control and Care
- App-Controlled Pelvic Rehab Trainer KM-533-4 for Easy Daily Therapy
- Wireless Pelvic Muscle Trainer KM-533-4 with EMG Biofeedback Features
- Pelvic Floor Exercise Trainer KM-533-4 for Home and Clinical Use
- Smart Pelvic Therapy Machine KM-533-4 for Muscle Strength Improvement
- EMG Biofeedback Kegel Trainer KM-533-4 for Recovery and Strength
- Wireless Pelvic Floor Therapy Device KM-533-4 with Smart Monitoring System
- Pelvic Muscle Strength Trainer KM-533-4 with Easy App Control
- Kegel Exercise Device KM-533-4 for Pelvic Muscle Stability and Strength
- Smart Pelvic Floor Rehab Machine KM-533-4 for Daily Therapy Routine
- EMG Pelvic Trainer KM-533-4 for Muscle Activation and Recovery
- Smart Pelvic Floor Trainer KM-533-4 with App-Based Therapy and Monitoring
- EMG Biofeedback Pelvic Device KM-533-4 for Muscle Strength and Daily Use
- Kegel Exercise Machine KM-533-4 for Pelvic Floor Strengthening and Control
- Pelvic Floor Therapy Device KM-533-4 with Easy Wireless App Control
- App-Based Pelvic Muscle Trainer KM-533-4 for Home Rehabilitation and Care
- Wireless Pelvic Floor Strength Device KM-533-4 with Smart Biofeedback Features
- EMG Pelvic Therapy Machine KM-533-4 for Muscle Recovery and Strength
- Smart Kegel Trainer KM-533-4 for Pelvic Muscle Strength and Stability
- Pelvic Muscle Training Device KM-533-4 with Biofeedback and App Support
- Kegel Therapy Device KM-533-4 for Better Pelvic Muscle Control and Health
- App-Controlled Pelvic Rehab Trainer KM-533-4 for Daily Therapy Routine
- Wireless Pelvic Muscle Trainer KM-533-4 with EMG Biofeedback System
- Pelvic Floor Exercise Trainer KM-533-4 for Home and Clinic Therapy Use
- Smart Pelvic Therapy Machine KM-533-4 for Muscle Strength Improvement
- EMG Biofeedback Kegel Trainer KM-533-4 for Recovery and Strength Building
- Wireless Pelvic Floor Therapy Device KM-533-4 with Smart Monitoring Support
- Pelvic Muscle Strength Trainer KM-533-4 with Easy App-Based Control
- Kegel Exercise Device KM-533-4 for Pelvic Muscle Stability and Training
- Smart Pelvic Floor Rehab Machine KM-533-4 for Daily Therapy and Care
- EMG Pelvic Trainer KM-533-4 for Muscle Activation and Rehabilitation
- Smart Pelvic Floor Trainer KM-533-4 with App-Based Control and EMG Biofeedback
- Wireless Pelvic Therapy Device KM-533-4 for Muscle Strength and Recovery
- Kegel Exercise Machine KM-533-4 for Pelvic Floor Strengthening and Stability
- Pelvic Floor Therapy Device KM-533-4 with Easy App Monitoring System
- App-Based Pelvic Muscle Trainer KM-533-4 for Daily Home Rehabilitation
- EMG Biofeedback Pelvic Device KM-533-4 for Muscle Activation and Control
- Wireless Pelvic Floor Strength Device KM-533-4 with Smart Features
- Smart Kegel Trainer KM-533-4 for Pelvic Muscle Strength and Care
- Pelvic Muscle Training Device KM-533-4 with Biofeedback Technology
- Kegel Therapy Device KM-533-4 for Better Pelvic Muscle Control
- App-Controlled Pelvic Rehab Trainer KM-533-4 for Easy Daily Use
- Wireless Pelvic Muscle Trainer KM-533-4 with EMG Biofeedback Support
- Pelvic Floor Exercise Trainer KM-533-4 for Home and Clinic Therapy
- Smart Pelvic Therapy Machine KM-533-4 for Muscle Strength Improvement
- EMG Biofeedback Kegel Trainer KM-533-4 for Recovery and Strength
- Wireless Pelvic Floor Therapy Device KM-533-4 with Smart Monitoring
- Pelvic Muscle Strength Trainer KM-533-4 with Easy App Control
- Kegel Exercise Device KM-533-4 for Pelvic Muscle Stability
- Smart Pelvic Floor Rehab Machine KM-533-4 for Daily Therapy Use
- EMG Pelvic Trainer KM-533-4 for Muscle Activation and Recovery
- Smart Pelvic Floor Trainer KM-533-4 with Wireless App-Based Therapy Support
- EMG Biofeedback Pelvic Device KM-533-4 for Muscle Strength and Control
- Kegel Exercise Machine KM-533-4 for Pelvic Floor Strength and Stability
- Pelvic Floor Therapy Device KM-533-4 with Easy App-Based Operation
- App-Based Pelvic Muscle Trainer KM-533-4 for Daily Rehabilitation Use
- Professional Pelvic Recovery Equipment with Adjustable EMG Stimulation
- KM-533-4 Digital Women Pelvic Therapy Machine for Muscle Endurance Training
- Advanced Pelvic Floor Muscle Coordination Device with Wireless Features
- Smart Kegel Therapy Equipment for Pelvic Stability and Strength Improvement
- KM-533-4 Portable Pelvic Biofeedback Rehabilitation Trainer for Women
- Wireless Pelvic Muscle Recovery Device with APP Guided Exercise Programs
- Professional EMG Pelvic Rehabilitation Machine for Physiotherapy Applications
- KM-533-4 Advanced Pelvic Floor Exercise Equipment for Women Recovery
- Smart Women Pelvic Therapy Trainer with ETS Muscle Activation Function
- KM-533-4 Intelligent Kegel Recovery Machine with Wireless Connectivity
- Advanced Pelvic Health Rehabilitation Device for Daily Physiotherapy Use
- Wireless Pelvic Floor Therapy Equipment with Real-Time EMG Monitoring
- KM-533-4 Smart Pelvic Muscle Recovery Trainer for Bladder Stability Support
- Professional Pelvic Rehabilitation System with APP Controlled Therapy Modes
- KM-533-4 Advanced EMG Biofeedback Device for Pelvic Muscle Training
- KM-533-4 Wireless Pelvic Floor Recovery System for Smart Kegel Rehabilitation
- Advanced Pelvic Therapy Equipment with EMG Guided Muscle Training
- Smart APP Controlled Pelvic Muscle Rehabilitation Device for Women
- KM-533-4 Digital Kegel Recovery Machine with Wireless Biofeedback Support
- Wireless Pelvic Floor Exercise Device for Bladder Control Improvement
- Professional Pelvic Muscle Therapy System with ETS Stimulation Technology
- KM-533-4 Smart Women Pelvic Rehabilitation Equipment for Daily Recovery
- KM-533-4 Smart Women Pelvic Rehabilitation Equipment for Daily Recovery
- Advanced EMG Pelvic Floor Trainer for Muscle Coordination and Stability
- Portable Pelvic Therapy Machine for Urinary Incontinence Rehabilitation
- KM-533-4 Intelligent Pelvic Recovery Device with APP Guided Exercises
- Wireless Pelvic Floor Strengthening Equipment for Women Physiotherapy
- Smart Kegel Muscle Stimulator with Real-Time EMG Biofeedback Analysis
- KM-533-4 Advanced Pelvic Rehabilitation Machine for Postpartum Care
- Professional Women Pelvic Therapy Device with Wireless APP Connectivity
- Wireless EMG Pelvic Recovery Trainer for Daily Physiotherapy Programs
- KM-533-4 Smart Pelvic Biofeedback Machine for Muscle Endurance Training
- Advanced Pelvic Floor Muscle Therapy Equipment for Bladder Support
- Portable Kegel Rehabilitation Device for Women Health Recovery Sessions
- KM-533-4 Intelligent Pelvic Strengthening System with ETS Activation
- Wireless Pelvic Muscle Exercise Machine for Clinical Rehabilitation Use
- Smart Pelvic Recovery Equipment for Urinary Leakage and Muscle Weakness
- KM-533-4 Advanced Women Health Therapy System with EMG Monitoring
- Professional Pelvic Floor Rehabilitation Device with Smart Digital Programs
- Wireless Kegel Exercise Trainer for Pelvic Stability Rehabilitation
- KM-533-4 APP Based Pelvic Muscle Recovery Equipment for Women
- Advanced Pelvic Biofeedback Therapy Machine for Daily Kegel Training
- Smart EMG Pelvic Rehabilitation Trainer with Adjustable Therapy Modes
- KM-533-4 Portable Pelvic Therapy Equipment for Home and Clinic Applications
- Wireless Pelvic Muscle Coordination Device for Women Recovery Programs
- Professional Kegel Recovery Machine with Real-Time Muscle Feedback
- KM-533-4 Smart Pelvic Floor Therapy System for Bladder Rehabilitation
- Advanced Women Pelvic Strengthening Equipment with Wireless Technology
- Wireless EMG Pelvic Muscle Recovery Device for Physiotherapy Treatment
- KM-533-4 Intelligent Pelvic Floor Exercise Trainer with APP Support
- Smart Pelvic Rehabilitation Machine with ETS Triggered Muscle Activation
- Professional Pelvic Muscle Stimulator for Kegel and Recovery Therapy
- KM-533-4 Advanced Biofeedback Pelvic Training System for Women
- Wireless Pelvic Floor Rehabilitation Equipment for Daily Muscle Exercises
- Smart Women Health Pelvic Recovery Device with Digital EMG Technology
- KM-533-4 Portable Kegel Therapy Trainer for Pelvic Muscle Strengthening
- KM-533-4 Wireless Pelvic Therapy Device for Advanced Kegel Rehabilitation
- Smart Pelvic Floor Biofeedback Machine with ETS Muscle Training Function
- Advanced Women Pelvic Recovery Equipment for Urinary Incontinence Therapy
- KM-533-4 Digital Pelvic Muscle Rehabilitation Trainer with APP Control
- Wireless Pelvic Floor Strengthening Machine for Postpartum Recovery Care
- Professional EMG Kegel Therapy System for Women Physiotherapy Programs
- KM-533-4 Smart Pelvic Recovery Device with Real-Time Muscle Monitoring
- Advanced Pelvic Floor Exercise Equipment for Bladder Stability Improvement
- Portable Pelvic Rehabilitation Machine with Wireless EMG Biofeedback
- KM-533-4 Intelligent Pelvic Therapy Trainer for Daily Recovery Sessions
- Wireless Women Health Rehabilitation Equipment for Pelvic Muscle Training
- Smart Kegel Rehabilitation Device with APP Guided Therapy Programs
- KM-533-4 Advanced Pelvic Muscle Stimulator with ETS Activation Technology
- Professional Pelvic Floor Therapy Machine for Muscle Endurance Training
- Wireless EMG Pelvic Recovery Equipment for Clinical Physiotherapy Use
- KM-533-4 Smart Biofeedback Pelvic Exercise Device for Women Health
- Advanced Pelvic Rehabilitation System for Daily Kegel Exercise Therapy
- Portable Pelvic Muscle Recovery Device with Adjustable Stimulation Levels
- KM-533-4 Intelligent Women Pelvic Strengthening Equipment with APP Support
- Wireless Pelvic Floor Muscle Therapy Trainer for Bladder Control Support
- Smart Pelvic Rehabilitation Equipment with Real-Time EMG Signal Analysis
- KM-533-4 Advanced Kegel Recovery Machine for Pelvic Stability Training
- Professional Women Pelvic Therapy Equipment with Wireless Connectivity
- Wireless Pelvic Muscle Recovery System for Postpartum Physiotherapy Care
- KM-533-4 Smart Pelvic Floor Trainer for Urinary Leakage Rehabilitation
- Advanced EMG Biofeedback Pelvic Device for Daily Muscle Coordination
- Portable Kegel Exercise Equipment for Women Pelvic Health Improvement
- KM-533-4 Intelligent Pelvic Floor Recovery Machine with Digital Programs
- Wireless Pelvic Therapy System for Muscle Activation and Rehabilitation
- Professional Pelvic Rehabilitation Trainer with APP Controlled Functions
- KM-533-4 Smart Women Pelvic Recovery Device with ETS Stimulation Support
- Advanced Pelvic Muscle Strengthening Machine for Physiotherapy Applications
- Wireless EMG Kegel Trainer for Bladder and Pelvic Stability Therapy
- KM-533-4 Portable Pelvic Biofeedback Equipment for Women Rehabilitation
- Smart Pelvic Therapy Device with Adjustable Kegel Exercise Modes
- Professional Pelvic Floor Recovery System for Daily Rehabilitation Programs
- KM-533-4 Advanced Pelvic Muscle Coordination Equipment with Wireless APP
- Wireless Pelvic Rehabilitation Machine for Women Health Physiotherapy
- Smart EMG Pelvic Exercise Trainer for Muscle Endurance and Recovery
- KM-533-4 Intelligent Kegel Therapy Equipment for Pelvic Floor Strengthening
- KM-533-4 Wireless Pelvic Floor Rehabilitation Trainer with Smart EMG Control
- Advanced Kegel Therapy Device for Women Pelvic Muscle Recovery Programs
- Smart Pelvic Rehabilitation Equipment with APP Based Biofeedback Training
- KM-533-4 Digital Pelvic Floor Strengthening Machine with ETS Technology
- Wireless Pelvic Muscle Therapy System for Urinary Incontinence Support
- Professional EMG Pelvic Recovery Trainer for Daily Physiotherapy Sessions
- KM-533-4 Smart Kegel Exercise Machine for Pelvic Stability Improvement
- Advanced Women Pelvic Therapy Equipment with Wireless Muscle Monitoring
- Portable Pelvic Rehabilitation Device for Home and Clinical Applications
- KM-533-4 Intelligent Pelvic Floor Recovery Equipment with APP Features
- Wireless Pelvic Muscle Coordination Trainer for Postpartum Rehabilitation
- Smart Biofeedback Kegel Therapy Device for Women Health Recovery
- KM-533-4 Advanced Pelvic Floor Exercise Equipment for Bladder Support
- Professional Pelvic Muscle Stimulator with Adjustable Therapy Programs
- Wireless EMG Pelvic Recovery System for Daily Kegel Exercise Therapy
- KM-533-4 Smart Women Pelvic Strengthening Machine with ETS Activation
- Advanced Pelvic Rehabilitation Therapy Device for Muscle Endurance Training
- Portable Kegel Recovery Equipment with Real-Time EMG Signal Feedback
- KM-533-4 Intelligent Pelvic Muscle Therapy Machine for Women Physiotherapy
- Wireless Pelvic Floor Rehabilitation Equipment for Clinical Therapy Use
- Smart Pelvic Health Recovery Device with APP Guided Exercise Programs
- KM-533-4 Advanced EMG Biofeedback Kegel Trainer for Muscle Activation
- Professional Women Pelvic Therapy Machine with Wireless Connectivity Support
- Wireless Pelvic Floor Strengthening Device for Daily Rehabilitation Programs
- KM-533-4 Smart Pelvic Exercise System for Bladder Stability Improvement
- Advanced Pelvic Recovery Trainer with ETS Triggered Muscle Therapy
- Portable Pelvic Biofeedback Machine for Women Health Physiotherapy
- KM-533-4 Intelligent Kegel Rehabilitation Equipment with Smart Monitoring
- Wireless Pelvic Muscle Recovery Device for Urinary Leakage Management
- Professional Pelvic Floor Exercise Trainer with Digital EMG Technology
- KM-533-4 Smart Pelvic Rehabilitation System for Women Recovery Programs
- Advanced Women Pelvic Health Equipment for Muscle Coordination Therapy
- Wireless EMG Pelvic Therapy Machine for Home and Clinic Rehabilitation
- KM-533-4 Portable Pelvic Floor Recovery Trainer with APP Connectivity
- Smart Kegel Muscle Strengthening Device for Physiotherapy Applications
- Professional Pelvic Rehabilitation Equipment with Adjustable ETS Stimulation
- KM-533-4 Advanced Pelvic Muscle Exercise Machine for Daily Recovery
- Wireless Pelvic Floor Therapy Trainer with Real-Time Biofeedback Analysis
- Smart Women Pelvic Recovery Equipment for Kegel Exercise Rehabilitation
- KM-533-4 Intelligent EMG Pelvic Rehabilitation Device for Muscle Strengthening
- KM-533-4 Wireless Pelvic Floor Therapy Machine with Smart Rehabilitation Modes
- Advanced EMG Kegel Recovery Equipment for Women Pelvic Health Programs
- Smart Pelvic Muscle Strengthening Device with APP Controlled Functions
- KM-533-4 Digital Pelvic Rehabilitation Trainer for Daily Therapy Sessions
- Wireless Pelvic Floor Exercise System for Bladder Control Improvement
- Professional Pelvic Therapy Equipment with ETS Triggered Stimulation
- KM-533-4 Smart Women Pelvic Recovery Machine with Biofeedback Analysis
- Advanced Pelvic Muscle Therapy Device for Postpartum Recovery Care
- Portable Pelvic Floor Rehabilitation Equipment with Wireless Connectivity
- KM-533-4 Intelligent Kegel Exercise Trainer for Muscle Coordination Therapy
- Wireless EMG Pelvic Strengthening System for Women Physiotherapy Clinics
- Smart Pelvic Rehabilitation Machine with Real-Time Muscle Feedback
- KM-533-4 Advanced Women Health Therapy Device with Adjustable Programs
- Professional Pelvic Floor Recovery Equipment for Daily Rehabilitation Use
- Wireless Pelvic Muscle Exercise Trainer for Urinary Leakage Management
- KM-533-4 Smart Biofeedback Pelvic Therapy System for Kegel Training
- Advanced Pelvic Rehabilitation Equipment with APP Guided Muscle Exercises
- Portable Women Pelvic Therapy Device for Home and Clinical Applications
- KM-533-4 Intelligent Pelvic Floor Recovery Trainer with ETS Technology
- Wireless Pelvic Health Rehabilitation System for Bladder Stability Support
- Smart EMG Kegel Therapy Equipment for Women Recovery Programs
- KM-533-4 Advanced Pelvic Muscle Stimulator with Wireless Monitoring
- Professional Pelvic Floor Therapy Machine for Muscle Endurance Improvement
- Wireless Pelvic Recovery Device for Daily Physiotherapy and Rehabilitation
- KM-533-4 Smart Women Pelvic Strengthening Trainer with APP Connectivity
- Advanced Pelvic Muscle Coordination Equipment for Kegel Rehabilitation
- Portable Pelvic Floor Biofeedback System with Adjustable Stimulation Levels
- KM-533-4 Intelligent Pelvic Exercise Machine for Women Health Recovery
- Wireless EMG Pelvic Therapy Equipment for Clinical Rehabilitation Sessions
- Professional Pelvic Rehabilitation Trainer with Real-Time Biofeedback Support
- KM-533-4 Smart Kegel Recovery Device for Pelvic Stability and Strength
- Advanced Women Pelvic Therapy Machine with Digital EMG Monitoring
- Wireless Pelvic Muscle Recovery Equipment for Daily Kegel Exercises
- UniGym Robot Gym for Physiotherapy Rehabilitation and Recovery
- UniGym Smart Robot Gym for Hand and Limb Rehabilitation
- UniGym Portable Rehabilitation Robot Gym for Home and Clinic Use
- UniGym Advanced Physiotherapy Rehabilitation Equipment
- UniGym Robot Gym for Upper and Lower Limb Physiotherapy
- UniGym Smart Rehabilitation Therapy Device for Stroke Recovery
- UniGym Robotic Rehab Gym Machine for Physical Therapy
- UniGym Motorized Robot Gym for Rehabilitation Exercise Therapy
- UniGym Smart Therapy Rehabilitation Equipment for Paralysis Recovery
- UniGym Robot Gym Physiotherapy Machine for Rehab Training
- UniGym Compact Rehabilitation Robot for Physiotherapy Clinics
- UniGym Digital Rehabilitation Therapy Equipment
- UniGym Smart Rehab Exercise Machine for Arm and Leg Therapy
- UniGym Robotic Physiotherapy Equipment for Home Rehabilitation
- UniGym Advanced Robot Gym for Stroke and Paralysis Rehabilitation
- UniGym Rehabilitation Training Device for Muscle Recovery Therapy
- UniGym Smart Physiotherapy Robot Gym Equipment
- UniGym Portable Physical Therapy and Rehabilitation Machine
- UniGym Robot Gym for Neurological and Orthopedic Rehabilitation
- UniGym Smart Motorized Rehab Equipment for Daily Physiotherapy
- UniGym Clinical Rehabilitation Robot Gym for Recovery Exercise
- UniGym Professional Physiotherapy Rehabilitation System
- UniGym Smart Recovery and Rehabilitation Exercise Device
- UniGym Automatic Robot Gym for Hand and Joint Rehabilitation
- UniGym Multifunctional Rehabilitation Therapy Equipment
- Pressure Therapy Equipment
- Advanced Class 4 Laser Therapy Equipment for Physiotherapy
- Professional Class 4 Laser Physiotherapy Device for Hospitals
- Digital hand grip analyzer measurement
- Exoskeleton Systems Designed for Precision Physiotherapy Training
- Hospital Use Transcranial And Vagus Stimulator
- Professional physiotherapy laser device Digital physiotherapy laser machine
- Shockwave Therapy Machine Electromagnetic ED Shock Wave Machine for Body Muscle Relieve Pain
- Forever Bubble Inclinometer Physiotherapy Equipment
- Acoustic Physical Pain Shockwave Therapy System
- Re-Usable Self-Adhesive TENS Units Electrodes, 4 pcs of Pads 2
- Wrist Rehabilitation Device
- Best Wrist CPM Machine for Pain Relief | Post-Surgery, Stroke Recovery, and Arthritis Therapy Equipment
- Wrist CPM Machine | Continuous Passive Motion Device for Wrist Therapy, Arthritis & Post-Surgery Recovery
- Economical Robotic Glove for Hand Therapy
- Premium Black Edition Pneumatic Shockwave Therapy Device for Physiotherapy and Sports Injury Recovery
- Cold Compression Therapy Device for Post-Surgery & Sports Injury Rehabilitation
- Portable Cryotherapy Compression System for Joint & Muscle Recovery
- TENS - Vaginal Probe Electrode for TENS - EMS - E-Stim Devices
- Digital 6 Chamber Pneumatic DVT Pump With Multiple Modes And Timer
- Smart Exoskeleton Rehabilitation Robot for Upper and Lower Limb Therapy
- Deep Penetration Focused Shockwave Therapy Equipment for Tissue Regeneration
- Clinical Brainwave Synchronization And Neuro Rehabilitation Machine
- Class 4 Laser Therapy Machine for Muscle Pain Treatment
- Therapy Machine
- Robotics Hand Rehabilitation Gloves
- Occupational Therapy Equipment
- Dvt Pump
- Bobath Tables
- Air Compression Therapy Equipment
- DVT Treatment Device Manual Sequential Compression Device
- Compact DVT pump Home use pump Portable edema device Lymphedema aid pump DVT aid pump
- Pneumatic Compression Therapy Equipment
- Compression therapy legs arms compression pump DVT arms legs compression pump
- Pneumatic Compression Therapy Device, For Hospital, Four Chamber
- Advanced Four-Chamber DVT Compression Pump for Varicose Vein Therapy
- Pressotherapy Slimming Lymphatic Drainage System Far-Infrared Sauna Thermal Blanket
- DVT prophylaxis equipment Medical compression therapy device
- ABS Plastic Multi-Chamber Compression Therapy DVT Machine
- DVT compression Portable relief device Travel therapy pump
- DVT portable aid Lightweight leg pump Wearable DVT pump
- Sequential Compression Therapy Device 4 Chamber Manual DVT pump
- DVT Effective compression therapy device Lymphedema and DVT treatment at home
- Biotronix DVT Air Compression Lymphedema Pump 4 chamber Digital
- DVT Pump
- Professional 6 Chamber Pressotherapy Sports Recovery Boots Dvt Pump Air Compression
- Best hospital pneumatic compression therapy device with four chambers
- DVT Trusted four-chamber pneumatic compression therapy device for hospitals
- DVT Affordable four-chamber pneumatic compression device for hospitals
- DVT Efficient hospital pneumatic compression therapy machine
- Physiotherapy Air compression Leg Massage Machine DVT pump 4 Chamber Manual
- four-chamber pneumatic compression therapy device for hospitals
- DVT Compression Air therapy 4 Chamber Digital for LEGS Waist Arm
- DVT compression therapy system
- Compression therapy machine Four-chamber pneumatic device
- Lymphatic Drainage Dvt Machine 4 Chamber Digital Physiotherapy Machine
- Air Compression Therapy Pump
- Digital Compression Therapy DVT Pump
- Plastic Compression Air Therapy Machine Digital, For Clinical
- ABS Plastic Exercise Equipment PORTABLE PNEUMATIC COMPRESSION DEVICE, Universal Size, Reusable
- Dvt Pump 4 Chamber Digital Model With Legs Attachments
- Plastics ABS Exercise Equipment Scd Machine Sequential Compression Device
- Lymphedema pump DVT pump Compression pump Portable pump Lymphedema relief
- Niscomed Lymphedema Dvt Pump For Legs And Compression Therapy Pump For Hospital
- DVT Treatment Device Manual Compression Device Sequential Compression Device 4-Chamber Compression Pump
- Four-chamber therapy device Medical compression equipment Hospital pneumatic therapy
- High-quality four-chamber pneumatic compression therapy system for hospitals
- Portable Plastic DVT Machine Digital 4 Chamber with Remote Control, For Four-chamber Compression Unit, Adjustable Compression
- Effective DVT Relief with Manual Four-Chamber Compression Pump Air Compression Device
- 4 chamber compression therapy system Deep vein thrombosis prevention tool
- Plastics ABS Portable Lymphedema Dvt Sequential Therapy Pump
- Plastics ABS Portable Lymphedema Compression Dvt Pump
- Lymphedema device DVT device Home pump Compact pump Edema pump
- 4-Chamber DVT Pump Manual DVT Pump Deep Vein Thrombosis Pump DVT Prevention Device
- 05 250 Air Compression Therapy Equipment, For Clinical, Four Chamber
- 4-Chamber Compression Pump Portable DVT Pump
- DVT Blood circulation improvement device Manual air compression therapy unit
- Compression Therapy Pump DVT Treatment Device Manual Compression Device
- DVT Prevention for Home Use Effective DVT Therapy Home DVT Pump
- 4-chamber Air Compression Pump Therapy
- Compact Four-Chamber DVT Compression Pump for Home and Clinic Use
- DVT Therapy Device Venous Thrombosis Prevention Leg Compression Pump
- DVT 4-Chamber Sequential Compression Devices
- dvt Leg and Foot Recovery Device Portable Leg Compression Massager
- Lymphedema management device Effective DVT compression pump
- ABS Plastic Rectangular Pneumatic Air Compression Therapy DVT pumps Digital Model
- 4 chamber DVT legs arms compression therapy
- DVT 50-60 Hz 20-250 Mmhg 6 Chamber Air Compression Lymphatic Drainage Massage Prevent
- 50-60 Hz 20-250 Mmhg Pneumatic Air compression Therapy Machine DVT Pump 6 Chamber, For Hospital
- DVT Pump 4 Air Chambers Compression
- DVT Compression Pump Venous Flow Pump Pneumatic Compression Device
- 4 Chamber Leg Compression Portable Therapy Pump pump
- DVT prophylaxis device compression pump for leg veins
- DVT support pump circulatory health device intermittent leg compression
- DVT compression therapy equipment leg pump for thrombosis
- DVT protection device air pump for blood clots lower extremity compression
- DVT multi-chamber compression pump therapeutic air pump
- DVT management device venous thromboembolism prevention
- DVT compression sleeves air compression therapy DVT prevention system
- DVT sequential air compression deep vein therapy
- DVT intermittent compression pump pneumatic leg compression vascular health device
- ROUNDED Portable Professional Four-Chamber Compression Pump
- Plastics ABS Portable DVT Prevention Device 4 Chamber Digital
- Compression Therapy Pump for Lymphedema Digital DVT Compression Therapy Device
- Plastics ABS Portable Air Compression Therapy DVT Pump 4 Chamber
- DVT compression Portable relief device Travel therapy pump Effective compression device
- Dvt Sequentail Compression Therapy 4 Chambers (manual), for Hospital
- DVT Four-chamber medical therapy device Compression therapy
- Portable DVT compression therapy Compression pump for home use
- ABS Plastic Compression Therapy Equipment Lymphedema Management DVT Machine, For Clinical, Four Chamber
- ABS Plastic Rectangular Pumps for Edema Lymphedema and Lipedema DVT 4 Chamber, Adjustable Compression
- 4 chamber DVT compression DVT compression legs Digital compression legs Leg DVT system
- Leg and Foot Pain Relief Air Compression Device for Home
- ABS Plastic Rectangular Lymphedema Compression Dvt Pump
- Air Pressure Therapy System Leg Massager Machine Shape Body
- DVT arms legs compression DVT legs arms compression pump
- Air Leg Massager Foot and Leg Massager for DVT Hospital Grade DVT Device
- 6 Chamber DVT Air Compression Therapy System Fullset
- dvt Foot Massager Air Compression Therapy Leg and Foot Massager Machine
- 50-60 Hz 20-220 Mmhg Air Pressure Therapy System w/Leg Cuff (4C-Digital/IPC02), For Hospital, Four Chamber
- dvt Foot and Leg Circulation Massager Air Compression Therapy for Legs
- DVT Machine DVT Pumps
- Dvt Pump Machine
- DVT therapy arms DVT pump arms Digital compression arms 4 chamber DVT legs
- Dvt Compression Therapy Manual 4 chamber Portable for Clinical and Home Use
- ABS Plastic Pneumatic Compression Therapy Equipment, For Clinical, Four Chamber
- DVT air pump Digital DVT pump 4 chamber DVT device
- DVT Pneumatic Air Compression Lymphedema Manual 4 Chamber LEGS ARM WAIST
- DVT Comprehensive four-chamber pneumatic compression therapy machine
- Air Compression Recovery Boots DVT Sleeves Leg Arm Foot Pressotherapy
- DVT Prevention and Treatment Lymphatic System Support Pump
- Air Compression Therapy DVT 4 Chamber Equipment
- Pneumatic Compression Air Therapy System
- ABS Plastic Pneumatic Compression Therapy Device, For Hospital, Four Chamber
- ABS Plastic Pneumatic Compression Therapy Equipment, For Clinical, Four Chamber
- ABS Plastic Pneumatic Compression Therapy Equipment, For Clinical, Four Chamber
- DVT treatment Wearable pump Lightweight pump Home therapy Travel compression
- Four-chamber compression therapy system compression equipment
- User-Friendly Four-Chamber DVT Compression Pump for Home Use manual DVP pump
- Innovative 4-Chamber DVT Compression Pump for Treatment of Varicose Veins
- 4-Chamber Compression Pump for Thrombosis in the Deep Veins Manual model Air Compression
- Novel 4-Chamber DVT Compression Pump for Varicose Vein Therapy
- Novel 4-Chamber DVT Compression Pump for Varicose Vein Therapy
- DVT compression therapy air compression pump for DVT
- DVT prevention equipment intermittent pneumatic compression
- Lymphatic Drainage Dvt Machine 4 Chamber Digital Physiotherapy Machine
- dvt Leg Lymphedema Treatment Pump Hospital Compression Pump System
- DVT Machine Digital Air Compression Therapy Pump
- Air Compression Leg Massager for Calves, Knees & Feet - Lymphedema, Circulation, and Pain Relief
- Air Compression Therapy Device (8 Ch) - 2 Legs, 1 Waist, 1 Arm Sleeve for Swelling, Pain Relief
- Portable Air Compression Therapy System (8 Channels) with Multi-Sleeves
- Plastics ABS Therapeutic Digital Air Compression Pump for DVT with 4 Chambers
- Air Compression Therapy Device (8 Ch) - 2 Legs, 1 Waist, 1 Arm Sleeve for Swelling, Pain Relief
- Air Compression Therapy 8 Ch with 2 Legs,1 Waist,1 Arm Sleeve
- Compression Device Leg Pump Machine
- Compression Device Manufacturers Air Relax Air Compression Leg Massager
- Air Compression Massager with Heat for Foot, Leg and Knee
- Compression Air Therapy Machine Digital, For Clinical
- DVT Air Compression Device 4 Ch Digital with 2Legs 1Waist 1Arm Sleeves
- Portable Compression Air Therapy DVT
- Compression Therapy System for Hospitals, For Clinical, Four Chamber
- 4 Chamber Digital Compression Machine
- Air Compression Leg Massager for Foot and Calf Circulation Massage
- DVT 4 Chamber Lymphatic Drainage Machine with Digital Control for Leg Therapy
- Air Compression Therapy Device
- Air Wave Pressure Massager Air Cycle Machine Leg Pulse Pressure Gas Recovery Massage
- 4 Chamber Air Compression Leg Massager for Lymphatic Drainage & Swelling Reduction
- Leg Massager, 3 Modes Feet Hot Air Timer for Therapy
- Dvt Pump Sequential Compression Device
- Leg Massager, 3 Modes Feet Hot Air Timer for Therapy
- Finger Dexterity Board
- Aesthetic Devices And Equipments
- Weight Loss Equipment
- Body Shaper Ultrasonic Cavitation RF Slimming Machine Weight Loss, For Clinical Purpose
- Skin Exfoliation and Hydration Machine Hydrafacial Device with Infusion Serums
- ABS Plastic Vacuum Scraping Cupping Therapy Machine chest Enlargement
- Vacuum Therapy Pump Chest Enhancement Instrument
- Biotronix 40k Ultrasonic liposuction Cavitation 8 Pads Laser Vacuum RF Skin Salon Spa Slimming
- Ultrasonic Cavitation Machine Multifunction Digital Touch Screen with Radio Frequency RF and Vacuum
- Ultrasonic Cavitation Machine with RF vacuum Slimming Fat loss Device Body Shaping LIPO Therapy
- Biotronix CAVITATION RF Vacuum LIPO LASER Slimming Machine Fat Loss Device
- lipotherapy unit ultrasonic RF cavitation 40kHz cavitation machine vacuum slimming device
- Biotronix 6 In1 kim 8 40k Ultrasonic liposuction Cavitation Pads Vacuum RF Skin Salon Slimming
- Lipotherapy Unit Ultrasonic RF 40Khz Cavitation Vacuum 5 in one Slimming Center Machine
- Slimming Machine Cavitation 6 in 1 Radiofrequency RF Vacuum LIPO LASER Digital Weight loss
- Biotronix 6 in one Touch Screen Weigh Loss Cavitation RF Vacuum LIPO LASER Device Slimming
- Ultrasonic Cavitation Machine 5 in one with Face RF Body RF Vacuum RF Slimming Machine
- Dermabrasion Facial Machine for Sensitive Skin Hydrafacial and LED Light Therapy Device
- Chest Enlargement Chest Enhancement Device Vacuum Therapy Machine
- Fat Loss Machine Cavitation Weight Loss RF Skin Tightening
- Body Shaper Vacuum Cavitation RF System Slimming Machine,, For Hospital
- Plastic Vacuum Therapy Equipment, For Clinical
- Vacuum Therapy Machine Lymph Drainage Machine
- ABS Plastic Vacuum Therapy Machine Lymph Drainage Machine chest Enlarger Pump, For Clinical
- Natural Chest Enhancement Safe and Effective Enlargement with a Vacuum Therapy Machine
- Vacuum Machine Vacuum Cupping Machine Lifter Vacuum Lift Machine Chest Enhancers
- Electric Home chest Massager Vacuum Therapy Machine Skin
- Biotronix 7 in 1 HydraFacial Machine (DOUBLE MOTOR)
- ABS Plastic Vacuum Therapy Machine Chest Enlargement Massage Device, For Clinical, Electrotherapy equipment
- Professional Hydrafacial Device Skin Rejuvenation Machine Facial Cleansing Equipment
- Biotronix Sauna Portable Far Infrared Sauna Heating Blanket Detox Home Professional Salon Beauty
- HydraFacial Rf Aqua Peel Skin Scrubber Facial Machine Skin Care Machines Professional Salon
- slimming body shaper wrap remote controlled weight loss wrap
- Automatic Ultrasonic Cavitation Slimming Equipment, For Hospital
- Suction Pump Enlargement Therapy for Large Chest Vacuum lift for the with cups
- Salon Hydra Facial Machine
- Cavitation Device 5 in 1 lLIPO Cavitation Ultrasonic Cavitation RF Vacuum Slimming Fat Loss Device
- Vacuum LIPO LASER Slimming System Cavitation RF Body Shaping
- Hydrafacial Machine for Hyperpigmentation Facial Exfoliation Device
- White Plastic HydraFacial Machine Trolley Beauty Cart, For Professional
- Professional Facial Treatment Equipment Hydrafacial System with Skin Brightening Technology
- Hydrafacial System with Oxygen Therapy Professional Skincare Device for Anti-Aging
- Facial Treatment System with Vacuum Extraction Professional Skincare Equipment for Home Facials
- Professional Skin Care Treatment Device Hydrafacial System for Deep Cleansing
- Advanced Hydrafacial Machine for Home Use Professional Skincare Device for Deep Cleansing
- Professional Facial Treatment System Hydrafacial Equipment for Fine Lines
- Cavitation Multipolar RF Vacuum and Laser Therapy Slimming Machine
- Cavitation RF Body Sculpting System Vacuum RF Slimming Equipment
- RF Cavitation Fat Blaster Device Cavitation RF and LIPO LASER Slimming System
- RF Cavitation and Vacuum Therapy LIPO LASER Fat Reduction Treatment
- Cavitation RF Fat Reduction Device RF Vacuum Slimming Treatment
- Cavitation 5 IN 1 RF Body Shaping Beauty Machine Body Tighten Face Body Skin Care
- Vacuum Therapy Lift Machine Body Shaping Massage Cups Chest Enlargement
- Largest Cup Lift Massager Chest Enlargement vacuum cupping therapy machine
- Biotronix Six in One Ultrasonic Cavitation RF Vacuum LASER Weight Loss Combination Device
- Vacuum Therapy Machine Chest Enlargement and Enhancement Device
- Biotronix Infrared Sauna Blanket Digital Far Infrared Heat Sauna Blanket Heating Detox Therapy
- Biotronix Cavitation 5 in one Slimming Fat loss Equipment with RF Radiofrequency Vacuum
- 5 in 1 Body Massage Shaping Machine with RF Vacuum Cavitation
- Lipo Lipolaser Weight Loss Equipment With CAVITATION RF VACCUM Digital Touch Screen Slimming Device
- 5 in 1 Body Massage Shaping Machine with RF Vacuum Cavitation
- Ultrasonic Cavitation Machine Face RF Body Rf Tripolar Rf Vacuum Rf 5 in one Slimming Device
- Lipolaser Ultrasound Liposuction Cavitation 8 Pads LLLT Lipo Fat LASER Slimming Machine
- Slimming Equipment 5 in 1 Lipo Ultrasonic Cavitation RF and Vacuum Therapy Weight Loss Device
- Electric weight reduction blanket thermal body wrap with zones
- 6 in 1 Multi-Functional Lipolaser RF Vacuum Ultrasonic Lipo Cavitation Machine
- 5 in 1 Ultrasonic Cavitation FACE BODY RF Vacuum Slimming Machine Fat loss Machine
- Body Shaper Ultrasonic Cavitation Machine, For Clinical Purpose
- Skincare Device with Anti-Aging Serums Hydrafacial System for Skin Brightening
- Chest Enlargement Device, For Clinical, Electrotherapy equipment
- Biotronix Body Shaper Weight Loss Sauna Thermal Heating Blanket 2 Parts Zone with Remote Control
- Body contouring sauna blanket infrared heating body shaper
- body shaper sauna blanket weight loss
- weight loss thermal wrap electric body shaper sauna
- portable slimming body wrap electric body contouring blanket
- Biotronix Far Infrared Detox Blanket 2Zone Spa Sauna Heating FIR Technology Zipper Type Digital
- Biotronix Full Body Heating Blanket Far IR Infrared 2 Zone/Parts Weight Loss Body Detox
- Remote controlled body slimming blanket body sculpting thermal wrap
- Hydrafacial System with Advanced Features Skin Care Device for Deep Cleansing and Hydration
- Anti-Aging Facial Machine HydraFacial for Pore Cleansing Salon Hydra Facial Machine
- Hydrafacial Machine for Sensitive Skin Skin Renewal Machine
- Skin Treatment Machine for Spa Home Use Dermabrasion Device
- Hydra Facial Machine Supplier
- Hydrafacial Beauty Machine Advanced Skin Care Device Facial Treatment System
- Portable 7 In 1 Hydro Peel Facial HydraFacial Machine Hydra Facial Therapy
- Hydrafacial System with Vacuum Suction Technology Facial Treatment Equipment for Skincare Clinics
- Hydra Facial Machine
- Hydrafacial System with Oxygen Infusion Facial Treatment Equipment for Home Spa
- Hydrafacial Equipment with Multiple Functions Skin Treatment Machine for Spa
- 7 in 1 Hydrafacial Machine Hydra Facial Microdermabrasion Double Motor Salons/Parlour & Skin Clinics
- Hydrafacial Machine with Vacuum Extraction Skin Care Device with Collagen Boosting
- Skin Care Machine for Acne and Scars Portable Hydrafacial System for Home Use
- Facial Treatment Device for Hyperpigmentation Professional Skin Care Treatment Machine
- Facial Machine for Professional Estheticians Hydrafacial Device with Infusion Serums
- Hydra Facial Machine Deep Skin Cleansing Machine China Beauty Equipment
- HydraFacial Machine Vacuum Blackhead Remover 7 In 1 H2o2 Hydra Facial Machine
- Hydrafacial Device for All Skin Types Facial Skin Care Equipment for Home
- Hydrafacial Beauty Machine
- Semi-Automatic 7 In 1 Face HydraFacial Machine, Normal Skin
- Hydrafacial Device with Ultrasonic Technology Facial Machine for Skin Detox
- Hydrafacial Machine with Microdermabrasion Features Facial Treatment Equipment with LED Light
- Professional Hydrafacial Dermabrasion Machine Esthetician Equipment for Home, Spa
- Hydra Facial Machine for Deeply Skin Cleaning China Beauty Equipment, Hydrafacial Machine
- Skin Lifting and Tightening Vacuum Therapy Body Massage Cupping Machine
- Vacuum Therapy Lift Machine Body Shaping Massage Cups for Chest Enlargement
- ABS Plastic Vacuum Therapy Machine, For Clinical, Electrotherapy equipment
- Vacuum Cupping Scraping Machine Strong Suction Pump Massage Cup with 32 Cups
- Slimming and Toning Machine Cavitation RF Vacuum Slimming
- Ultrasonic Cavitation Rf Vacuum Machine
- Slimming Equipment Body Shaping Fat loss Device Cavitation RF Vacuum Weight loss Device
- Professional Grade Skin Care Equipment Facial Equipment with Anti-Aging Features
- Professional Facial Machine for Estheticians Hydrafacial Device for Skin Exfoliation
- Hydrafacial Equipment for Anti-Aging Facials Skin Care Device with Hydration Therapy
- Chest Cup Enhancement Sucking with a Vacuum Therapy Machine Nursing Chest Enhancement
- Biotronix Infrared Sauna Blanket Body Detox Professional Therapy Sweating Sauna Bed Pain Relief
- Biotronix Far Infrared Heat Sauna Blanket,Portable Sauna Bag Blanket Weight Loss and Physiotherapy
- Biotronix Sauna Thermal Blanket Infrared Blanket Thermal Blanket Beauty Detoxification Weight Loss
- Biotronix Portable Sauna Heating IR Blanket 2 Zone with Remote Digital Full Body Sweating Blanket
- Biotronix Slimming Equipment Far Infrared Heating Blanket 2 Zone Parts Digital Full Body Therapy
- Biotronix Infrared Sauna Blanket Foldable Sauna Fitness Blanket Steam Sauna Blanket Slimming Blanket
- Vacuum Cavitation Slimming Device RF LIPO LASER Fat Blaster
- RF Cavitation Fat Reduction LIPO LASER and RF Slimming
- Vacuum Cavitation Fat Reduction RF and LIPO LASER Body Contouring
- Cavitation RF Weight Loss System LIPO LASER and Vacuum Slimming Machine
- RF Cavitation Fat Loss Machine LIPO LASER and RF Fat Reduction
- 5 in 1 lLIPO Cavitation Ultrasonic Cavitation RF Vacuum Slimming Fat Loss Device
- Cavitation RF Slimming Equipment LIPO LASER and RF Body Sculptor
- Cavitation RF Weight Loss Treatment LIPO LASER Body Shaping Device
- LIPO LASER Slimming Equipment Cavitation RF Weight Loss
- Cavitation RF Fat Blaster Machine Vacuum RF and LIPO LASER Slimming
- LIPO LASER Fat Reduction Cavitation RF Fat Loss Device
- Fat Loss Cavitation Machine RF Body Slimming Device
- Biotronix Ultrasonic Cavitation Machine with Radio Frequency RF Vacuum LIPO LASER Weight Loss
- Lipo Lipolaser Weight Loss Equipment
- Vacuum 5 in one Slimming Center Machine
- Biotronix 6 in 1 Cavitation Multipolar RF Vacuum and Laser Therapy Slimming Machine
- Body Shaper Automatic Cavitation RF Vacuum Therapy Imported Slimming Equipment, For Weight Loss
- Ultrasonic Cavitation Machine Face RF Body Rf Tripolar Rf Vacuum Rf 5 in one Slimming Device
- 5 in 1 lLIPO Cavitation Ultrasonic Cavitation RF Vacuum Slimming Fat Loss Device
- RF Cavitation Fat Loss Machine LIPO LASER and RF Fat Reduction
- Hydrafacial Equipment with Multiple Functions Skin Treatment Machine for Spa
- Hydrafacial System with Skin Rejuvenation Dermabrasion Facial Machine with LED Light
- Hydrafacial System for Skin Care Professionals Skincare Device for Home Use
- Hydrafacial Equipment for Spas Hydrafacial Machine Wholesale Skin Hydration Facial Machine
- Compressible Limb Therapy System
- Top choice pneumatic compression therapy system for hospitals
- 50-60 Hz 20-250 Mmhg Compression Limb Therapy Device 6 Chamber, For Hospital
- 50-60 Hz 20-250 Mmhg 6 Chamber DVT Air Compression Therapy System Fullset, For Hospital
- Pneumatic Compression Pumps, Lymphedema Pumps, Edema Pumps 2,450Get Latest Price
- INTERMITTENT PNEUMATIC COMPRESSION SYSTEM DVT TREATMENT PUMP
- Rectangular ABS Plastic VENOUS INSUFFICIENCY COMPRESSION PUMP LYMPHATIC SYSTEM SUPPORT DEVICE, Universal Size, Reusable
- Plastic Flexible DVT System for Compression Leg Therapy (DVT Manual, 4-Chamber), For Pain Relief, Reusable
- Exercise Equipment Dvt Pump Sequential Compression Device, Universal Size, Reusable
- DVT manual blood circulation device Air compression therapy for lymphedema
- Air compression leg and foot massager machine air compression therapy
- DVT Management Device Compression Therapy Equipment
- Air Compression Therapy Blood Circulation Compression Therapy Device Diabetic Foot Ulcers Treatment
- Compressible Limb Therapy System DVT Prevention for Home Use
- DVT Best performing four-chamber pneumatic compression device for hospitals
- Physical Equipment Compression Therapy Sleeve Rehabilitation Recovery Massager Dvt Pump
- ABS Plastic Rectangular Pneumatic Compression Dvt Pumps Physiotherapy
- Foot Wound Healing Edema Reduction DVT Pump 4 Chamber Air Compression Therapy with Mild Pressure
- Plastics ABS Portable Dvt Pumps 4 Chamber Digital
- Four-Chamber DVT Compression Pump Manual Analogue Model: Small and Lightweight
- Air Therapy Recovery Device, Lymphatic Drainage Machine Pneumatic Compression for Edema Relief
- ABS Plastic Rectangular 6 Chamber DVT Machine, Adjustable Compression
- DVT 4-Chamber Compression Therapy Device for Home Care
- Niscomed Lymphedema DVT Pump Compression Therapy Pump for Hospital Use
- ABS Plastic Rectangular DVT device for Spider vein Varicose Vein, Adjustable Compression
- ABS Plastic Rectangular Biotronix DVT Compression Pump, For Hospital And Clinic, For Chronic Venous Insufficiency
- Biotronix Sequential 6Ch Compression Air Therapy Pneumatic Digital 6 Modes Remote Arm Legs Waist
- DVT Compression therapy machine Four-chamber pneumatic device
- Lymphatic Drainage Pump for Circulation DVT Compression Therapy Equipment
- Air compression therapy recovery boots with six chambers
- Vascular Health Support Leg Edema Management Vein Health Maintenance DVT Compression System
- dvt Leg Massager with Air Compression Relax Air Compression Therapy
- dvt Compression therapy system DVT compression arm
- DVT compression device 4 chamber air therapy system Digital therapy for legs and arms
- Dvt Compression Therapy Manual
- Plastics ABS Portable Digital DVT Compression
- Plastic round Four-Chamber DVT Compression Pump for Improved Blood Flow, For Body Relief
- dvt Digital compression therapy DVT therapy legs 4 chamber DVT leg pump
- DVT prevention Compression therapy Mobile pump Travel pump Leg pump
- Plastic Circular Physiotherapy Pressotherapy DVT PUMP air compression Therapy
- DVT therapy for arms Compression air therapy system 4 chamber DVT device
- Air Compression Therapy Leg Massager Gym Leg Massager Airbags
- Leg Compression Therapy Machine DVT Prevention Pump
- Leg Compression Pump System Improve Circulation, Reduce Swelling, and Sports Recovery
- Sequential Compression Therapy Device
- Air compression therapy recovery boots with six chambers compression therapy
- Biotronix 6 Chamber Lymphedema Pump Digital With Remote with 2 Legs Cuffs ,1 Arm Cuff ,1 Waist Cuff
- Intermittent Pneumatic Compression Device DVT Prevention Pump 6 Chamber
- DVT Prevention Device Therapeutic Compression Massager Home Use Compression Pump
- Air Pressure Machine to Help Blood Circulation with 4 Chamber Leg Cuff
- Digital DVT compression therapy 4 chamber DVT air therapy Compression therapy for legs
- 50-60 Hz 20-250 Mmhg Biotronix Digital Automatic DVT Air Compression Therapy Pneumatic, For Hospital, Six Chamber
- Home DVT prevention device Lymphatic drainage compression machine
- White Vibrating Airbags Air Pressure Leg Massager With Air Compression Therapy 4 bags, For Gym
- Lymphatic circulation Aid DVT Prevention and Treatment Lymphatic System Support Pump
- Biotronix Intermittent DVT Deep Vein Thrombosis Prophylaxis Compression Therapy Boots
- Sequential Limb compression Therapy Air Pressure 4 Chamber
- Pneumatic compression device Hospital compression device Four-chamber therapy device
- Pneumatic Compression Therapy Devices 4 Chamber for DVT PUMP
- Compressible Limb Therapy System
- 4 Chamber DVT Pump Air Compression Therapy Machine
- Black Relax Air compression leg and foot massager machine air compression therapy, For Household, (w) 18
- DVT Digital Pump Chinese 4 Chamber Lymphatic Drainage Device
- Air compression therapy recovery boots with six chambers compression therapy
- Air compression therapy recovery boots with six chambers compression therapy
- Compression Pump for DVT Lymphatic Drainage Pump Air Therapy for Lymphedema
- Circulation Enhancement Therapy Compression for Venous Insufficiency Leg pump for swelling
- Pneumatic Sequential Lymphatic Drainage Lymphedema Pressotherapy Compression Air Therapy 6 Chamber
- Osteopathy Tables
- Cavitation Machine
- Tattoo Removal Device
- Digital Algometer
- Equipment Trolley
- Beauty Rolling Trolley Cart Equipment Mobile Salon Spa Service Instrument Hairdressing Storage Tray
- Biotronix Suspended Gait Trainer Device Electrical Partial Body Weight Support System
- Plastic Trolley Movable with Storage Tray & Wheels for Physiotherapy Machines
- Rolling Trolley Cart Beauty Salon SPA Storage Equipment Machine Organizer Stand
- Desktop Beauty Salon Trolley Beauty Equipment Trolley Beauty Machine Trolley
- Fully Automatic Hair Salon Equipment Furniture Beauty Equipment Trolley Cart Equipment
- Multi-functional beauty cart Beauty salon mobile workstation
- Beauty Salon Multipurpose Cart A Beauty Mobile Trolleys With Storage Space
- Beauty Salon Nail Salon Trolley
- Professional Salon Hairdressing Trolley Imported China Hair Salon Equipment Trolley
- White Plastic Multifunctional Makeup Trolley with Wheels for Spa Beauty Instruments
- Trolley ABS Space Saving Beauty Trolley Stand Rolling Salon Cart Holder Spa
- Beauty salon trolley with integrated cable management
- Trolley Movable And Height Adjustable Trolley For Hydra Machine For Professionals
- Beauty salon mobile trolley Salon tools organizer with wheels
- Hair salon utility trolley with storage Beauty equipment trolley with shelves
- Salon cart for beauty supplies Professional beauty salon storage cart
- Salon trolley with adjustable trays Hair salon equipment mobile trolley
- Beauty salon tool caddy
- Trolly Portable salon equipment cart Automatic salon furniture
- Salon cart for beauty salon tools and equipment
- Wheels With Storage Tray 30 kg Capacity Imported Trolley Stylish Design
- Beauty salon tools organizer Salon equipment transport cart
- Equipment Trolley Physio Equipment Beauty Salon Foldable Trolley
- Beauty Storage Cart Hair Dryer Holder Stylist Equipment Lockable Rolling Wheels Salon Trolley
- Storage Trolley Cart Serving Trolley ABS Plastic Steel Universal Wheel Storage Basket Beauty Salons
- Fully Automatic Trolley Cart beauty Salon Equipment
- Multifunctional Medical Cart Medical Cart Wheels Beauty Machine Trolley Spa Trolley for Beauty Salon
- Beauty Equipment Salon Trolley Case Beauty Spa Trolley Cart Bracket Instrument
- Salon utility trolley with organizers
- Beauty Cart,Salon Rolling Trolley Cart with Wheels Trolley Stand Beauty Instrument Tray Rolling Cart
- Salon equipment cart with handle
- Beauty Rolling Pull Cart Trolley Stand For Beauty Equipment Salon Beauty Trolley For Spa 4 Wheels
- Trolley Beauty Salon Equipment Furniture Hospital Equipment Machine Trolley Simple Storage Tray
- Salon trolley with compartments Hair salon equipment organizer
- Beauty salon rolling trolley with trays Salon equipment organizer with drawers
- Salon trolley for hair salon equipment Hair salon tools organizer trolley
- Professional Salon Hairdressing Trolley
- Professional Beauty Salon Furniture High Quality White Trolley
- Rolling Trolley Salon Pedestal Rolling Cart Beauty Salon Wheel Trolley ABS for Hair Salon
- Beauty Instrument Tray Rolling Salon Spa Service Trolley with Metallic Pillar ABS Tray with Wheels
- Salon trolley cart with wheels
- Wheels With Storage Tray 30 kg Capacity Imported Trolley Stylish Design
- Storage Trolley Cart Serving Trolley ABS Plastic Steel Universal Wheel Storage Basket Beauty Salons
- Rolling Trolley Cart Beauty Salon SPA Storage Equipment Machine Organizer Stand
- Portable Metal White Facial Cosmetology Spa Beauty Salon Rolling Trolley with Storage Tray Wheels
- Beauty salon trolley with cable management system
- Infrared Thermometer
- Spine Therapy Device
- Shockwave Therapy Machine and Equipment
- Low-energy Shockwave Therapy Digital Touch Screen ESWT Device Orthopedic Conditions Rehabilitation
- Shock Wave Device Portable Shockwave Therapy Chronic Pain Relief
- Shock wave therapy for long-lasting pain relief
- Touch screen shock wave therapy equipment for comprehensive pain relief
- Physiotherapy Rehabilitation Radial Shock Wave Device ED Treatment Portable Shockwave Therapy Device
- Home Use ED Treatment ESWT Radial Shock Wave Device Physiotherapy
- Physiotherapy Rehabilitation Radial Shock Wave Device Treatment
- Radial Shockwave Therapy for Chronic Pain Management
- Electromagnetic Shockwave Therapy Portable ESWT Device Digital Touch Screen
- Electromagnetic ESWT Machine Muscle Recovery Physiotherapy Home Use
- Shockwave Therapy Tool Extracorporeal ESWT Radial Shock Wave Shock Wave Device Portable
- Home Use Rehabilitation ED Treatment ESWT Radial Shock Wave Device, For Hospital Shockwave Therapy
- Shockwave Therapy Device Home Use Pain Relief Portable ESWT Treatment, For Clinical Purpose
- Shockwave Therapy for Rehabilitation Portable Digital Touch Screen Physiotherapy
- Shockwave Therapy Machine SW18 Model, For Hospital Portable digital pain relief device
- Digital Touch Screen ESWT System Pain Relief Rehabilitation Home Use Treatment
- Extracorporeal ESWT Radial Shock Wave Shock Wave Device Portable Shockwave Therapy
- Portable Digital Touch Screen Physiotherapy Treatment, For Hospital, Radial shockwave therapy
- Electromagnetic Shock Wave Device Rehabilitation Treatment Physiotherapy Portable Touch Screen
- Physiotherapy ESWT Shockwave Therapy for Physiotherapy ED Treatment
- Digital therapy Portable mini shockwave therapy device with digital interface
- Radial Shock Wave Device Physiotherapy ED Treatment ESWT Electromagnetic Portable
- Shockwave Therapy Machine for Physical Therapy Clinics
- ESWT Radial Shockwave Therapy Device
- Portable Digital Touch Screen Treatment Physiotherapy Electromagnetic ESWT
- Shockwave Therapy for Physiotherapy Portable Digital Touch Screen
- Shockwave Therapy Device Adjustable Intensity Orthopedic Conditions, For Hospital
- Portable Shockwave Therapy, For Hospital
- Portable Electromagnetic Shockwave Therapy
- Portable Digital Touch Screen Physiotherapy Rehabilitation
- SW18 Shockwave Therapy Device
- Low-energy Shockwave Therapy Digital Touch Screen ESWT Device
- Portable ESWT System Digital Touch Screen Shockwave Equipment, For Hospital
- ESWT Unit Professional Grade for Targeted Orthopedic Relief Shockwave Therapy
- LCD Digital Touch Screen ESWT System Sports Injuries Muscle Recovery
- Electromagnetic Shockwave Therapy Machine Home Use Pain Relief, For Hospital
- Home use Pain relief Portable ESWT Machine Pain Relief Rehabilitation Digital Touch Screen
- PalsticABS Portable Digital Touch Screen Physiotherapy Rehabilitation
- Digital Touch Screen ESWT Device Electromagnetic Shockwave Therapy System Portable ESWT Machine
- Shock wave therapy for lower back pain treatment
- Shockwave Therapy Unit Orthopedic Conditions Pain Relief Portable
- Shockwave Treatment Portable Device Digital Touch Screen ESWT Treatment, For Hospital
- Physiotherapy Equipment with Digital Touch Screen
- Portable Shockwave Therapy Digital Touch Screen Physiotherapy, For Hospital
- Physiotherapy ED Treatment ESWT Electromagnetic Portable shockwave Therapy
- Home Use Physiotherapy Rehabilitation Radial Shock Wave Device ED Treatment
- Plastics ABS Exercise Equipment Electrical Motorized Rehabilitation Pedal Cycle Motorized, Manual
- Physiotherapy, Digital Touch Screen Shockwav,e Equipment
- PalsticABS Portable ESWT Machine Pain Relief Rehabilitation Digital Touch Screen
- Physiotherapy Radial Shockwave Machine
- Shockwave Therapy for Physiotherapy Portable Digital Touch Screen, For Hospital
- Shockwave Therapy Portable Device Home Use Adjustable Intensity, For Clinical Purpose
- shockwave therapy with touch screen for sports therapy for home rehabilitation
- Digital therapy Portable shockwave therapy machine with therapeutic benefits
- Shockwave Pain Relief Rehabilitation Portable Digital Touch Screen Physiotherapy Rehab Device
- Touch screen shock wave treatment device for home and clinic Shockwave Therapy
- SW18 Shockwave Therapy for Physical Therapists
- Pain relief Portable shockwave therapy machine for home use
- Shock wave therapy devices for professional pain clinics
- Physiotherapy Portable Touch Screen, For Clinical Purpose for Shockwave Therapy
- shockwave therapy for physical rehabilitation Portable mini shockwave therapy device with physical therapy benefits
- Pain relief Physiotherapy Rehabilitation Radial Shock Wave Device ED Treatment
- touch screen shockwave therapy for personal use Digital therapy machine
- Portable Shockwave Therapy System Tendonitis Treatment Digital Touch Screen
- ESWT Machine Muscle Recovery Physiotherapy Home Use
- Shock Impulse Waves Therapy Machine for ED & Pain Relief,Multi-Functional Touch Screen
- Electromagnetic ESWT for Enhanced Recovery Shockwave Therapy Machine
- Touch Screen Physiotherapy ESWT Home Use ED Treatment Physiotherapy Portable Shockwave Therapy Device
- Portable mini shockwave therapy device for home pain relief
- Shock Wave Machine Touch Screen Therapy Pain Relief Treatment
- Shockwave for Pain Shockwave Therapy Equipment, For Hospital Shockwave Therapy Machine
- Portable Shockwave Therapy Device Digital Touch Screen ESWT Equipment
- Shockwave Therapy Equipment Portable ESWT Treatment, For Hospital
- Mini Shockwave Therapy Portable ESWT System Digital Touch Screen Shockwave Equipment, For Hospital
- Automatic Legs And Arm Stroke Recovery Biotronix Solution Forever Electrical Motorized
- Low-energy Radial Shockwave Therapy
- Shockwave Therapy Portable Device Tendonitis Treatment Digital Display SW 18 Touch Screen
- Shockwave Touch screen shock wave therapy machine for precise and effective therapy
- Touch Screen ESWT Unit Professional Grade for Targeted Orthopedic Relief
- Home Use Rehabilitation ED Treatment ESWT Radial Shock Wave Device, For Hospital, Model Name/Number: Shockwave Therapy
- Compact Shockwave Therapy Portable Mini Compact Touch Screen Equipment
- Rehabilitation Shockwave Machine with Digital Screen
- Electromagnetic Shockwave Therapy Portable ESWT Device Digital Touch Screen Shockwave Machine
- Shock Wave Device Portable Digital Touch Screen Model
- Digital Touch Screen Shockwave Therapy Machine Electromagnetic ESWT System for Rehabilitation
- Rehabilitation with Digital Shockwave Therapy Machine
- Portable Shockwave Therapy Unit Orthopedic Conditions Pain Relief
- Electronic Computerized Advanced Shockwave Therapy for Treatment and Physiotherapy
- Biotronix Solution Forever Shockwave Therapy Portable Mini Compact Touch Screen
- Full Body 7 Heads ED Treatment Physiotherapy Shockwave Therapy
- Electromagnetic ESWT SW18 Portable Shockwave Therapy Equipment, For Hospital
- Shockwave Therapy Machine SW18 Model, For Hospital
- Home Use Physiotherapy Rehabilitation Radial Shock Wave Device Treatment
- Shockwave Therapy for ED Treatment Physiotherapy Rehabilitation SW18 Model Electromagnetic
- Electromagnetic Equipment Portable ESWT Treatment, For Hospital Shockwave Therapy
- Focused Shockwave Therapy Multifunctional Extracorporeal Shockwave Device ED & Pain Relief
- Digital Touch Screen Shockwave Therapy Equipment for Rehabilitation Centers
- Radial Shock Wave Device ED Treatment ESWT Shockwave Therapy touch screen Model
- Portable Shockwave Therapy Touch Screen 7 Heads 10 Treatment Area
- Extracorporeal ESWT Radial Shock Wave Shock Wave Device Portable Shockwave Therapy Device
- Shockwave Physiotherapy Rehabilitation Radial Device ED Treatment
- Shockwave Therapy Machine for Physiotherapy Treatment
- Radial Shock Wave Device Rehabilitation Treatment
- Electromagnetic Extracorporeal Shockwave Machine
- Shockwave Therapy Equipment
- Pneumatic Shockwave Therapy Machine
- ESWT Device Electromagnetic Shockwave Therapy System Portable ESWT Machine
- Extracorporeal Shock Wave Machine Touch Screen Therapy Pain Relief Treatment
- Extra Corporal Shockwave Therapy Digital Touch Screen Physiotherapy Compact Mini Model
- Digital Touch Screen ESWT Unit Professional Grade for Targeted Orthopedic Relief
- Digital Touch Screen ESWT Unit Home Use Sports Injuries Muscle Recovery
- shockwave therapy for healthcare Physical therapy Rehabilitation Clinical use
- Physiotherapy Home Use Electromagnetic ESWT Machine Muscle Recovery
- Digital therapy Portable shockwave therapy machine with therapeutic benefits
- Digital therapy Portable shockwave therapy machine with therapeutic benefits
- Electrotherapy Device
- Physiotherapy Machine Accessories
- Home Page
- Company Profile
- Contact Us

- Home
- Products
- Physiotherapy Equipment
- KM-580 Advanced Biofeedback Therapy Device for Stroke and Paralysis Rehabilitation
KM-580 Advanced Biofeedback Therapy Device for Stroke and Paralysis Rehabilitation
Price 15000 INR/ Piece
KM-580 Advanced Biofeedback Therapy Device for Stroke and Paralysis Rehabilitation Specification
- Capacity
- Occupational Therapy EMG Device Kg
- Noise Level
- Touch Display EMG Biofeedback System db
- Operating Type
- Clinical Biofeedback Equipment
- Material
- Neuro Rehabilitation EMG System
- Power Consumption
- EMG Mirror Therapy System Watt (W)
- Voltage
- EMG Game Based Therapy Machine Statampere (sA)
- Function
- Post-Stroke Hand Rehabilitation Equipment
- Warranty
- Advanced Physiotherapy Biofeedback System
- Magnification Power
- PC Connectivity Physiotherapy Device mm
- Size
- Physiotherapy Rehabilitation Equipment India
- Set Contains
- EMG Work and Rest Training Device
- Portable
- Digital EMG Biofeedback Machine
- Lighting Type
- Physiotherapy Machine with Report Printing
- Operation Mode
- EMG Data Recording and Analysis System
- Feature
- EMG Therapy for Neuromuscular Re-education
- Application
- NMES FES ETS Combo Therapy Device
- Wall Mounted
- Rechargeable Biofeedback Device
- Accuracy
- Post-Stroke Hand Rehabilitation Equipment mg
- Use
- Lower Limb Rehabilitation Equipment
- Suitable For Use
- Muscle Re-education Biofeedback Unit
- Power
- Shoulder and Elbow Physiotherapy Device Volt (v)
- Attributes
- Hand and Arm Rehabilitation Device
- Dimension (L*W*H)
- Physical Therapy Biofeedback Device Inch (in)
- Equipment Type
- Paralysis Recovery Device
- Storage Instructions
- Post-Stroke Hand Rehabilitation Equipment
- Color
- Hospital Grade EMG Therapy Unit
- Real-Time Operation
- Biofeedback Device with Report Printing Week
- Technology
- Swallowing EMG Therapy Equipment
- Adhesive Type
- Portable Biofeedback Device for Clinics
- Condition
- Pelvic Floor Biofeedback Training Device
- Shape
- Pain Management Therapy Device
- Recyclable
- No
- Recommended For
- Shoulder, Joints, Cervical, Back, Muscles, Nerves
- Treatment
- TENS Physiotherapy Machine
- Measuring Range
- KM-580 Biofeedback Therapy Device Rankine
- Pressure Range
- Stroke Rehabilitation Equipment
- Frequency Range
- EMG Biofeedback Physiotherapy Equipment Hertz (HZ)
- Usage
- EMG Biofeedback Physiotherapy Equipment
- Age Group
- Children, Adults, Women, Elders, Infants
- Display
- Touch Screen
- Power Source
- Electric
- Weight
- NMES FES Electrical Stimulation Device Kilograms (kg)
KM-580 Advanced Biofeedback Therapy Device for Stroke and Paralysis Rehabilitation Trade Information
- Minimum Order Quantity
- 1 Piece
- FOB Port
- New Delhi
- Payment Terms
- Paypal, Cash Against Delivery (CAD), Cash on Delivery (COD), Cash Advance (CA), Cash in Advance (CID), Cheque, Days after Acceptance (DA), Delivery Point (DP), Letter of Credit at Sight (Sight L/C), Telegraphic Transfer (T/T), Western Union, Letter of Credit (L/C)
- Supply Ability
- 1 Piece Per Day
- Delivery Time
- 1 Week
- Sample Available
- Yes
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- as per communication
- Main Export Market(s)
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- Main Domestic Market
- All India
- Certifications
- as per communication customer
About KM-580 Advanced Biofeedback Therapy Device for Stroke and Paralysis Rehabilitation
Biotronix KM-580 Advanced Biofeedback Therapy Device Stroke & Paralysis Rehabilitation
Compact, clinic-grade biofeedback system with surface EMG, ETS, NMES/FES, and TENSabuilt for neuro-rehab, pelvic health, and musculoskeletal therapy. Touch display, rechargeable battery, PC connectivity, and printable progress reports.
The KM-580 is designed for real-world physiotherapy departments and rehab centers that need objective data and engaging patient training. From post-stroke upper-limb re-education to pelvic floor retraining, it combines EMG biofeedback, electrical stimulation, and guided protocols on a bright touch interface. Therapists get clear metrics and reports; patients get simple visuals, games, and faster learning.
Key Features
-
Surface EMG biofeedback with real-time visual & audio cues
-
ETS (EMG-Triggered Stimulation) for task-specific motor relearning
-
NMES/FES & TENS modes with pre-set and custom programs
-
Touch display with intuitive workflow and therapy templates
-
Mirror & game-based training to boost patient engagement
-
Work/Rest assessments and fatigue analysis for dosing decisions
-
PC connectivity, data storage, and report printing (PDF/Excel)
-
Rechargeable battery + mainsaportable for ward and bedside use
-
Multi-patient profiles with secure data management
-
Compact, lightweight, and easy to disinfect
Clinical Applications
-
Post-stroke & traumatic brain injury upper/lower limb re-education
-
Peripheral nerve injury and muscle re-education
-
Pelvic floor rehab (stress/urge incontinence, postpartum)
-
Dysphagia/swallowing EMG training support
-
Orthopedic & sports rehab: quad/hamstring activation, shoulder, ankle
-
Chronic pain modulation with TENS
-
Hand therapy & fine-motor control with mirror/game feedback
Benefits for Clinics
-
Objective progress tracking with printable reports
-
Higher patient motivation via visual feedback & games
-
Faster setup, consistent protocols, less chair-time per session
-
Versatileacovers neuro, pelvic, MSK, and pain workflows
-
Portable for home-care programs and bedside sessions
Whatas in the Box
-
KM-580 main unit (touch display)
-
Reusable EMG leads and sEMG electrodes (starter pack)
-
Stimulation leads & electrode pads
-
USB cable and PC software/driver
-
Power adapter & carry case
-
Quick start guide + user manual
Technical SpecificationsA
EMG Biofeedback
-
Channels: 2-channel sEMG (simultaneous)
-
Input type: Differential, surface EMG
-
Bandwidth: 20a500 Hz (A10%)
-
Sampling rate: a1000 samples/sec/channel
-
Input impedance: a100 MI
-
Noise: a1 AV RMS
-
Display: RMS/Mean/Peak, bar/graph, trend line
-
Thresholds: Manual/auto with adaptive gain
ETS (EMG-Triggered Stimulation)
-
Trigger: User-set EMG threshold (per channel)
-
Delay: 0a5 s; On-time: 1a10 s; Off-time: 1a20 s
-
Safety: Auto-abort on artifact/over-threshold
Stimulation (NMES/FES/TENS)
-
Waveform: Symmetrical biphasic pulsed
-
Amplitude: 0a120 mA (500 I load)
-
Pulse width: 50a400 As
-
Frequency: 1a150 Hz (1-Hz steps)
-
Ramps: 0a5 s (up & down)
-
Duty cycles: Custom & presets (Strength/Endurance/Edema)
-
Output channels: 2 independent
Therapy Content
-
EMG tests: 2 standard (Baseline, Work/Rest)
-
Templates: a5 guided protocols (Upper limb, Lower limb, Pelvic, Shoulder, Hand)
-
Games: a6 EMG games + 10 mirror therapy modes
-
Custom programs: Save/load therapist presets
User Interface & Data
-
Display: 4.3a7" capacitive touch (anti-glare)
-
Memory: a1000 sessions; multi-patient profiles
-
Connectivity: USB to PC; report export (PDF/Excel)
-
Printing: Direct via PC software
-
Language: English (others on request)
Power & Build
-
Power: 100a240 V AC, 50/60 Hz; internal Li-ion battery
-
Battery life: 6a8 hours typical use
-
Dimensions: ~210 A 120 A 55 mm; Weight: ~0.9 kg
-
Safety: Class II, Type BF applied parts; over-current & leakage protection
-
Compliance: IEC 60601-1, IEC 60601-1-2 (EMC) or equivalent
-
Operating conditions: 10a40AC; 30a80% RH (non-condensing)
Accessories (Optional)
-
Extra EMG electrodes (adult/pediatric)
-
Pelvic floor probes (consult indications)
-
Electrode pad kits & lead sets
-
Mobile stand and protective case
KM-580 biofeedback device, EMG biofeedback physiotherapy, EMG-triggered stimulation, ETS therapy system, NMES FES stimulator, pelvic floor biofeedback, swallowing EMG therapy, stroke rehabilitation device, paralysis rehabilitation equipment, neurorehabilitation EMG, mirror therapy physiotherapy, TENS physiotherapy unit, digital biofeedback machine, rechargeable biofeedback system, PC connectivity physiotherapy, report printing rehab device, clinic grade EMG, hand therapy rehabilitation, shoulder rehabilitation device, lower limb activation device, occupational therapy EMG, physiotherapy equipment India, hospital rehabilitation equipment, portable biofeedback unit
Comprehensive Neurological Rehabilitation
The KM-580 targets post-stroke and paralysis rehabilitation through advanced EMG biofeedback and TENS physiotherapy. Its multi-functional approach aids muscle re-education for various body parts, from the shoulder and elbow to hand and pelvic floor. Combining mirror therapy and functional electrical stimulation (FES), the device supports occupational therapy goals and real-world functional improvements.
User-Friendly, Portable Design
Engineered for versatility and portability, the KM-580 offers a compact structure, making it suitable for clinics or at-home therapy. The intuitive touch screen and digital interface streamline operation, while report printing enables clear tracking of patient progress. Durable plastic construction ensures reliable usage with minimal maintenance, keeping the device suitable for repeated daily therapies.
FAQ's of KM-580 Advanced Biofeedback Therapy Device for Stroke and Paralysis Rehabilitation:
Q: How does the KM-580 biofeedback therapy device assist in stroke and paralysis rehabilitation?
A: The KM-580 uses EMG-triggered stimulation, neuromuscular re-education, and real-time biofeedback to help patients regain muscle control and strength. Its game-based therapy encourages engagement and consistent practice, vital for effective post-stroke and paralysis recovery.Q: What areas of the body can be treated using the KM-580 device?
A: The KM-580 is versatile, supporting therapy for the shoulder, joints, cervical, back, muscles, nerves, hand, arm, pelvic floor, and lower limbs, making it suitable for a wide range of rehabilitation applications.Q: When should patients use the KM-580 for optimal results?
A: For best outcomes, patients should follow their clinician's recommended therapy schedule, often involving regular sessions several times a week, customized to the stage of recovery and the targeted muscle groups.Q: Where can the KM-580 biofeedback therapy device be used?
A: With its portable and user-friendly design, the KM-580 can be used in hospital clinics, rehabilitation centers, or at home, offering flexibility for continuous therapy in both professional and personal settings.Q: What is the process for using the KM-580 therapy device?
A: The user attaches the EMG electrodes to target areas, initiates the therapy session via the touch screen, and follows on-screen instructions. Real-time biofeedback and printable reports monitor progress and help therapists adjust therapy as needed.Q: How does the EMG data and report printing benefit rehabilitation?
A: EMG data offers objective insight into muscle activity, allowing clinicians to tailor interventions. Printable reports help track patient progress over time, motivating both patients and therapists with measurable milestones.Q: What are the benefits of using KM-580 for muscle re-education?
A: The KM-580 enhances neuromuscular re-education by using interactive, evidence-based protocols. This not only helps restore motor function faster but also improves patient compliance due to engaging therapy modules and clear feedback.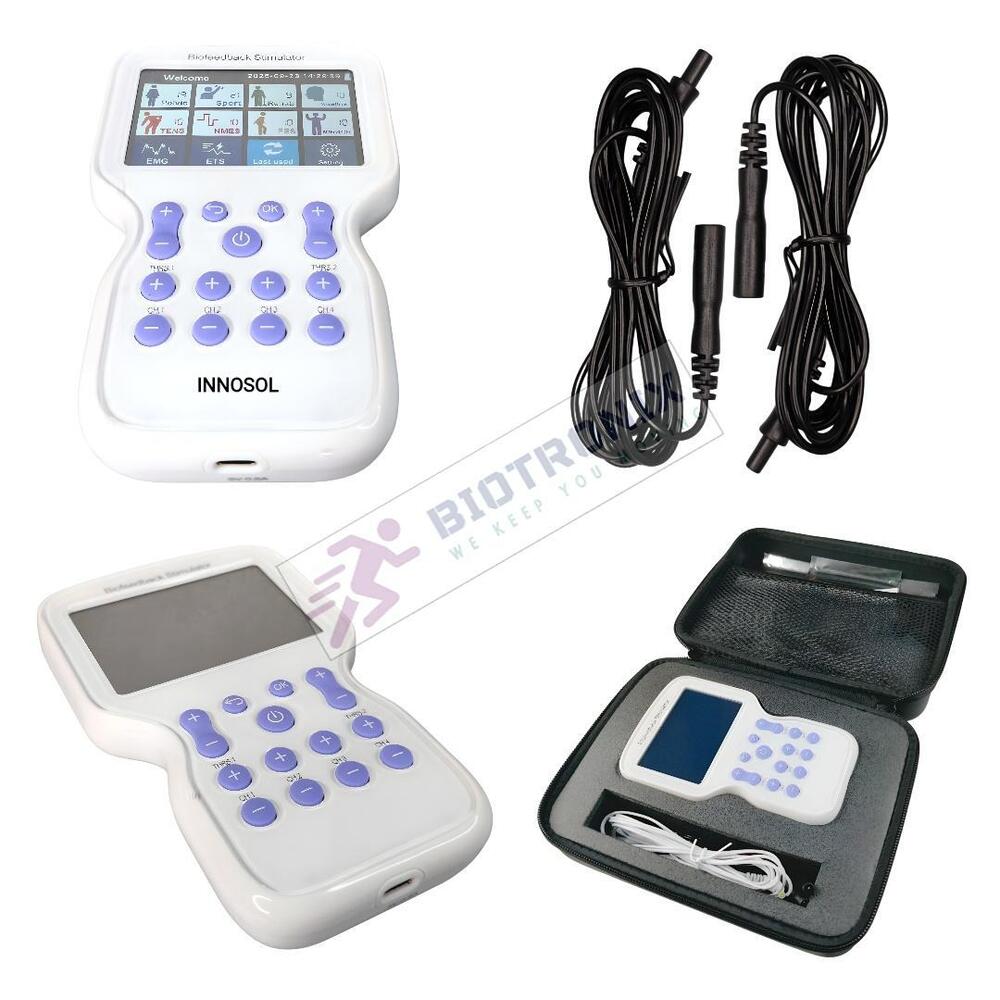
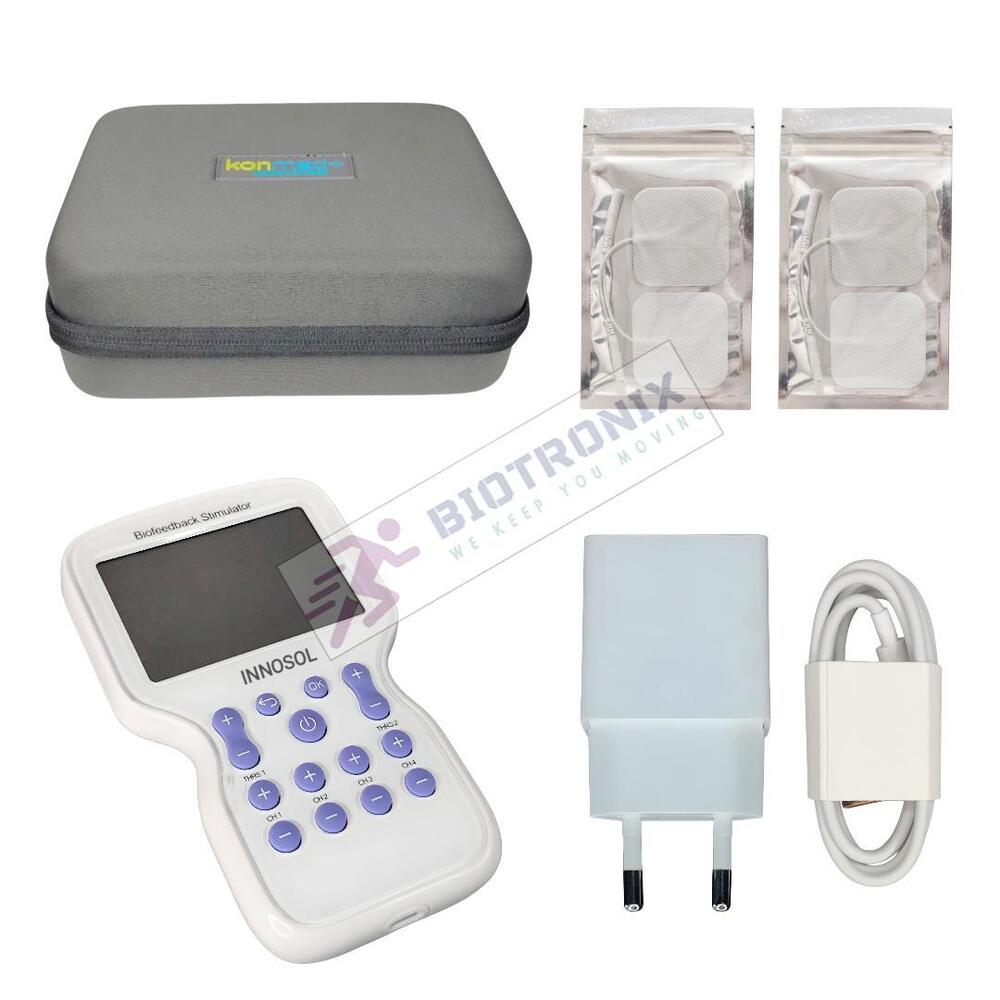
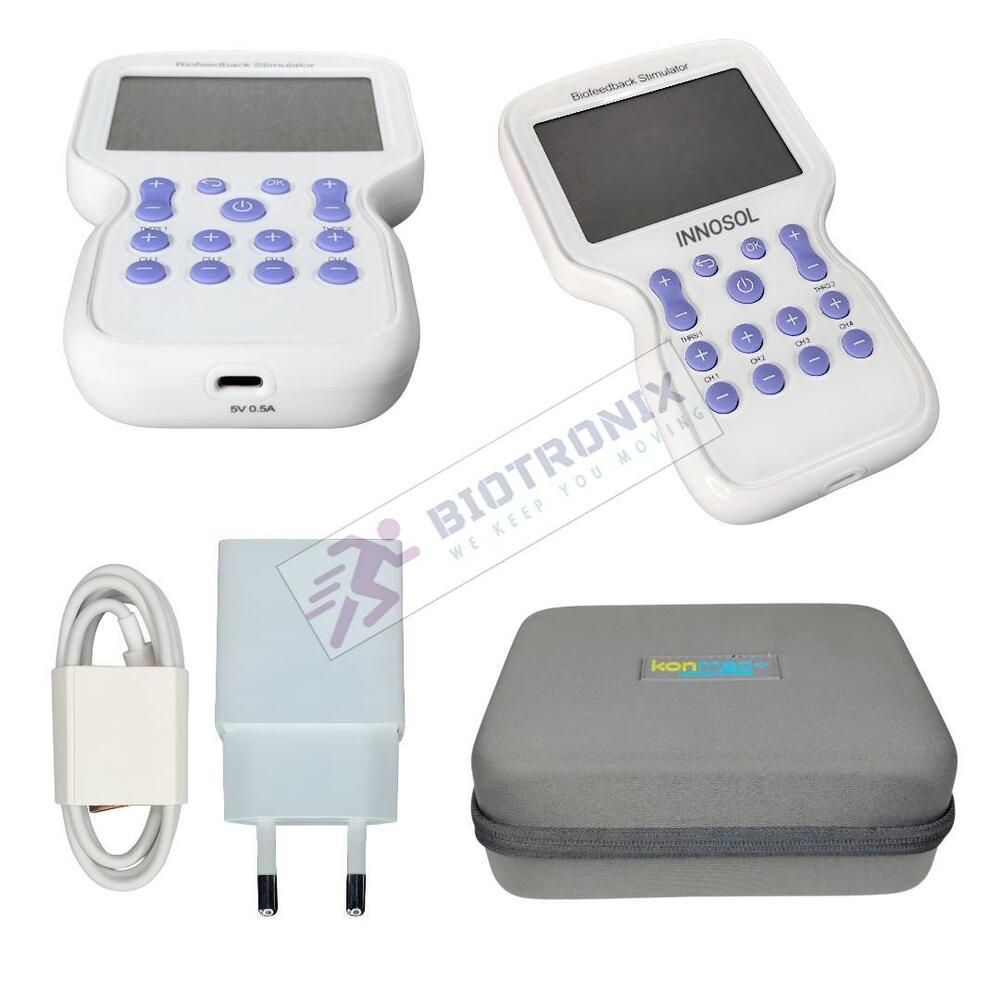

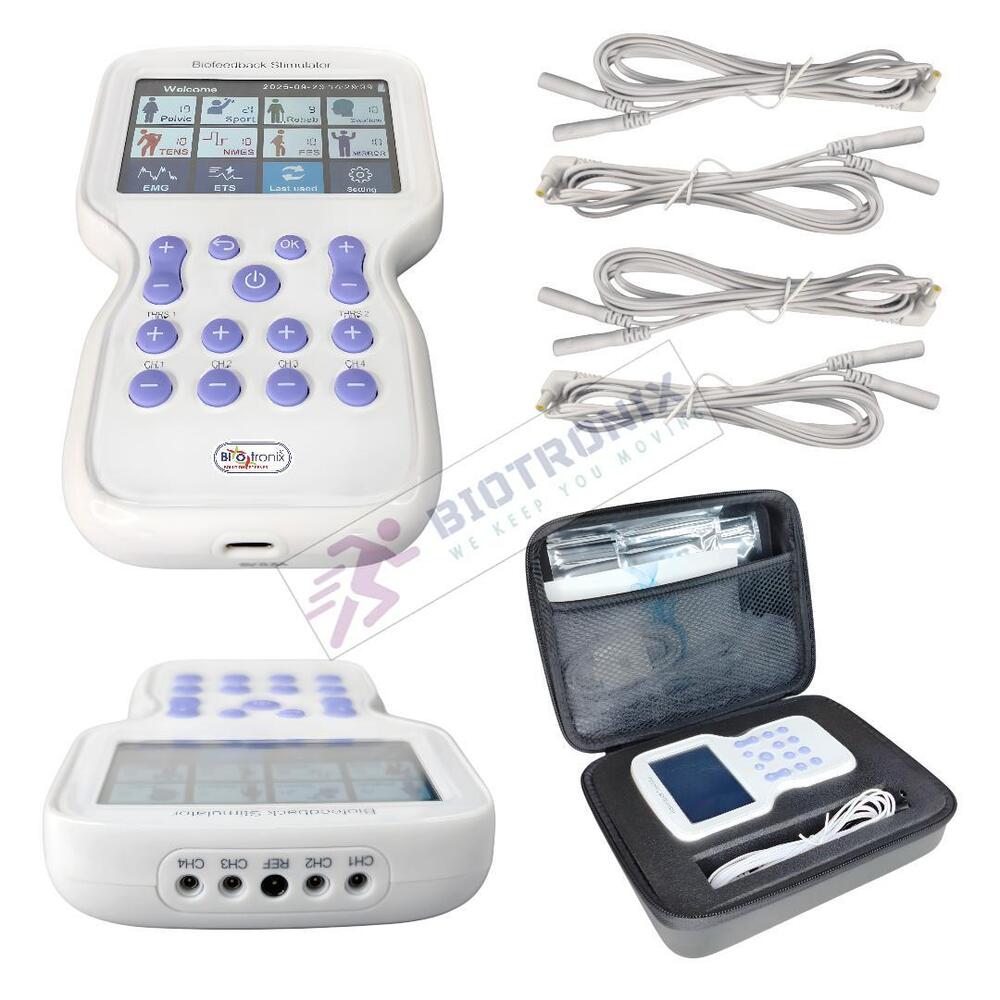


Price:
- 50
- 100
- 200
- 250
- 500
- 1000+

More Products in Physiotherapy Equipment Category
Exoskeleton Rehabilitation Systems for Strength and Control
Price 20000 INR / Piece
Minimum Order Quantity : 1 Piece
Frequency Range : Advanced wearable exoskeleton systems Hertz (HZ)
Pressure Range : Clinical exoskeleton rehab solutions
Treatment : Exoskeletonassisted physiotherapy
Age Group : Children, Adults, Women, Elders, Infants
Konmed KM-560 Electrotherapy Biofeedback Device for Physiotherapy
Price 20000 INR / Piece
Minimum Order Quantity : 1 Piece
Frequency Range : 1120 Hz Hertz (HZ)
Pressure Range : Not Applicable
Treatment : Treatment record recall system
Age Group : Children, Adults, Women, Elders, Infants
Konmed KM-560 4 Channel EMG Biofeedback System for hospital Therapy
Price 20000 INR / Piece
Minimum Order Quantity : 1 Piece
Frequency Range : 1120 Hz Hertz (HZ)
Pressure Range : Not Applicable
Treatment : Treatment record recall system
Age Group : Women, Elders, Children, Adults, Infants
Professional EMG Kegel Therapy System for Women Physiotherapy Programs
Price 25000.0 INR / Piece
Minimum Order Quantity : 1 Piece
Frequency Range : 80 Hz Hertz (HZ)
Pressure Range : 0300 mmHg
Treatment : Urinary Incontinence (Stress, Urge & Mixed)
Age Group : Elders, Adults, Children, Women, Infants






Thank You!
Thank You for your valuable time. We have received your details and will get back to you shortly.
Our Products
- Medical Equipment
- Beauty Equipment
- Slimming Equipment
- Chiropractor Tools
- Physiotherapy Equipment
- Pressure Therapy Equipment
- Therapy Machine
- Robotics Hand Rehabilitation Gloves
- Occupational Therapy Equipment
- Dvt Pump
- Bobath Tables
- Air Compression Therapy Equipment
- Finger Dexterity Board
- Aesthetic Devices And Equipments
- Weight Loss Equipment
- Compressible Limb Therapy System
- Osteopathy Tables
- Cavitation Machine
- Tattoo Removal Device
- Digital Goniometer
- Digital Algometer
- Equipment Trolley
- Infrared Thermometer
- Spine Therapy Device
- IASTM Tools
- Shockwave Therapy Machine and Equipment
- Massage Guns
- Body Massagers Device
- Chiropractic Guns and IASTM Tools
- Knee Elbow Shoulder CPM ( Continuous Passive Motio
- DVT Air Compression Therapy Lymphedema Device
- Cupping Therapy Device
- Kinesio Tapes
- Electrotherapy Device
- Evaluation ROM Measurement Devices
- Hot and Cold therapy Hydrotherapy Equipments
- LASER Therapy Equipment
- Hand Rehabilitation Glove and Devices
- Traction Therapy Device
- Treatment Examination Manipulation Tables
- Un Weigh Mobility Trainer Partial Body Weight Supp
- Sensory Motor Tools Equipment
- Physiotherapy Machine Accessories
- Hair Removal Devices
- Tattoo Removal Devices
- Hydrafacial Machines


Developed and Managed by Infocom Network Private Limited.

 Send Inquiry
Send Inquiry

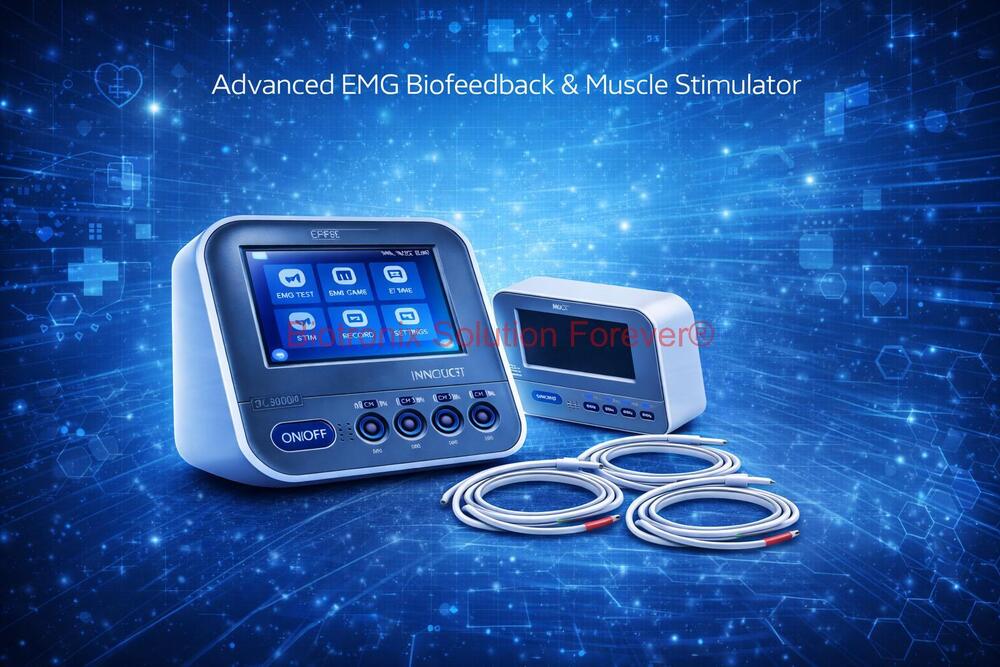

 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free